Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 360-370.DOI: 10.19852/j.cnki.jtcm.2026.02.009
• Original Articles • Previous Articles Next Articles
Exploring the potential mechanism of Huang’e capsule (黄莪胶囊) against spontaneous benign prostatic hyperplasia in beagle dogs using high-performance liquid chromatography-quadrupole-time-of-flight tandem mass spectrometry, gas chromatography-mass spectrometry, and network pharmacology
MA Aicui1,2,3,4, PAN Qi1,2,3,4, CHEN Dingshi1,2,3,4, WANG Zhonghui1,2,3,4, ZHENG Chiyang1,2,3,4, ZHANG Junfang1,2,3,4, YAN Jianyan1,2,3,4, LI Lei1,2,3,4, YAN Han1,2,3,4, YAO Shunheng1,2,3,4, GUO Jun1,2,3,4( ), SUN Zuyue1,2,3,4(
), SUN Zuyue1,2,3,4( )
)
- 1
Shanghai Institute for Biomedical and Pharmaceutical Technologies Shanghai 200237, China
2National Evaluation Center for the Toxicology of Fertility Regulating Drugs Shanghai 200237, China
3NHC Key Laboratory of Reproduction Regulation Shanghai 200237, China
4Shanghai Engineering Research Center of Reproductive Health Drug and Devices Shanghai 200237, China
-
Received:2025-01-13Accepted:2025-06-11Online:2026-04-15Published:2026-04-04 -
Contact:Prof. SUN Zuyue, Shanghai Institute for Biomedical and Pharmaceutical Technologies, Shanghai 200237, China. sunzy64@163.com; Dr. GUO Jun, Shanghai Institute for Biomedical and Pharmaceutical Technologies, Shanghai 200237, China. guojun@sibpt.cn; Telephone: +86-13501754505 -
About author:MA Aicui and PAN Qi are co-first authors and contributed equally to this work
Cite this article
MA Aicui, PAN Qi, CHEN Dingshi, WANG Zhonghui, ZHENG Chiyang, ZHANG Junfang, YAN Jianyan, LI Lei, YAN Han, YAO Shunheng, GUO Jun, SUN Zuyue. Exploring the potential mechanism of Huang’e capsule (黄莪胶囊) against spontaneous benign prostatic hyperplasia in beagle dogs using high-performance liquid chromatography-quadrupole-time-of-flight tandem mass spectrometry, gas chromatography-mass spectrometry, and network pharmacology[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 360-370.
