Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (3): 489-495.DOI: 10.19852/j.cnki.jtcm.20231204.005
• Original articles • Previous Articles Next Articles
Exploring the multicomponent synergy mechanism of Zuogui Wan (左归丸) on postmenopausal osteoporosis by a systems pharmacology strategy
FENG Yanchen1, LIU Yali1, DANG Xue1, LIN Zixuan2, ZHANG Yunke2, CHE Zhiying1, LI Xiang3, PAN Xiaolong1, LIU Feixiang2( ), ZHENG Pan1(
), ZHENG Pan1( )
)
- 1 Traditional Chinese Medicine (Zhong Jing) School, Henan University of Chinese Medicine, Zhengzhou 450046, China
2 Hospital of Encephalopathy, the First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, China
3 State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, National Clinical Research Center for Infectious Diseases, Collaborative Innovation Center for Diagnosis and Treatment of Infectious Diseases, the First Affiliated Hospital, Zhejiang University School of Medicine, Research Units of Infectious Disease and Microecology, Chinese Academy of Medical Sciences, Hangzhou 31000, China
-
Received:2023-02-22Accepted:2023-07-14Online:2024-06-15Published:2023-12-04 -
Contact:LIU Feixiang, Hospital of Encephalopathy, the First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, China,spiritofwestlfx@126.com ; ZHENG Pan, Traditional Chinese Medicine (Zhong Jing) School, Henan University of Chinese Medicine, Zhengzhou 450046, China,zhengpanhn@163.com Telephone: +86-371-66221540 -
Supported by:National Natural Science Foundation of China: Research on Mechanism of Zuogui Wan in Treating Postmenopausal Osteoporosis by Regulating Feed-forward Loop of Oxytocin/Oxytocin Receptor Based on Transcriptome so as to Explain the Theory of “All Marrows Dominated by Brain”(82104730);Based on Protein Kinase Cθ/Nuclear Factor Kappa-B Signal Transduction Regulation, Shexiang Huangqi Compound Dropping Pills Regulate the Migration and Differentiation Mechanism of Mesenchymal Stem Cells Through the Blood-brain Barrier after Cerebral Ischemia(81974564);NThe 72nd Batch of China Postdoctoral Science Foundation(2022M721065);Central Plains Talent Program-science and Technology Innovation Leading Talent Project(224200510027)
