Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (6): 877-884.DOI: 10.19852/j.cnki.jtcm.20220425.002
• Research Articles • Previous Articles Next Articles
Celastrol promotes apoptotic cell death in children neuroblastoma cells through caspases dependent pathway
YANG Ye, ZHANG Aihui, LI Anmao( )
)
- Department of Thoracic Surgery, Xi 'an Children's Hospital, Xi 'an 710003, China
-
Received:2021-10-09Accepted:2022-01-21Online:2022-12-15Published:2022-04-25 -
Contact:LI Anmao -
About author:LI Anmao, Department of Thoracic Surgery, Xi'an Children's Hospital, Xi'an 710003, China. lianmaoxian@aliyun.com,Telephone: +86-298-7458067
-
Supported by:Scientific Research Project of Shaanxi Provincial Department of Health: the Protective Role of Celastrol and its Active Mechanisms in Neuroblastoma Treatment(2020A1515003129)
Cite this article
YANG Ye, ZHANG Aihui, LI Anmao. Celastrol promotes apoptotic cell death in children neuroblastoma cells through caspases dependent pathway[J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 877-884.
share this article
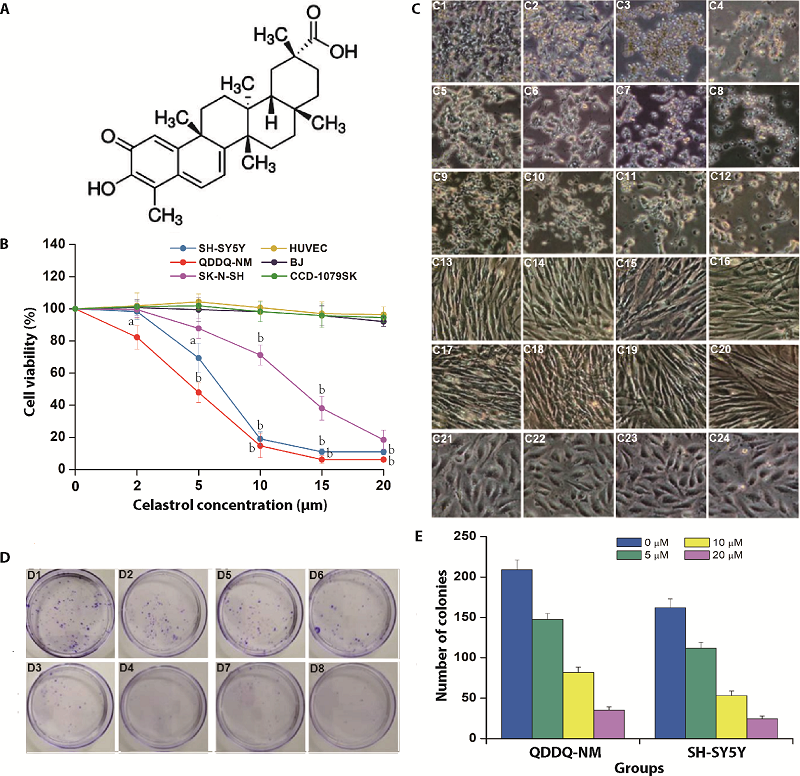
Figure 1 Celastrol inhibits cell viability and proliferation of neuroblastoma cells A: molecular structure of celastrol; B: QDDQ-NM, SK-N-SH and SH-SY5Y neuroblastoma cell lines and three normal cell lines were incubated with different concentrations (0-20 µM) of celastrol, CCK-8 assay was applied to determine cells viability. aP < 0.05; bP < 0.01 versus 0 μM celastrol treatment group; C: C1-C4: QDDQ-NM cell lines were incubated with different concentrations (0-20 µM) of celastrol; C5-C8: SK-N-SH cells were incubated with different concentrations (0-20 µM) of celastrol; C9-C12: SH-SY5Y cells were incubated with different concentrations (0-20 µM) of celastrol; C13-C16: BJ cells were incubated with different concentrations (0-20 µM) of celastrol; C17-C20: CCD-1079SK cells were incubated with different concentrations (0-20 µM) of celastrol; C21-C24: HUVEC cells were incubated with different concentrations (0-20 µM) of celastrol, cell morphology changes were detected by optical microscope. Scale bar = 100 μm; D: colony formation results after celastrol treatment (0-20 µM) in QDDQ-NM (D1-D4) and SH-SY5Y cells (D5-D8). aP < 0.05; bP < 0.01; cP < 0.001 versus control group; E: number of colonies after different concentrations of celastrol treatment 0, 5, 10, 20 µM. CCK-8: cell counting kit-8.