share this article
| Group | n | Qmax (mL/s) | Prostate volume by B-scan (cm3) | |||||
|---|---|---|---|---|---|---|---|---|
| 0 week | 4 weeks | Difference | 0 week | 4 weeks | Difference | |||
| BPH | 7 | 3.6±1.7 | 3.6±1.3 | -0.0±0.8a | 21.5±2.4 | 21.7±1.4 | 0.1±2.6c | |
| HEC-0.04 | 7 | 3.9±1.3 | 4.6±1.2 | 0.6±1.3 | 21.5±3.4 | 19.3±3.5 | -2.2±1.4c | |
| HEC-0.16 | 7 | 3.4±1.5 | 4.8±2.4 | 1.4±1.7b | 22.3±3.4 | 16.8±2.1 | -5.5±3.7d | |
| HEC-0.64 | 7 | 3.4±1.4 | 4.8±2.9 | 1.4±2.2 | 21.5±2.9 | 16.4±1.3 | -5.1±3.3b | |
| HEC-2.56 | 7 | 3.3±1.1 | 4.0±1.4 | 0.7±1.1 | 20.9±2.5 | 14.5±1.7 | -6.4±1.8d | |
| Fina | 7 | 4.8±1.3 | 6.9±1.8 | 2.1±1.8b | 20.7±1.6 | 12.0±1.7 | -8.7±1.6d | |
Table 1 Effects on Qmax and prostate volume by B-mode ultrasonography of HEC pre- and post-administration in beagle dogs with spontaneous BPH ($\stackrel{-}{x}\pm $s)
| Group | n | Qmax (mL/s) | Prostate volume by B-scan (cm3) | |||||
|---|---|---|---|---|---|---|---|---|
| 0 week | 4 weeks | Difference | 0 week | 4 weeks | Difference | |||
| BPH | 7 | 3.6±1.7 | 3.6±1.3 | -0.0±0.8a | 21.5±2.4 | 21.7±1.4 | 0.1±2.6c | |
| HEC-0.04 | 7 | 3.9±1.3 | 4.6±1.2 | 0.6±1.3 | 21.5±3.4 | 19.3±3.5 | -2.2±1.4c | |
| HEC-0.16 | 7 | 3.4±1.5 | 4.8±2.4 | 1.4±1.7b | 22.3±3.4 | 16.8±2.1 | -5.5±3.7d | |
| HEC-0.64 | 7 | 3.4±1.4 | 4.8±2.9 | 1.4±2.2 | 21.5±2.9 | 16.4±1.3 | -5.1±3.3b | |
| HEC-2.56 | 7 | 3.3±1.1 | 4.0±1.4 | 0.7±1.1 | 20.9±2.5 | 14.5±1.7 | -6.4±1.8d | |
| Fina | 7 | 4.8±1.3 | 6.9±1.8 | 2.1±1.8b | 20.7±1.6 | 12.0±1.7 | -8.7±1.6d | |
| Group | n | Prostate volume by water displacement (cm3) | Prostate weight (g) | Prostate index (%) |
|---|---|---|---|---|
| BPH | 7 | 21.07±4.96a | 22.16±5.49a | 1.48±0.48a |
| HEC-0.04 | 7 | 20.68±4.54a | 21.17±4.82a | 1.38±0.41a |
| HEC-0.16 | 7 | 19.90±3.56a | 20.93±3.72a | 1.32±0.25d |
| HEC-0.64 | 7 | 18.16±3.65a | 18.45±3.69d | 1.34±0.31d |
| HEC-2.56 | 7 | 14.13±1.08b | 14.76±0.91b | 1.05±0.13b |
| Fina | 7 | 10.85±2.80c | 11.41±2.99c | 0.74±0.18c |
