Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (3): 478-488.DOI: 10.19852/j.cnki.jtcm.20240408.003
• Original articles • Previous Articles Next Articles
Investigation of the active ingredients and mechanism of Shuangling extract in dextran sulfate sodium salt induced ulcerative colitis mice based on network pharmacology and experimental verification
CHU Mengzhen, WANG Yu, LIN Zhijian, Lyu Jintao, ZHANG Xiaomeng, ZHANG Bing( )
)
- Department of Clinical Chinese Pharmacy, School of Chinese Materia Medica, Beijing University of Chinese Medicine, Beijing 102488, China
-
Received:2023-02-22Accepted:2023-06-15Online:2024-06-15Published:2024-04-08 -
Contact:ZHANG Bing, School of Chinese Materia Medica, Liangxiang Campus, Beijing University of Chinese Medicine, Beijing 102488, China.Zhangb@bucm.edu.cn Telephone: +86-1390113756 -
Supported by:Innovation Team and Talents Support Program of National Administration of Traditional Chinese Medicine: Construction and Practice of Pharmacovigilance Theory System in Line with the Characteristics of Traditional Chinese Medicine(ZYYCXTD-C-202005-11);National Leading Talents Support Plan of Traditional Chinese Medicine ‘Qihuang Scholar’ Plan: Chinese Medicine Inheritance and Innovation "One Hundred Million" Talent Project (Qihuang Project) Qihuang Scholars(1040063320004)
Cite this article
CHU Mengzhen, WANG Yu, LIN Zhijian, Lyu Jintao, ZHANG Xiaomeng, ZHANG Bing. Investigation of the active ingredients and mechanism of Shuangling extract in dextran sulfate sodium salt induced ulcerative colitis mice based on network pharmacology and experimental verification[J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 478-488.
share this article
| ID | Pathway | Gene ratio | P value |
|---|---|---|---|
| hsa05200 | Pathways in cancer | 29/38 | 4.67E-28 |
| hsa05161 | Hepatitis B | 22/38 | 1.37E-26 |
| hsa05212 | Pancreatic cancer | 16/38 | 6.01E-22 |
| hsa05219 | Bladder cancer | 14/38 | 4.64E-21 |
| hsa05205 | Proteoglycans in cancer | 20/38 | 2.97E-20 |
| hsa04917 | Prolactin signaling pathway | 15/38 | 2.02E-19 |
| hsa05142 | Chagas disease (American trypanosomiasis) | 16/38 | 1.22E-18 |
| hsa04668 | TNF signaling pathway | 16/38 | 1.91E-18 |
| hsa04620 | Toll-like receptor signaling pathway | 15/38 | 7.87E-17 |
| hsa05215 | Prostate cancer | 14/38 | 2.68E-16 |
| hsa05213 | Endometrial cancer | 12/38 | 1.09E-15 |