Cite this article
FENG Yanchen, LIU Yali, DANG Xue, LIN Zixuan, ZHANG Yunke, CHE Zhiying, LI Xiang, PAN Xiaolong, LIU Feixiang, ZHENG Pan. Exploring the multicomponent synergy mechanism of Zuogui Wan (左归丸) on postmenopausal osteoporosis by a systems pharmacology strategy[J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 489-495.
share this article
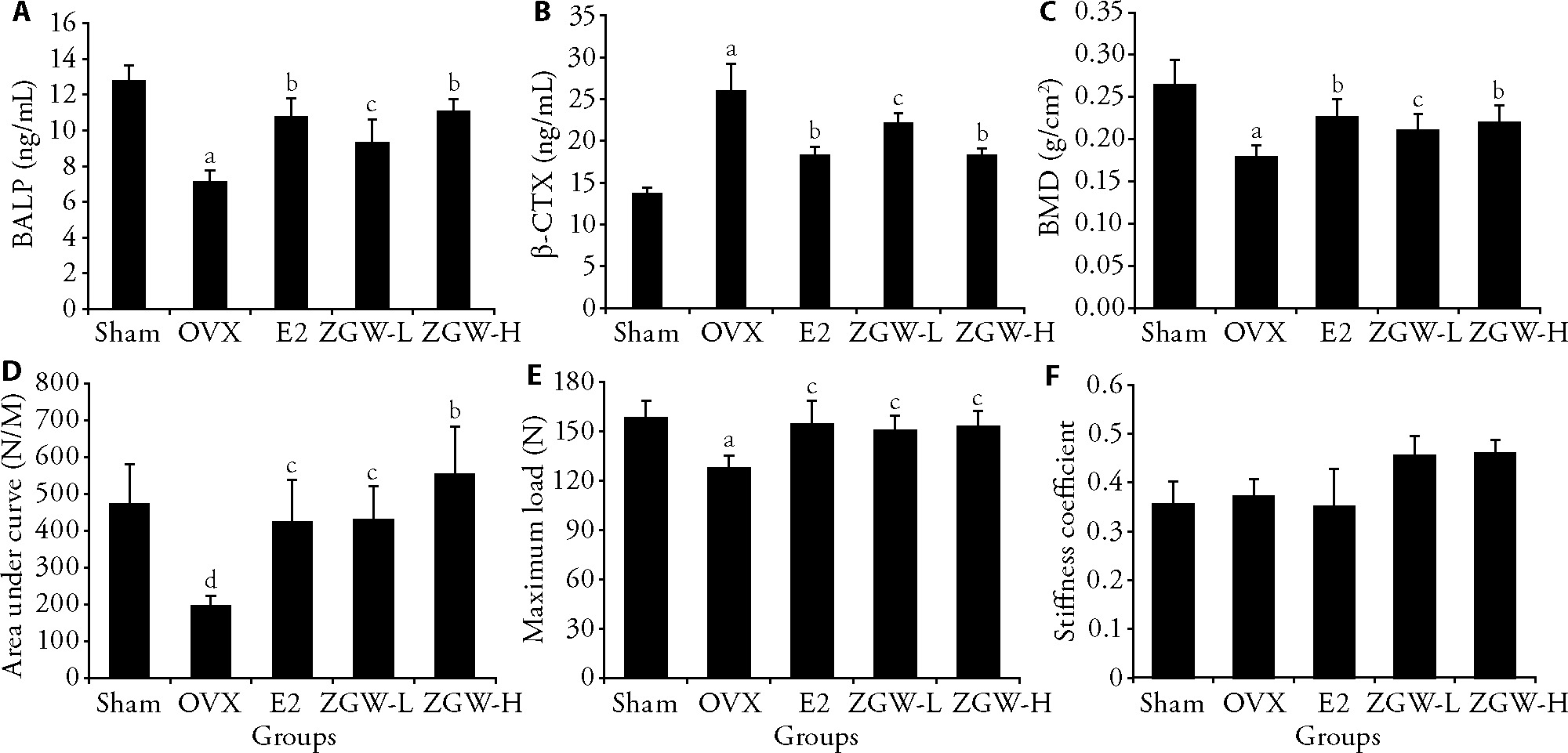
Figure 1 Effects of ZGW on bone-related indexes in rats A: serum levels of BALP were evaluated using ELISA kits (n = 5); B: serum levels of BALP and β-CTX were evaluated using ELISA kits (n = 5); C: changes in BMD levels in rats from each group following ZGW intervention (n = 7); D: changes in the levels of the area under the curve of the femoral stem (n = 4); E: changes in the levels of the maximum load of the femoral stem (n = 4); F: changes in the levels of the stiffness coefficient of the femoral stem (n = 4). Sham: sham-operated, treated with water for 12 weeks; OVX: ovariectomized, treated with water for 12 weeks; E2: 17β-estradiol, treated with 17β-E2 for 12 weeks (50 μg·kg-1·d-1); ZGW-L: low-dose ZGW (2.3 g·kg-1·d-1), treated with low-dose lyophilized ZGW powder dissolved in water for 12 weeks; ZGW-H: high-dose ZGW (4.6 g·kg-1·d-1), treated with high-dose lyophilized ZGW powder dissolved in water for 12 weeks. ZGW: Zuogui Wan. BALP: bone-specific alkaline phosphatase. β-CTX: β-C-terminal telopeptide of type I collagen. ELISA: enzyme-linked immunosorbent assay. BMD: bone mineral density. dP < 0.05, compared with the Sham group; aP < 0.01, compared with the Sham group; cP < 0.05, compared with the OVX group; bP < 0.01, compared with the OVX group. Data were presented as mean ± standard error of the mean. One-way Analysis of variance, followed by Student-Newman-Keuls test and the Student's t-test.