Table 2 Effects on prostate volume and prostate weight of HEC in beagle dogs with spontaneous BPH ($\stackrel{-}{x}\pm $s)
| Group | n | Prostate volume by water displacement (cm3) | Prostate weight (g) | Prostate index (%) |
|---|---|---|---|---|
| BPH | 7 | 21.07±4.96a | 22.16±5.49a | 1.48±0.48a |
| HEC-0.04 | 7 | 20.68±4.54a | 21.17±4.82a | 1.38±0.41a |
| HEC-0.16 | 7 | 19.90±3.56a | 20.93±3.72a | 1.32±0.25d |
| HEC-0.64 | 7 | 18.16±3.65a | 18.45±3.69d | 1.34±0.31d |
| HEC-2.56 | 7 | 14.13±1.08b | 14.76±0.91b | 1.05±0.13b |
| Fina | 7 | 10.85±2.80c | 11.41±2.99c | 0.74±0.18c |
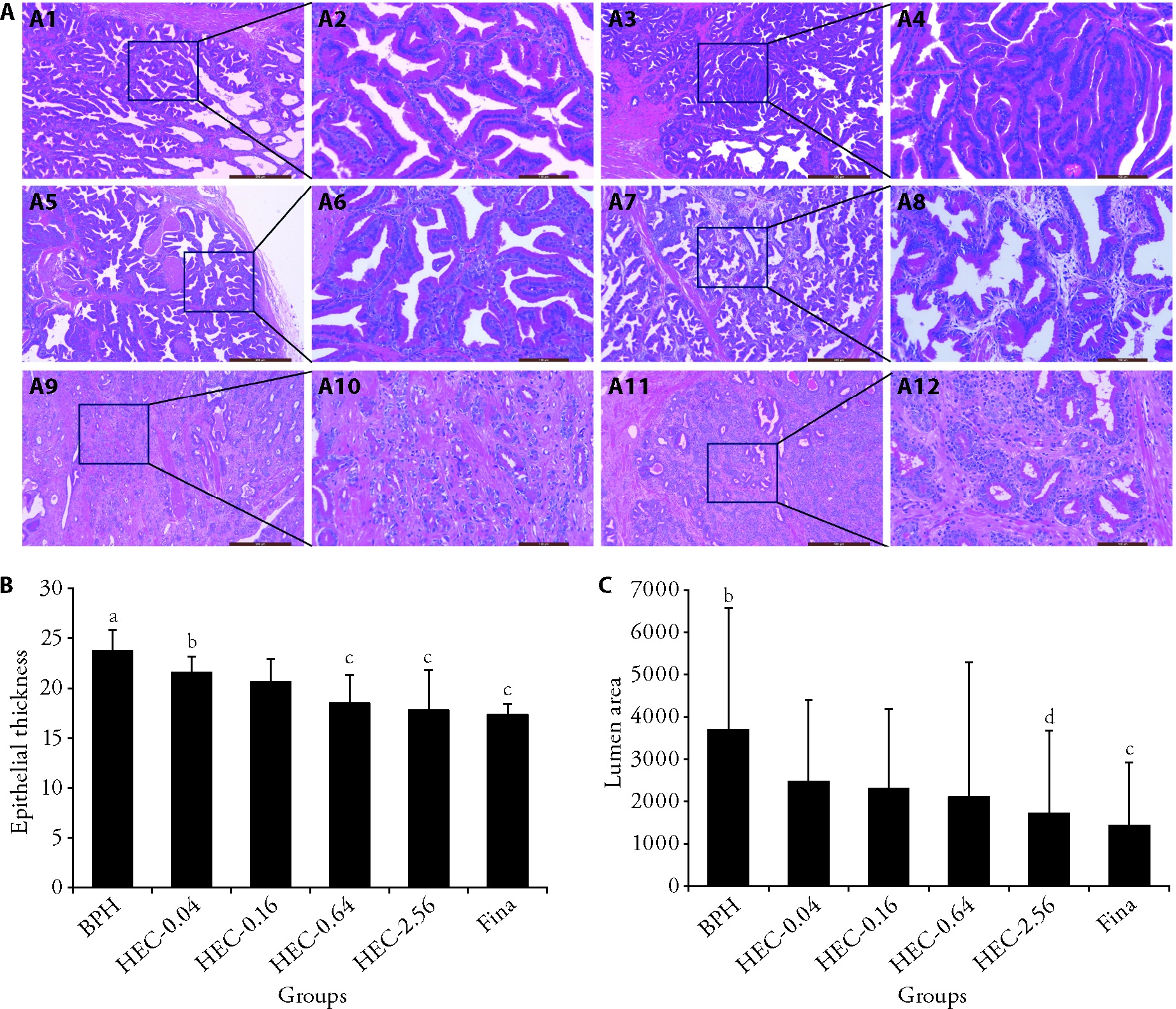
Figure 1 Effect of HEC in prostate tissues in beagle dogs with spontaneous BPH A: histopathological study of prostate tissues in beagle dogs with spontaneous BPH using HE staining; A1: BPH group (× 50, scale bar = 500 μm); A2: BPH group (× 200, scale bar = 100 μm); A3: HEC-0.04 group (× 50, scale bar = 500 μm); A4: HEC-0.04 group (× 200, scale bar = 100 μm); A5: HEC-0.16 group (× 50, scale bar = 500 μm); A6: HEC-0.16 group (× 200, scale bar = 100 μm); A7: HEC-0.64 group (× 50, scale bar = 500 μm); A8: HEC-0.64 group (× 200, scale bar = 100 μm); A9: HEC-2.56 group (× 50, scale bar = 500 μm); A10: HEC-2.56 group (× 200, scale bar = 100 μm); A11: Fina group (× 50, scale bar = 500 μm); A12: Fina group (× 200, scale bar = 100 μm); B: the epithelial thickness of the prostates in beagle dogs with spontaneous BPH; C: the lumen area of the prostates in beagle dogs with spontaneous BPH. BPH: the BPH group; HEC-0.04, HEC-0.16, HEC-0.64, HEC-2.56: 0.04, 0.16, 0.64, 2.56 g/kg HEC-treated group, respectively; Fina: 0.24 mg/kg finasteride-treated group. HE: hematoxylin and eosin; HEC: Huang’e capsule; BPH: benign prostatic hyperplasia. Differences among three or more groups were analyzed by analysis of variance. Data are presented as mean ± standard deviation (n = 7 per group). aP 0.01, bP 0.05, compared to Fina group; cP 0.01, dP 0.05, compared to BPH group.