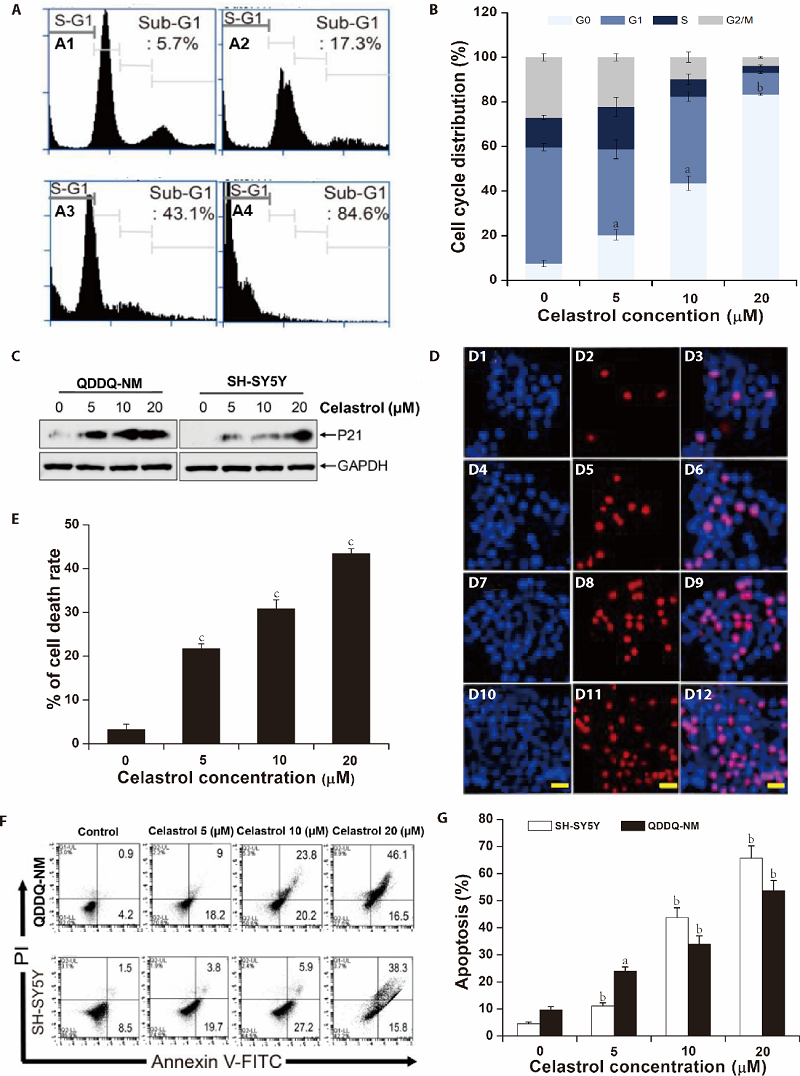
Figure 2 Celastrol inhibits cell cycle progression and induces cell apoptosis in QDDQ-NM and SH-SY5Y cells A: flow cytometry assay we applied to detect the cells cycle distribution in QDDQ-NM cells after treatment with different concentrations of celastrol (A1) 0 µM, (A2) 5 µM, (A3) 10 µM,(A4) 20 µM for 1 d; B: quantitative analysis of cell cycle distribution was shown after celastrol treatment; C: the protein expressions of P21 were detected by immunoblotting in QDDQ-NM and SH-SY5Y cells after different concentrations of celastrol treatment; D: QDDQ-NM cells with Hoechst (bright blue) and PI (red) fluorescence signals were identified as apoptotic cells, (D1, D4, D7, D10) Hoechst stanning for 0, 5, 10, 20 µM celastrol treatment; (D2, D5, D8, D11) PI stanning for 0, 5, 10, 20 µM celastrol treatment; (D3, D6, D9, D12) Merged stanning for 0, 5, 10, 20 µM celastrol treatment; scale bar = 50 μm. E: the percentage of cell death rate after celastrol treatment. F, G: flow cytometry assay was applied to determine the apoptosis of QDDQ-NM (a) and SH-SY5Y (b) cells after celastrol treatment. aP < 0.05, bP < 0.01, cP < 0.001 compared to control.