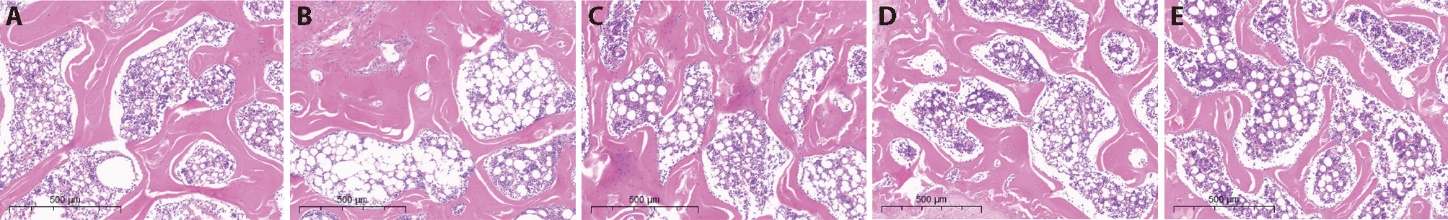
Figure 2 Changes in HE staining of the distal femur in groups after ZGW intervention (× 40) A: Sham group (n = 4). B: OVX group (n = 4). C: E2 group (n = 4). D: ZGW-L group (n = 4). E: ZGW-G group (n = 4). Sham: sham-operated, treated with water for 12 weeks; OVX: ovariectomized, treated with water for 12 weeks; E2: 17β-estradiol, treated with 17β-E2 for 12 weeks (50 μg·kg-1·d-1); ZGW-L: low-dose ZGW (2.3 g·kg-1·d-1), treated with low-dose lyophilized ZGW powder dissolved in water for 12 weeks; ZGW-H: high-dose ZGW (4.6 g·kg-1·d-1), treated with high-dose lyophilized ZGW powder dissolved in water for 12 weeks. HE: hematoxylin-eosin. ZGW: Zuogui Wan.
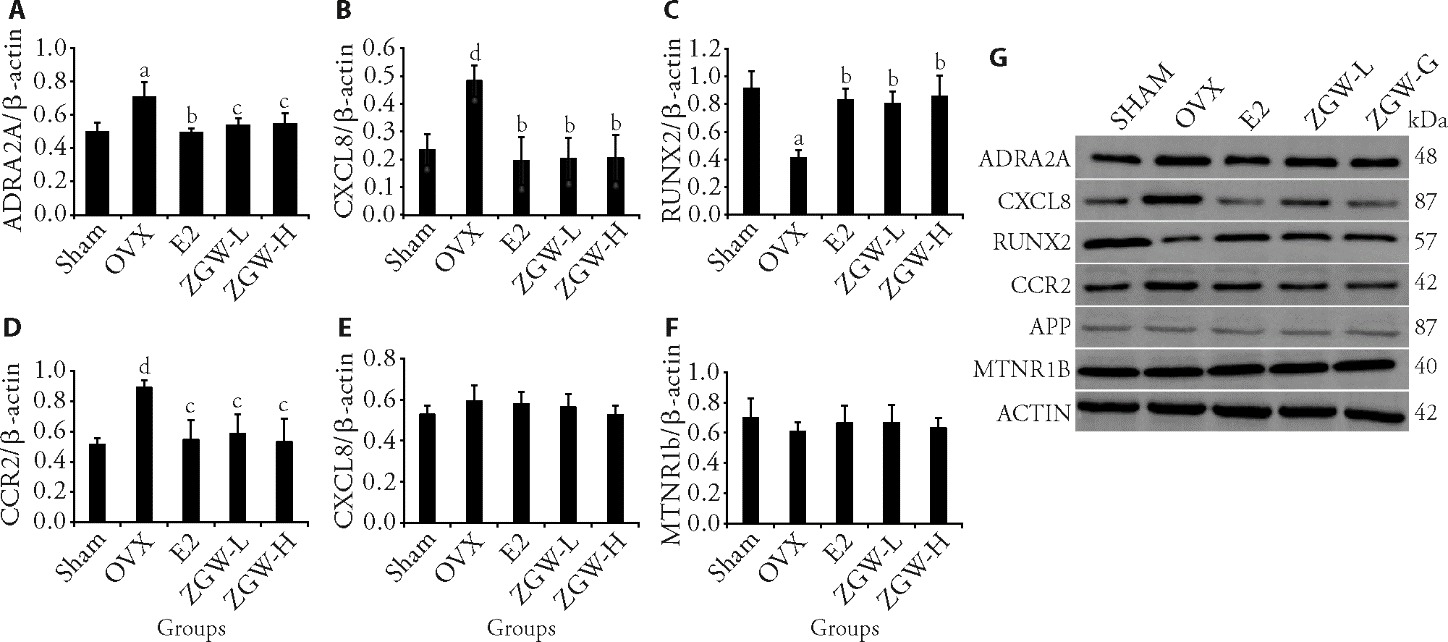
Figure 3 Changes in core gene expression levels after ZGW intervention A: protein expression levels of ADRA2A (n = 3); B: protein expression levels of CXCL8 (n = 3); C: protein expression levels of Runx2 (n = 3); D: protein expression levels of CCR2 (n = 3); E: protein expression levels of APP (n = 3); F: protein expression levels of MTNR1B (n = 3); G: Western blotting representative images of ADRA2A, CXCL8, RUNX2, CCR2, APP and MTNR1B (n = 3). Sham: sham-operated, treated with water for 12 weeks; OVX: ovariectomized, treated with water for 12 weeks; E2: 17β-estradiol, treated with 17β-E2 for 12 weeks (50 μg·kg-1·d-1); ZGW-L: low-dose ZGW (2.3 g·kg-1·d-1), treated with low-dose lyophilized ZGW powder dissolved in water for 12 weeks; ZGW-H: high-dose ZGW (4.6 g·kg-1·d-1), treated with high-dose lyophilized ZGW powder dissolved in water for 12 weeks. ZGW: Zuogui Wan; ADRA2A: rabbit anti-alpha-2a active receptor; CXCL8: rabbit anti-chemokine living 8; RUNX2: runt-related transcription factor 2; CCR2: C-C chemokine receptor type 2; APP: amyloid-beta A4 protein; MTNR1B: melatonin receptor type 1B. aP < 0.01, dP < 0.05, compared with Sham group; bP < 0.01, cP < 0.05, compared with OVX group. Data were presented as mean ± standard error of the mean. One-way Analysis of variance, followed by Student-Newman-Keuls test and the Student's t-test.