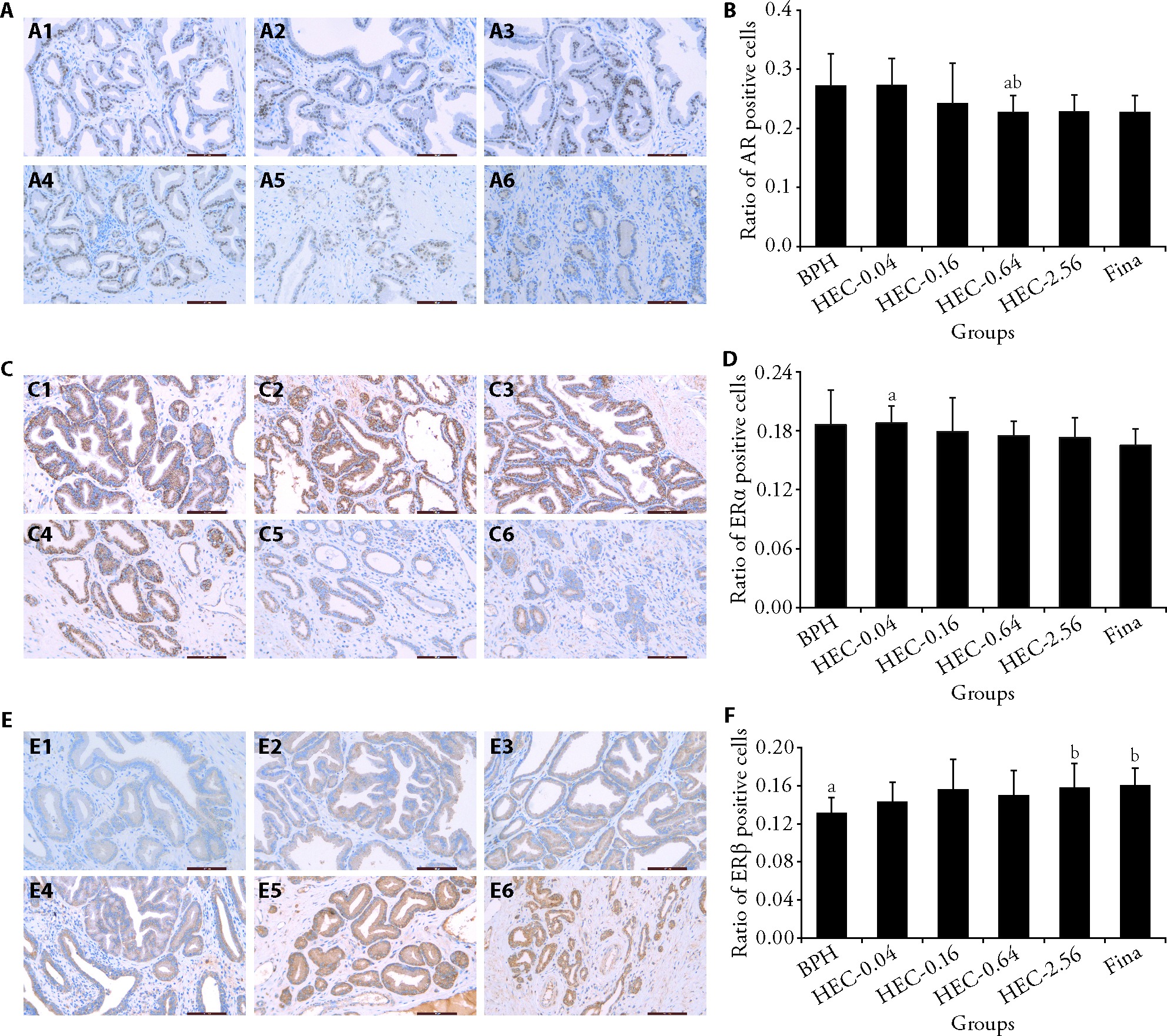
Figure 2 Effect of HEC on the expression of AR, ERα, and ERβ in prostate tissues in beagle dogs with spontaneous BPH A: slices of prostate tissue from each group were examined for AR using IHC (scale bar = 100 μm); A1: BPH group; A2: HEC-0.04 group; A3: HEC-0.16 group; A4: HEC-0.64 group; A5: HEC-2.56 group; A6: Fina group; B: the proportion of AR-positive cells in prostatic cells was estimated; C: slices of prostate tissue from each group were examined for ERα using IHC (scale bar = 100 μm); C1: BPH group; C2: HEC-0.04 group; C3: HEC-0.16 group; C4: HEC-0.64 group; C5: HEC-2.56 group; C6: Fina group; D: the proportion of cells that were ERα positive was calculated; E: slices of prostate tissue from each group were examined for ERβ using IHC (scale bar = 100 μm); E1: BPH group; E2: HEC-0.04 group; E3: HEC-0.16 group; E4: HEC-0.64 group; E5: HEC-2.56 group; E6: Fina group; F: the proportion of cells that were ERβ positive was calculated. BPH: the BPH group; HEC-0.04, HEC-0.16, HEC-0.64, HEC-2.56: 0.04, 0.16, 0.64, 2.56 g/kg HEC-treated group, respectively; Fina: 0.24 mg/kg finasteride-treated group. HEC: Huang’e capsule; BPH: benign prostatic