| hsa04915 | Estrogen signaling pathway | 14/38 | 1.33E-15 |
| hsa04660 | T cell receptor signaling pathway | 14/38 | 1.52E-15 |
| hsa05133 | Pertussis | 13/38 | 1.65E-15 |
| hsa05160 | Hepatitis C | 15/38 | 2.19E-15 |
| hsa04068 | FoxO signaling pathway | 15/38 | 2.42E-15 |
| hsa05210 | Colorectal cancer | 12/38 | 8.93E-15 |
| hsa05140 | Leishmaniasis | 12/38 | 4.38E-14 |
| hsa05220 | Chronic myeloid leukemia | 12/38 | 5.15E-14 |
| hsa05223 | Non-small cell lung cancer | 11/38 | 1.62E-13 |
Table 1 KEGG enrichment analysis of related pathways
| ID | Pathway | Gene ratio | P value |
|---|---|---|---|
| hsa05200 | Pathways in cancer | 29/38 | 4.67E-28 |
| hsa05161 | Hepatitis B | 22/38 | 1.37E-26 |
| hsa05212 | Pancreatic cancer | 16/38 | 6.01E-22 |
| hsa05219 | Bladder cancer | 14/38 | 4.64E-21 |
| hsa05205 | Proteoglycans in cancer | 20/38 | 2.97E-20 |
| hsa04917 | Prolactin signaling pathway | 15/38 | 2.02E-19 |
| hsa05142 | Chagas disease (American trypanosomiasis) | 16/38 | 1.22E-18 |
| hsa04668 | TNF signaling pathway | 16/38 | 1.91E-18 |
| hsa04620 | Toll-like receptor signaling pathway | 15/38 | 7.87E-17 |
| hsa05215 | Prostate cancer | 14/38 | 2.68E-16 |
| hsa05213 | Endometrial cancer | 12/38 | 1.09E-15 |
| hsa04915 | Estrogen signaling pathway | 14/38 | 1.33E-15 |
| hsa04660 | T cell receptor signaling pathway | 14/38 | 1.52E-15 |
| hsa05133 | Pertussis | 13/38 | 1.65E-15 |
| hsa05160 | Hepatitis C | 15/38 | 2.19E-15 |
| hsa04068 | FoxO signaling pathway | 15/38 | 2.42E-15 |
| hsa05210 | Colorectal cancer | 12/38 | 8.93E-15 |
| hsa05140 | Leishmaniasis | 12/38 | 4.38E-14 |
| hsa05220 | Chronic myeloid leukemia | 12/38 | 5.15E-14 |
| hsa05223 | Non-small cell lung cancer | 11/38 | 1.62E-13 |
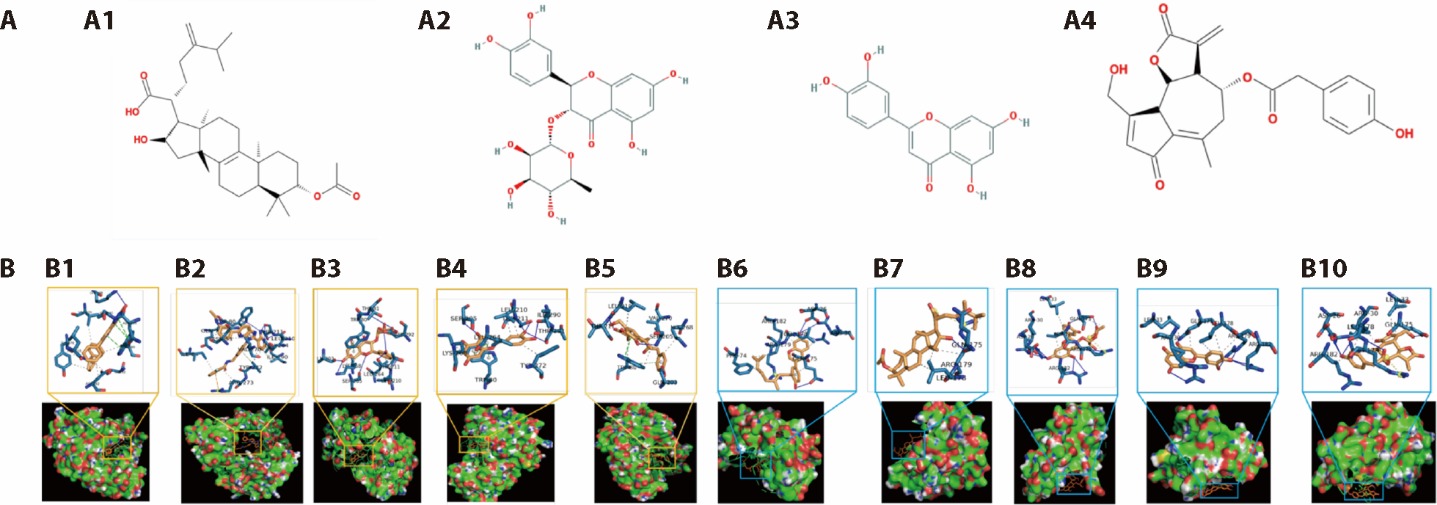
Figure 1 Molecular docking research A: structural formula of the main active ingredient. A1: pachymic acid; A2: astilbin; A3: luteolin; A4: lactucopicrin. B: molecular docking diagram of active components of Shuangling extract and key targets. B1-B5: docking results of MK-2206 dihydrochloride, pachymic acid, astilbin, luteolinandlactucopicrin active ingredients with AKT1 target protein. B6-B10: docking results of Corylifol A, pachymic acid, astilbin, luteolinand lactucopicrin active ingredients with IL-6 target protein. IL-6: interleukin-6; AKT1: AKT serine/threonine kinase 1.