Figure 3 Celastrol facilitates apoptosis in QDDQ-NM and SH-SY5Y cells by activating caspases pathway A: mRNA expressions of Caspase-9, Bcl-2, PUMA, Bax, Noxa, and Caspase-3 were examined via RT-PCR assay; B: quantitative analysis of RT-PCR results; C-F: Western blotting and quantification results of the expressions of anti-apoptosis and pro-apoptosis proteins in QDDQ-NM and SH-SY5Y cells after celastrol treatment. G: cell viability of QDDQ-NM and SH-SY5Y were examined by CCK-8 assay after combined treatment with Z-VAD-FKM (caspase inhibitor) and celastrol. PUMA: p53 up-regulated modulator of apoptosis; RT-PCR: reverse transcription polymerase chain reaction; CCK-8: cell counting kit-8. aP < 0.05, aP < 0.01 versus control and cP < 0.01 versus celastrol treatment group.

Figure 4 ROS production was involved in celastrol-mediated apoptosis A, B: ROS production was examined under fluorescence microscope by DCFH-DA staining and the relative DCF fluorescence intensities was quantified by Imagelab; A1-A3: QDDQ-NM cells for control group, celastrol group, celastrol with NAC group; A4-A6: SH-SY5Y cells for control group, celastrol group, celastrol with NAC group; C: flow cytometry assay was also applied to assess the effect of NAC on celastrol-mediated ROS production; D: the percentage of cell viability after celastrol treatment. aP < 0.01 compared to control, bP < 0.01 compared to celastrol-pretreated group. E, F: chromatin-condensed nuclei (red arrow) indicated cell apoptosis after staining with Hoechst 33442 dye. E1-E4: QDDQ-NM cells for control group, NAC group, celastrol group, celastrol with NAC group; E5-E8: SH-SY5Y cells for control group, NAC group, celastrol group, celastrol with NAC group. The images were detected by the fluorescence microscope. Scale bar = 50 μm. The proportion of Hoechst 33442+ cells were quantified by Imagelab. G, H: Annexin V and PI were used to stain the apoptosis cells by flow cytometry. The percentage of apoptosis cells of QDDQ-NM and SH-SY5Y were quantified by Imagelab. aP < 0.01, compared to untreated cells; bP < 0.01, cP < 0.05, compared to celastrol treatment cells. ROS: reactive oxygen species; DCFH-DA: 2, 7-Dichlorodihydrofluorescein diacetate; DCF: 2′, 7′-Dichlorodihydrofluorescein; NAC: N-Acetyl-L-cysteine.
| [1] |
Polito L, Bortolotti M, Pedrazzi M, Mercatelli D, Battelli MG, Bolognesi A. Apoptosis and necroptosis induced by stenodactylin in neuroblastoma cells can be completely prevented through caspase inhibition plus catalase or necrostatin-1. Phytomedicine 2016; 23: 32-41.
DOI PMID |
| [2] |
Naveen CR, Gaikwad S, Agrawal-Rajput R. Berberine induces neuronal differentiation through inhibition of cancer stemness and epithelial-mesenchymal transition in neuroblastoma cells. Phytomedicine 2016; 23: 736-44.
DOI PMID |
| [3] | Fukuyama K, Kakio S, Nakazawa Y, et al. Roasted coffee reduces beta-amyloid production by increasing proteasomal beta-secretase degradation in human neuroblastoma SH-SY5Y cells. Mol Nutr Food Res 2018; 62: e1800238. |
| [4] | Alberto F, Mario L, Sara P, Settimio G, Antonella L. Electro-magnetic information delivery as a new tool in translational medicine. Int J Clin Exp Med 2014; 7: 2550-6. |
| [5] | Unno K, Pervin M, Nakagawa A, et al. Blood-brain barrier permeability of green tea catechin metabolites and their neuritogenic activity in human neuroblastoma SH-SY5Y cells. Mol Nutr Food Res 2017; 61. |
| [6] |
Franco-Luzon L, Garcia-Mulero S, Sanz-Pamplona R, et al. Genetic and immune changes associated with disease progression under the pressure of oncolytic therapy in a neuroblastoma outlier patient. Cancers (Basel) 2020; 12: 1104.
DOI URL |
| [7] |
Szanto CL, Cornel AM, Vijver SV, Nierkens S. Monitoring immune responses in neuroblastoma patients during therapy. Cancers (Basel) 2020; 12: 519.
DOI URL |
| [8] |
Shanmugam MK, Ahn KS, Lee JH, et al. Celastrol attenuates the invasion and migration and augments the anticancer effects of bortezomib in a xenograft mouse model of multiple myeloma. Front Pharmacol 2018; 9: 365.