hyperplasia; AR: androgen receptor; ERα: estrogen receptor alpha, ERβ: estrogen receptor beta; IHC: immunohistochemical. Differences among three or more groups were analyzed by analysis of variance. Data are presented as mean ± standard deviation (n = 7 per group). aP 0.05, compared to Fina group; bP 0.05, compared to BPH group.
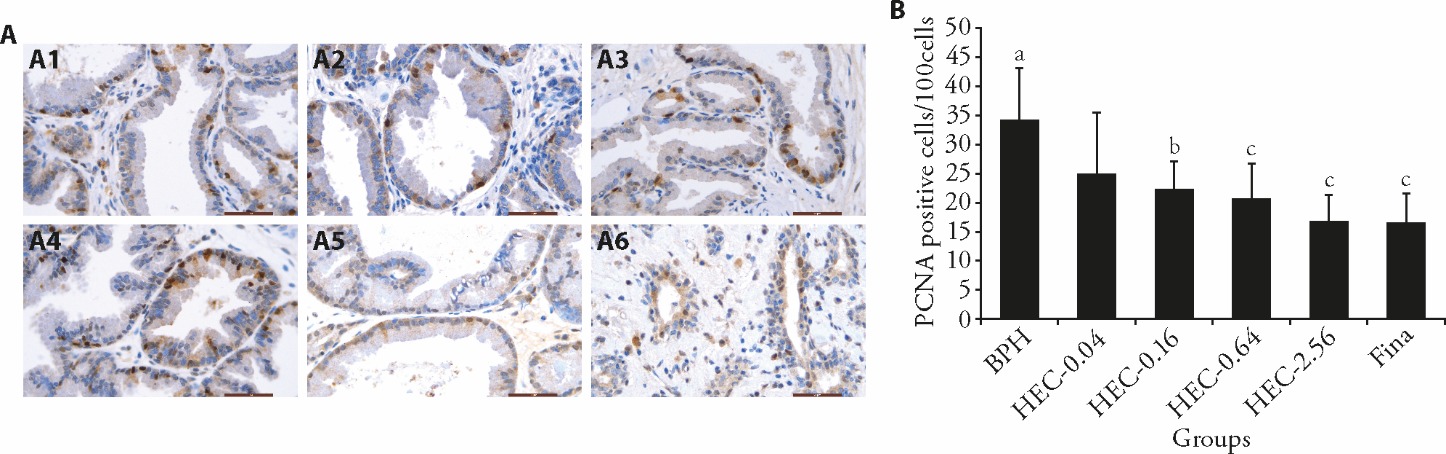
Figure 3 Effect of HEC on prostatic cell proliferation in beagle dogs with spontaneous BPH A: the localization of PCNA in each group was analyzed by IHC tests (scale bar = 50 μm); A1: BPH group; A2: HEC-0.04 group; A3: HEC-0.16 group; A4: HEC-0.64 group; A5: HEC-2.56 group; A6: Fina group; B: the proportion of PCNA-positive cells percentages was displayed. BPH: the BPH group; HEC-0.04, HEC-0.16, HEC-0.64, HEC-2.56: 0.04, 0.16, 0.64, 2.56 g/kg HEC-treated group, respectively; Fina: 0.24 mg/kg finasteride-treated group. PCNA: proliferating cell nuclear antigen; HEC: Huang’e capsule; BPH: benign prostatic hyperplasia. Differences among three or more groups were analyzed by analysis of variance. Data are presented as mean ± standard deviation (n = 7 per group). aP 0.05, compared to Fina group; bP 0.05, cP 0.01, compared to BPH group.