| 1. |
Leder BZ, Tsai JN, Uihlein AV, et al. Denosumab and teriparatide transitions in postmenopausal osteoporosis (the DATA-Switch study): extension of a randomised controlled trial. Lancet 2015; 386: 1147-55.
DOI PMID |
| 2. |
Black DM, Rosen CJ. Clinical practice. Postmenopausal osteoporosis. N Engl J Med 2016; 374: 254-62.
DOI URL |
| 3. |
Si Y, Wang C, Guo Y, Xu G, Ma Y. Prevalence of osteoporosis in patients with type 2 diabetes mellitus in the Chinese mainland: a systematic review and Meta-analysis. Iran J Public Health 2019; 48: 1203-14.
PMID |
| 4. |
Cosman F. Long-term treatment strategies for postmenopausal osteoporosis. Curr Opin Rheumatol 2018; 30: 420-6.
DOI PMID |
| 5. |
Le QA, Hay JW, Becker R, Wang Y. Cost-effectiveness analysis of sequential treatment of abaloparatide followed by alendronate versus teriparatide followed by alendronate in postmenopausal women with osteoporosis in the United States. Ann Pharmacother 2019; 53: 134-43.
DOI PMID |
| 6. | Li J, Lian JW. Pharmacology of traditional Chinese medical formulae. 4th ed. Beijing: China Press of Traditional Chinese Medicine, 2016: 138-9. |
| 7. | Hu Y, Zheng S, Fan Y, Sun L. Effects of Zuogui Wan on bone metabolism and fall risk in elderly diabetic patients. An Mo Yu Kang Fu Yi Xue 2017; 8: 51-4. |
| 8. | Liu F, Tan F, Tong W, et al. Effect of Zuogui Wan on osteoporosis in ovariectomized rats through RANKL/OPG pathway mediated by β2AR. Biomed Pharmacother 2018; 103: 1052-60. |
| 9. |
Kang X, Chen L, Yang S, et al. Zuogui Wan slowed senescence of bone marrow mesenchymal stem cells by suppressing Wnt/β-catenin signaling. J Ethnopharmacol 2022; 294: 115323.
DOI URL |
| 10. |
Artero A, Artero A, Tarín JJ, Cano A. The impact of moderate wine consumption on health. Maturitas 2015; 80: 3-13.
DOI PMID |
| 11. |
Ahmad Khan M, Sarwar AHMG, Rahat R, Ahmed RS, Umar S. Stigmasterol protects rats from collagen induced arthritis by inhibiting proinflammatory cytokines. Int Immunopharmacol 2020; 85: 106642.