DOI PMID |
| [9] |
Hong M, Wang N, Tan HY, Tsao SW, Feng Y. MicroRNAs and Chinese medicinal herbs: new possibilities in cancer therapy. Cancers (Basel) 2015; 7: 1643-57.
DOI URL |
| [10] |
Luo Y, Feng Y, Song L, et al. A network pharmacology-based study on the anti-hepatoma effect of Radix Salviae Miltiorrhizae. Chin Med 2019; 14: 27.
DOI URL |
| [11] | Zhu F, Li C, Jin XP, et al. Celastrol may have an anti-atherosclerosis effect in a rabbit experimental carotid ather-osclerosis model. Int J Clin Exp Med 2014; 7: 1684-91. |
| [12] |
Lin FZ, Wang SC, Hsi YT, et al. Celastrol induces vincristine multidrug resistance oral cancer cell apoptosis by targeting JNK1/2 signaling pathway. Phytomedicine 2019; 54: 1-8.
DOI URL |
| [13] |
Liu X, Zhao P, Wang X, et al. Celastrol mediates autophagy and apoptosis via the ROS/JNK and Akt/mTOR signaling pathways in glioma cells. J Exp Clin Cancer Res 2019; 38: 184.
DOI URL |
| [14] |
Wagh PR, Desai P, Prabhu S, Wang J. Nanotechnology-based celastrol formulations and their therapeutic applications. Front Pharmacol 2021; 12: 673209.
DOI URL |
| [15] |
Chen X, Zhao Y, Luo W, et al. Celastrol induces ROS-mediated apoptosis via directly targeting peroxiredoxin-2 in gastric cancer cells. Theranostics 2020; 10: 10290-308.
DOI PMID |
| [16] |
Hu XL, He QW, Long H, et al. Synthesis and biological evaluation of celastrol derivatives with improved cytotoxic selectivity and antitumor activities. J Nat Prod 2021; 84: 1954-66.
DOI URL |
| [17] |
Niu W, Wang J, Wang Q, Shen J. Celastrol loaded nanoparticles with ROS-response and ROS-inducer for the treatment of ovarian cancer. Front Chem 2020; 8: 574614.
DOI URL |
| [18] |
Puangpraphant S, Berhow MA, Vermillion K, Potts G, Gonzalez de Mejia E. Dicaffeoylquinic acids in Yerba mate (Ilex paraguariensis St. Hilaire) inhibit NF-kappa B nucleus translocation in macrophages and induce apoptosis by activating caspases-8 and -3 in human colon cancer cells. Mol Nutr Food Res 2011; 55: 1509-22.
DOI PMID |
| [19] |
Hong M, Almutairi MM, Li S, Li J. Wogonin inhibits cell cycle progression by activating the glycogen synthase kinase-3 beta in hepatocellular carcinoma. Phytomedicine 2020; 68: 153174.
DOI URL |
| [20] | Agidigbi TS, Kim A, Reactive oxygen species in osteoclast differentiation and possible pharmaceutical targets of ROS-mediated osteoclast diseases. Int J Mol Sci 2019; 20. |
| [21] | Rohn TT, Kokoulina P, Eaton CR, Poon WW. Caspase activation in transgenic mice with Alzheimer-like pathology: results from a pilot study utilizing the caspase inhibitor Q-VD-OPh. Int J Clin Exp Med 2009; 2: 300-8. |
| [22] | Hong M, Li J, Li S, Almutairi MM. Resveratrol derivative, trans-3, 5, 4'-trimethoxystilbene, prevents the developing of ather-osclerotic lesions and attenuates cholesterol accumulation in macrophage foam cells. Mol Nutr Food Res 2020: e1901115. |
| [23] | Lamichhane S, Bastola T, Pariyar R, et al. ROS production and ERK activity are involved in the effects of d-beta-hydroxybutyrate and metformin in a glucose deficient condition. Int J Mol Sci 2020: e1901115. 2017; 18. |
| [1] | LI Xi, LIN Xiangquan, CHEN Dongdong, LIU Hui. B-cell lymphoma-2 phosphorylation at Ser70 site-related autophagy mediates puerarin-inhibited the apoptosis of MC3T3-E1 cells during osteoblastogenesis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 27-34. |
| [2] | ZHUANG Xingxing, LIU Tao, WEI Liangbing, SONG Junmei, GAO Jiarong. Qiteng Xiaozhuo granules (芪藤消浊颗粒) medicated serum inhibits excessive proliferation and promotes apoptosis of human glomerular mesangial cells by targeting fat mass and obesity associated proteins [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1072-1080. |