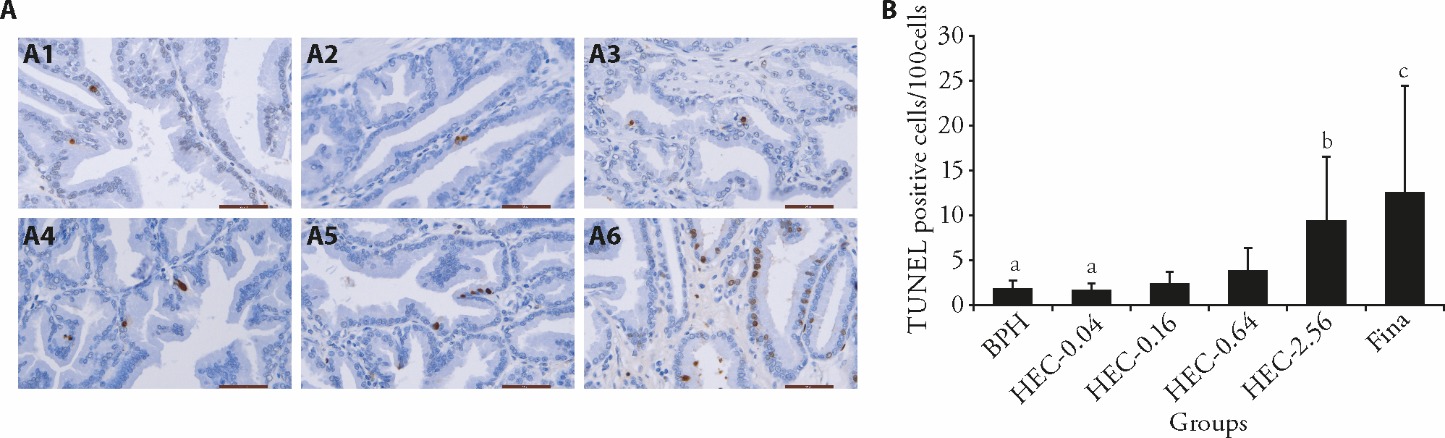
Figure 4 Effect of HEC on prostatic cell apoptosis in beagle dogs with spontaneous BPH A: the localization of TUNEL-positive cells in each group (scale bar = 50 μm); A1: BPH group; A2: HEC-0.04 group; A3: HEC-0.16 group; A4: HEC-0.64 group; A5: HEC-2.56 group; A6: Fina group; B: the proportion of TUNEL-positive cells was displayed. BPH: the BPH group; HEC-0.04, HEC-0.16, HEC-0.64, HEC-2.56: 0.04, 0.16, 0.64, 2.56 g/kg HEC-treated group, respectively; Fina: 0.24 mg/kg finasteride-treated group. TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling; HEC: Huang’e capsule; BPH: benign prostatic hyperplasia. Differences among three or more groups were analyzed by analysis of variance. Data are presented as mean ± standard deviation (n = 7 per group). aP 0.05, compared to Fina group; bP 0.01, cP 0.05, compared to BPH group.
| 1. |
Choi YJ, Fan M, Wedamulla NE, Tang Y, Kim EK. Alleviatory effect of isoquercetin on benign prostatic hyperplasia via IGF-1/PI3K/Akt/mTOR pathway. Food Sci Hum Wellness 2024; 13: 1698-710.