DOI URL |
| 12. |
Yang L, Chen Q, Wang F, Zhang G. Antiosteoporotic compounds from seeds of Cuscutachinensis. J Ethnopharmacol 2011; 135: 553-60.
DOI URL |
| 13. | Li SF, Tang JJ, Chen J, et al. Regulation of bone formation by baicalein via the mTORC1 pathway. Drug Des Devel Ther 2015; 9: 5169-83. |
| 14. |
Chauhan S, Sharma A, Upadhyay NK, et al. In-vitro osteoblast proliferation and in-vivo anti-osteoporotic activity of Bombaxceiba with quantification of Lupeol, gallic acid and β-sitosterol by HPTLC and HPLC. BMC Complement Altern Med 2018; 18: 233.
DOI |
| 15. | Geng X, Yang L, Zhang C, Qin H, Liang Q. Wogonin inhibits osteoclast differentiation by inhibiting NFATc1 translocation into the nucleus. ExpTher Med 2015; 10: 1066-70. |
| 16. |
Ma J, Han L, Zhou X, Li Z. Clinical significance of Vitamin-D and other bone turnover markers on bone mineral density in patients with gestational diabetes mellitus. Pak J Med Sci 2022; 38: 23-7
DOI PMID |
| 17. |
Neri AA, Galanis D, Galanos A, Pepe AE, et al. The effect of Ceratonia siliqua supplement on bone mineral density in ovariectomy-induced osteoporosis in rats. In Vivo 2023; 37: 270-85.
DOI URL |
| 18. |
Yousefzadeh N, Kashfi K, Jeddi S, Ghasemi A. Ovariectomized rat model of osteoporosis: a practical guide. Excli J 2020; 19: 89-107.
DOI PMID |
| 19. |
Jung YK, Kang YM, Han S. Osteoclasts in the inflammatory arthritis: implications for pathologic osteolysis. Immune Netw 2019; 19: e2.
DOI URL |
| 20. |
Pagani S, Fini M, Giavaresi G, Salamanna F, Borsari V. The active role of osteoporosis in the interaction between osteoblasts and bone metastases. Bone 2015; 79: 176-82.
DOI PMID |
| 21. | Yang P. Expression prolife of cytokines and chemokines in patients with osteoarthritis and the function of CXCL8 and CXCL11 in the regulation of chondrocytes. Xi'an: The Fourth Military Medical University, 2017: 1-86. |
| 22. | Yang Y. The mechanism of osteoblastic CXCL2 in regulation of bone formation and osteoblastic differentiation. Guangzhou: Southern Medical University, 2019: 1-73. |
| 23. |
Li S, Liu B, Zhang L, Rong L. Amyloid beta peptide is elevated in osteoporotic bone tissues and enhances osteoclast function. Bone 2014; 61: 164-75.
DOI PMID |
| 24. | Liu L, Wang T, Yang X, et al. MTNR1B loss promotes chordoma recurrence by abrogating melatonin-mediated β-catenin signaling repression. J Pineal Res 2019; 67: e12588. |
| 25. |
Li Y, Zhou J, Wu Y, et al. Association of osteoporosis with genetic variants of circadian genes in Chinese geriatrics. Osteoporos Int 2016; 27: 1485-92.
DOI PMID |
| 26. |
Mlakar V, Jurkovic Mlakar S, Zupan J, et al. ADRA2A is involved in neuro-endocrine regulation of bone resorption. J Cell Mol Med 2015; 19: 1520-9.