| Compound | Estimated ΔG (kcal/mol) | |||||
|---|---|---|---|---|---|---|
| MK-2206 dihydrochloride | Corylifol A | pachymic acid | astilbin | luteolin | lactucopicrin | |
| AKT1 | -12.02 | - | -7.41 | -9.59 | -7.09 | -9.87 |
| IL-6 | - | -6.65 | -5.39 | -4.2 | -4.95 | -5.45 |
Table 2 Molecular docking results of active Ingredients of Shuangling extract and core targets
| Compound | Estimated ΔG (kcal/mol) | |||||
|---|---|---|---|---|---|---|
| MK-2206 dihydrochloride | Corylifol A | pachymic acid | astilbin | luteolin | lactucopicrin | |
| AKT1 | -12.02 | - | -7.41 | -9.59 | -7.09 | -9.87 |
| IL-6 | - | -6.65 | -5.39 | -4.2 | -4.95 | -5.45 |
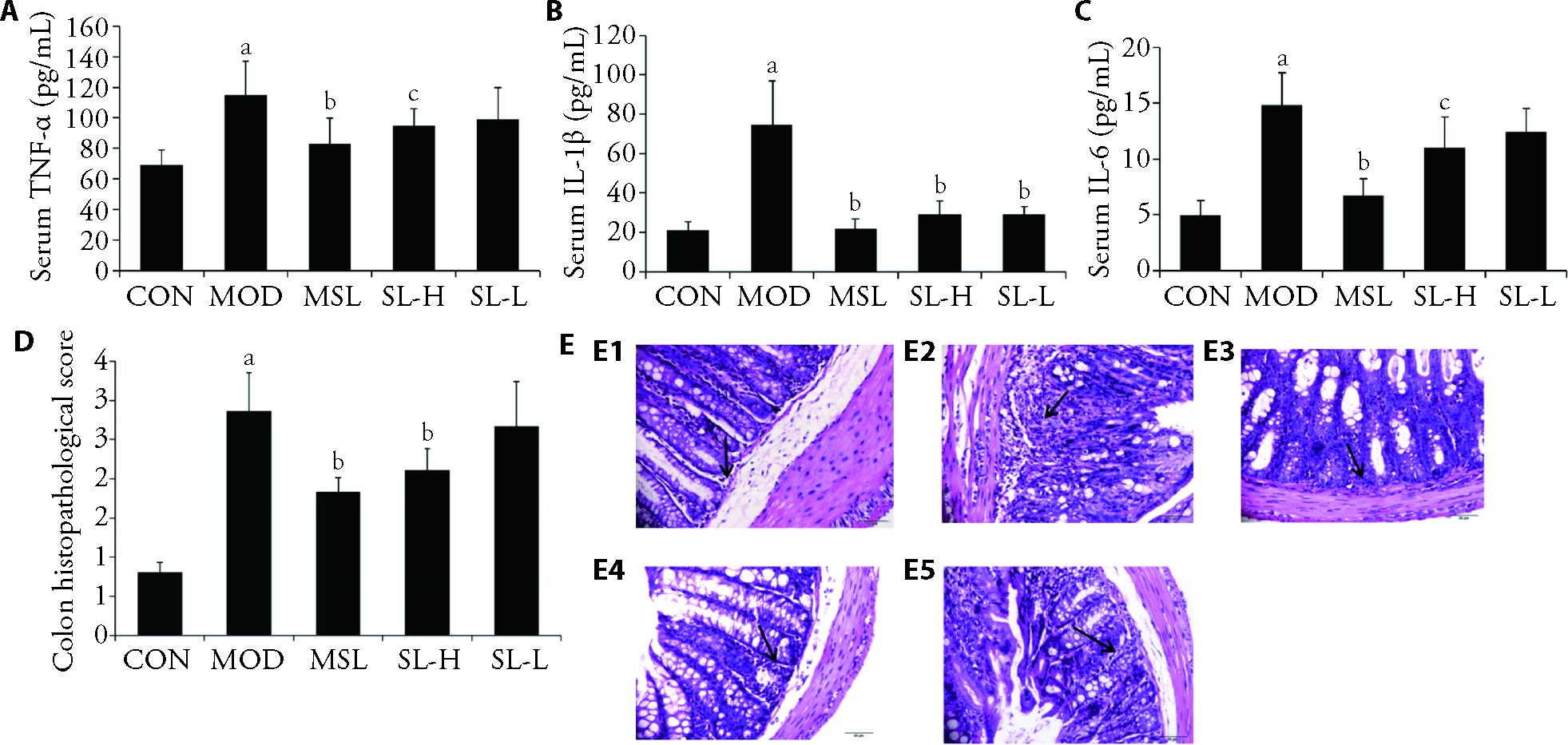
Figure 2 Serum inflammatory cytokine levels and histopathological HE staining A: levels of serum TNF-α; B: levels of serumIL-1β; C: levels of serumIL-6; D: colon tissue pathology HE staining score; E: representative HE stained slices of Shuangling extract in anti-UC (× 400). E1: CON; E2: MOD; E3: MSL; E4: SL-H; E5: SL-L. CON group: normal operation (n = 6); MOD group: 3% DSS without treatment (n = 6); MSL group: 3% DSS rats treated with Mesalazine water solution at a daily dose of 400 mg/kg (n = 6); SL-H group: 3% DSS rats treated with Shuangling extract at a daily dose of 18.34 g/kg for 7 d (n = 6). SL-L group: 3% DSS rats treated with Shuangling extract at a daily dose of 9.17 g/kg for 7 d (n = 6). TNF-α: tumor necrosis factor-α; IL-1β: interleukin-1β; IL-6: interleukin-6; HE: hematoxylin-eosin. CON: control; MOD: model; MSL: mesalazine; SL-H: Shuangling extract high-dose; SL-L: Shuangling extract low-dose; DSS: dextran sulfate