| [3] | SU Chengguo, ZHAO Xiaoyan, YE Jiangnan, ZHANG Xin, JIANG Yuqing, GUO Junjie, ZHANG Xiyuan, QI Wenchuan, ZHU Jun. Effect of Tuina along “bladder meridian” alleviating intervertebral disc degeneration by regulating the transforming growth factor-β1/Smad signaling pathway in a rabbit model [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 991-1000. |
| [4] | ZHU Bin, YU Ning, WANG Lei, TIAN Yue, WU Mingfen, ZHAO Zhigang. Synergistic effect of schizandrin A and DNase I knockdown on high glucose induced beta cell apoptosis by decreasing intracellular calcium concentration [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 661-666. |
| [5] | JIANG Yiqian, ZHOU Xibin, PU Wenyuan, ZHOU Chunxiang. Sanwu Baisan decoction (三物白散) inhibits colorectal cancer progression in mice by remodeling gut microbiota and tumorigenesis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 466-473. |
| [6] | WANG Tianqi, LI Zining, CHEN Ting, CHEN Rui, JIN Ya, Oduro Patrick kwabena, ZHANG Han, WANG Yi. Zuogui (左归丸) and Yougui pills (右归丸) improve perimenopausal syndrome via regulation of apoptosis in mice [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 474-483. |
| [7] | XIE Jing, BI Zheng, WANG Sihai, SHEN Guoming, FANG Zhaohui. Danzhi Jiangtang capsule (丹蛭降糖胶囊) reduces renal injury in rats with diabetes induced by high fat diet and streptozotocin via downregulating toll-like receptor 4-nuclear factor-κB pathway and apoptosis [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 312-321. |
| [8] | DING Yajie, LIU Feng, LI Zhaoyan, Xu Yan, CAO Nida, ZHANG Guangao, WANG Rui, ZHAO Aiguang. Efficacy of luteolin on the human gastric cancer cell line MKN45 and underlying mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 34-41. |
| [9] | JIANG Wen, ZHANG Wei, ZHANG Yuxiang, YANG Hao, PAN Xiaomei, CHEN Qiang, CHEN Junhui. Tilianin extracted from Xiangqinglan (Herba Dracocephali Moldovicae) inhibits apoptosis induced by mitochondrial pathway and endoplasmic reticulum stress in H9c2 cells after oxygen-glucose deprivation/reoxygenation [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 42-50. |
| [10] | ZHENG Wei, WANG Mingxing, LIU Shanxue, LUAN Chao, ZHANG Yanqiu, XU Duoduo, WANG Jian. Buyang Huanwu Tang (补阳还五汤) protects H2O2-induced RGC-5 cell against oxidative stress and apoptosis via reactive oxygen species-mitogen-activated protein kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 885-891. |
| [11] | YANG Mengzhe, ZHANG Beibei, LIANG Zhenqiang, CHENG Nannan, Lü Anqiao, YANG Jianyu, GUO Xingzhe, BAI Xianyu, HUANG Yuanjiao, JIAO Aijun, XU Ning. Sanguinarine suppresses cell proliferation, migration and invasion in nasopharyngeal carcinoma via inhibiting mTOR signaling [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 687-692. |
| [12] | DING Jiamin, XING Yifeng, CHEN Zuoliang, CHEN Wanlu, MA Zhongxiong, XIE Yunde, ZHOU Lin. Qilan preparation (芪蓝颗粒) inhibits proliferation and induces apoptosis by down-regulating microRNA-21 in human Tca8113 tongue squamous cell carcinoma cells [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 693-700. |
| [13] | LI Miao, ZHENG Jialu, WANG Shuangshuang, CHEN Lei, PENG Xiao, CHEN Jinfang, AN Hongmei, HU Bing. Tenglong Buzhong granules (藤龙补中颗粒) inhibits the growth of SW620 human colon cancer in vivo [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 701-706. |
| [14] | YANG Liping, YU Xinglin, ZHANG Chao, CHEN Pu, DUAN Xiaohua. Yajieshaba prevents lipopolysaccharide-induced intestinal barrier injury via anti-inflammatory and anti-apoptosis [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 707-714. |
| [15] | WANG Wei, LI Qingling, MA Qiang, XIA Ran, GAO Bing, WANG Yi, WANG Jing. Effects of moxibustion at bilateral Feishu (BL13) and Xinshu (BL15) combined with benazepril on myocardial cells apoptosis index and apoptosis-related proteins cytochrome c and apoptosis-inducing factor in rats with chronic heart failure [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 227-233. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