DOI URL |
| 2. |
Zhu C, Wang D, Zi H, et al. Epidemiological trends of urinary tract infections, urolithiasis and benign prostatic hyperplasia in 203 countries and territories from 1990 to 2019. Mil Med Res 2021; 8: 64.
DOI PMID |
| 3. |
Rho J, Seo CS, Park HS, et al. Asteris Radix et Rhizoma suppresses testosterone-induced benign prostatic hyperplasia in rats by regulating apoptosis and inflammation. J Ethnopharmacol 2020; 255: 112779.
DOI URL |
| 4. |
Wang Z, Mao Q, Yuan Y, Wang C, Wei H. Shuangshi Tonglin capsule treats benign prostatic hyperplasia through the ROS/NLRP3 signaling pathway. Int Urol Nephrol 2024; 56: 1259-71.
DOI |
| 5. |
Liu Y, Shao R, Suo T, et al. Traditional Chinese Medicine Danzhi qing’e decoction inhibits inflammation-associated prostatic hyperplasia via inactivation of ERK1/2 signal pathway. J Ethnopharmacol 2023; 309: 116354.
DOI URL |
| 6. |
Csikós E, Horváth A, Ács K, et al. Treatment of benign prostatic hyperplasia by natural drugs. Molecules 2021; 26: 7141.
DOI URL |
| 7. |
Krishnamoorthi R, Ganapathy AA, Hari Priya VM, Kumaran A. Future aspects of plant derived bioactive metabolites as therapeutics to combat benign prostatic hyperplasia. J Ethnopharmacol 2024; 330: 118207.
DOI URL |
| 8. | Dong J, Jia J, Ma W, Li J, Jiao Y, Luo S. Expressions of endothelin-I in benign prostatic hyperplasia rats treated with Long Bi Xiao capsule. Zhong Guo Nan Ke Xue Za Zhi 2009; 23: 35-8. |
| 9. | Ma W, Jia J, Jiao Y, et al. Effects of Chinese herbal medicine Long-Bi-Xiao capsule on fibronectin and collagen IV expression in human prostatic stromal cells cultured in vitro. Zhong Guo Nan Ke Xue Za Zhi 2008; 22: 7-10+14. |
| 10. |
Zhu PX, Su ZQ, Fan QY, Zhang C, Wang T. Network pharmacology and animal experiments revealed the protective effects of Guilong prescription (归龙方) on chronic prostatitis and its possible mechanisms. J Tradit Chin Med 2025; 45: 89-99.
DOI |
| 11. |
Sun F, Báez-Díaz C, Sánchez-Margallo FM. Canine prostate models in preclinical studies of minimally invasive interventions: part II, benign prostatic hyperplasia models. Transl Androl Urol 2017; 6: 547-55.
DOI PMID |
| 12. | National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals. 8th ed. Washington: National Academies Press, 2011: 42-76. |
| 13. |
Chen X, He S, Lu Y, et al. Inhibition of spontaneous canine benign prostatic hyperplasia by an Urtica fissa polysaccharide fraction. Planta Med 2015; 81: 10-4.
DOI PMID |
| 14. | Yuan B, Duan F, Zhang J, Zhang H, Wang M. Establishment of hormone-induced canine benign prostatic hyperplasia model: a prospective, controlled study. Heliyon 2022; 8: e11352. |
| 15. |
Angrimani DSR, Brito MM, Rui BR, Nichi M, Vannucchi CI. Reproductive and endocrinological effects of benign prostatic hyperplasia and finasteride therapy in dogs. Sci Rep 2020; 10: 14834.
DOI PMID |
| 16. |
Kanu SC, Ejezie FE, Ejezie CS, Eleazu CO. Effect of methanol extract of Plectranthus esculentus N.E.Br tuber and its fractions on indices of benign prostatic hyperplasia in Wistar rats. J Ethnopharmacol 2024; 331: 118301.
DOI URL |
| 17. | Jiang S, Xu Z, Jia J. Efficacy of Huang’e capsules on benign prostatic hyperplasia with Qi-deficiency, blood stasis and damp heat block: a stratified analysis. Zhong Hua Nan Ke Xue 2018; 24: 819-23. |
| 18. |
Lucas Cava V, Sánchez Margallo FM, Báez Díaz C, Dávila Gómez L, Lima Rodríguez JR, Sun F. Prostatic artery embolization with polyethylene glycol microspheres: evaluation in a canine spontaneous benign prostatic hyperplasia model. CVIR Endovasc 2020; 3: 44.