DOI PMID |
| [1] | YANG Qinjun, YIN Dandan, WANG Hui, GAO Yating, WANG Xinheng, WU Di, TONG Jiabing, WANG Chuanbo, LI Zegeng. Uncovering the action mechanism of Shenqi Tiaoshen formula (参芪调肾方) in the treatment of chronic obstructive pulmonary disease through network pharmacology, molecular docking, and experimental verification [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 770-783. |
| [2] | HU Yuanyuan, LIU Xinguang, ZHAO Peng, WU Jinyan, YAN Xinhua, HOU Runsu, WANG Xiangcheng, YANG Fan, TIAN Xinrong, LI Jiansheng. Integration of serum pharmacochemistry with network pharmacology to reveal the potential mechanism of Yangqing Chenfei formula (养清尘肺方) for the treatment of silicosis [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 784-793. |
| [3] | LUO Shan, YANG Fan, CHEN Yuanchun, ZHAO Ruoxi, LIU Haiye, GAO Fei, MA Wencan, GAO Weijuan, YU Wentao. Sanhua Tang (三化汤) protects against ischemic stroke by preventing blood-brain barrier injury: a network pharmacology and in vivo experiments [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 794-803. |
| [4] | CHU Mengzhen, WANG Yu, LIN Zhijian, Lyu Jintao, ZHANG Xiaomeng, ZHANG Bing. Investigation of the active ingredients and mechanism of Shuangling extract in dextran sulfate sodium salt induced ulcerative colitis mice based on network pharmacology and experimental verification [J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 478-488. |
| [5] | WANG Yan, DENG Fanying, LIU Shiqi, WANG Yingli. Network pharmacology and experimental validation to reveal the pharmacological mechanisms of Sini decoction (四逆汤) against renal fibrosis [J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 362-372. |
| [6] | ZHI Guoguo, SHAO Bingjie, ZHENG Tianyan, JI Shaoxiu, LI Jingwei, DANG Yanni, LIU Feng, WANG Dong. Efficacy of Ganshuang granules (肝爽颗粒) on non-alcoholic fatty liver and underlying mechanism: a network pharmacology and experimental verification [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 122-130. |
| [7] | YANG Ye, CHEN Xiaoyang, YAO Junkai, HU Yueyao, WANG Wei. Efficacy of Danlou tablet (丹蒌片) on myocardial ischemia/ reperfusion injury assessed by network pharmacology and experimental verification [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 131-144. |
| [8] | ZHANG Qi, CHEN Dexuan, ZHU Guixiang, ZHANG Shihu, FENG Xiao, MA Chaoqun, ZHANG Yi. Efficacy of Tounongsan decoction (透脓散方) on pyogenic liver abscess: network pharmacology and clinical trial validation [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 145-155. |
| [9] | REN Hui, ZHAO Lintao, GAO Kai, YANG Yuanyuan, CUI Xiaomin, HU Jing, CHEN Zhiyong, LI Ye. Deciphering the chemical profile and pharmacological mechanism of Jinlingzi powder (金铃子散) against bile reflux gastritis using ultra-high performance liquid chromatography coupled with Q exactive focus mass spectrometry, network pharmacology, and molecular docking [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1209-1218. |
| [10] | YANG Xirui, ZHAO Hui, SHAN Muhammad, DONG Feixue, ZHANG Dandan, WANG Jixue, YUAN Xingxing. Efficacy of bioactive compounds of Chaihu (Radix Bupleuri Chinensis) on glaucomatous optic atrophy through interleukin-6/hypoxia inducible factor-1α signal pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1219-1226. |
| [11] | HAN Huagang, LI Ziqiang, OUYANG Jingfeng, WANG Tianquan, DONG Lingyan, CAO Junling. Mechanism of Lingbao Huxin Dan (灵宝护心丹) in the treatment of bradyarrhythmia complicated with coronary heart disease: a network pharmacology analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1001-1009. |
| [12] | PANG Fengtao, LI Kesong, ZHANG Yi, TANG Xiaopo, ZHOU Xinyao. Efficacy of Lushi Runzao decoction (路氏润燥汤) on ameliorating Sjogren's syndrome: a network pharmacology and experimental verification-based study [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 751-759. |
| [13] | JIA Lihua, KUANG Haodan, XU Yuan. Efficacy of Buzhong Yiqi decoction (补中益气汤) on benign prostatic hyperplasia and its possible mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 533-541. |
| [14] | JIA Lihong, TIE Defu, FAN Zhaohui, CHEN Dan, CHEN Qizhu, CHEN Jun, BO Huaben. Mechanism underlying Fanmugua (Fructus Caricae) leaf multicomponent synergistic therapy for anemia: data mining based on hematopoietic network [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 542-551. |
| [15] | LI Yue, WEN Shuting, ZHAO Runyuan, FAN Dongmei, ZHAO Dike, LIU Fengbin, MI Hong. Efficacy of active ingredients in Qingdai (Indigo Naturalis) on ulcerative colitis: a network pharmacology-based evaluation [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 124-133. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