DOI PMID |
| 19. |
Hanh TTH, Hang DTT, Van Minh C, Dat NT. Anti-inflammatory effects of fatty acids isolated from Chromolaena odorata. Asian Pac J Trop Med 2011; 4: 760-3.
DOI PMID |
| 20. |
Park SY, Seetharaman R, Ko MJ, et al. Ethyl linoleate from garlic attenuates lipopolysaccharide-induced pro-inflammatory cytokine production by inducing heme oxygenase-1 in RAW264.7 cells. Int Immunopharmacol 2014; 19: 253-61.
DOI PMID |
| 21. |
Lao Z, Fan Y, Huo Y, et al. Physcion, a novel inhibitor of 5α-reductase that promotes hair growth in vitro and in vivo. Arch Dermatol Res 2022; 314: 41-51.
DOI |
| 22. |
Pan X, Wang H, Tong D, et al. Physcion induces apoptosis in hepatocellular carcinoma by modulating miR-370. Am J Cancer Res 2016; 6: 2919-31.
PMID |
| 23. |
Wang X, Zhu J, Yan H, et al. Kaempferol inhibits benign prostatic hyperplasia by resisting the action of androgen. Eur J Pharmacol 2021; 907: 174251.
DOI URL |
| 24. |
Hj K, Br J, Hj A. Psoralea corylifolia L. extract ameliorates benign prostatic hyperplasia by regulating prostate cell proliferation and apoptosis. J Ethnopharmacol 2021; 273: 113844.
DOI URL |
| 25. |
Cheng C, Xu C, Zhou W, et al. Integrated network pharmacology and serum metabonomics analysis to explore the potential mechanism of anemarrhena asphodeloides Bunge-Phellodendron chinense Schneid herb pair in the treatment of benign prostatic hyperplasia. J Pharm Biomed Anal 2024; 248: 116264.
DOI URL |
| 26. |
Wang Y, Liu L, Cheng C, et al. Study on mechanism of Zishen pill treating benign prostatic hyperplasia based on serum pharmacochemistry and network pharmacology. J Pharm Biomed Anal 2023; 234: 115480.
DOI URL |
| 27. |
Sreenivasulu K, Nandeesha H, Dorairajan LN, et al. Over expression of PI3K-AkT reduces apoptosis and increases prostate size in benign prostatic hyperplasia. Aging Male 2020; 23: 440-6.
DOI URL |
| 28. |
Zhou Y, Bolton EC, Jones JO. Androgens and androgen receptor signaling in prostate tumorigenesis. J Mol Endocrinol 2015; 54: R15-29.
DOI URL |
| 29. |
Jang YG, Go RE, Hwang KA, Choi KC. Resveratrol inhibits DHT-induced progression of prostate cancer cell line through interfering with the AR and CXCR4 pathway. J Steroid Biochem Mol Biol 2019; 192: 105406.
DOI URL |
| 30. |
Tao R, Miao L, Yu X, et al. Cynomorium songaricum Rupr demonstrates phytoestrogenic or phytoandrogenic like activities that attenuates benign prostatic hyperplasia via regulating steroid 5-α-reductase. J Ethnopharmacol 2019; 235: 65-74.
DOI URL |
| 31. | Meng F, Tang L. Challenges and prospect in research of Chinese materia medica network pharmacology. Zhong Cao Yao 2020, 51: 2232-7. |
| 32. |
Duan ZL, Wang YJ, Lu ZH, et al. Wumei Wan attenuates angiogenesis and inflammation by modulating RAGE signaling pathway in IBD: network pharmacology analysis and experimental evidence. Phytomedicine 2023; 111: 154658.
DOI URL |
| 33. |
Li X, Wei S, Niu S, et al. Network pharmacology prediction and molecular docking-based strategy to explore the potential mechanism of Huanglian Jiedu decoction against sepsis. Comput Biol Med 2022; 144: 105389.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