solution; UC: ulcerative colitis. The statistical significances were assessed using a one-way analysis of variance followed by Dunnett's test. All results were presented as mean ± standard deviation. aP < 0.01, compared with the CON group; bP < 0.01 and cP < 0.05, compared with the MOD group.

Figure 3 Expression of the CD3 and Foxp3 protein in the colon A: immunohistochemical staining of CD3 protein in mouse colon (× 400). A1: CON; A2: MOD; A3: MSL; A4: SL-H; A5: SL-L. B: immunohistochemical staining of FOXP3 protein in mouse colon (× 400). B1: CON; B2: MOD; B3: MSL; B4: SL-H; B5: SL-L. C: average optical density of CD3 protein in colon tissue of mice in each group; D: average optical density of FOXP3 protein in colon tissue of mice in each group. CON group: normal operation (n = 6); MOD group: 3% DSS without treatment (n = 6); MSL group: 3% DSS rats treated with Mesalazine water solution at a daily dose of 400 mg/kg (n = 6); SL-H group: 3% DSS rats treated with Shuangling extract at a daily dose of 18.34 g/kg for 7 d (n = 6). SL-L group: 3% DSS rats treated with Shuangling extract at a daily dose of 9.17 g/kg for 7 d (n = 6). CD3: cluster of differentiation 3; FOXP3: forkhead box p3; DSS: dextran sulfate solution; UC: ulcerative colitis; CON: control; MOD: model; MSL: mesalazine; SL-H: Shuangling extract high-dose; SL-L: Shuangling extract low-dose; DSS: dextran sulfate solution. The statistical significances were assessed using a one-way analysis of variance followed by Dunnett's test. All results were presented as mean ± standard deviation. aP < 0.05, compared with the CON group; bP < 0.05, compared with the MOD group.
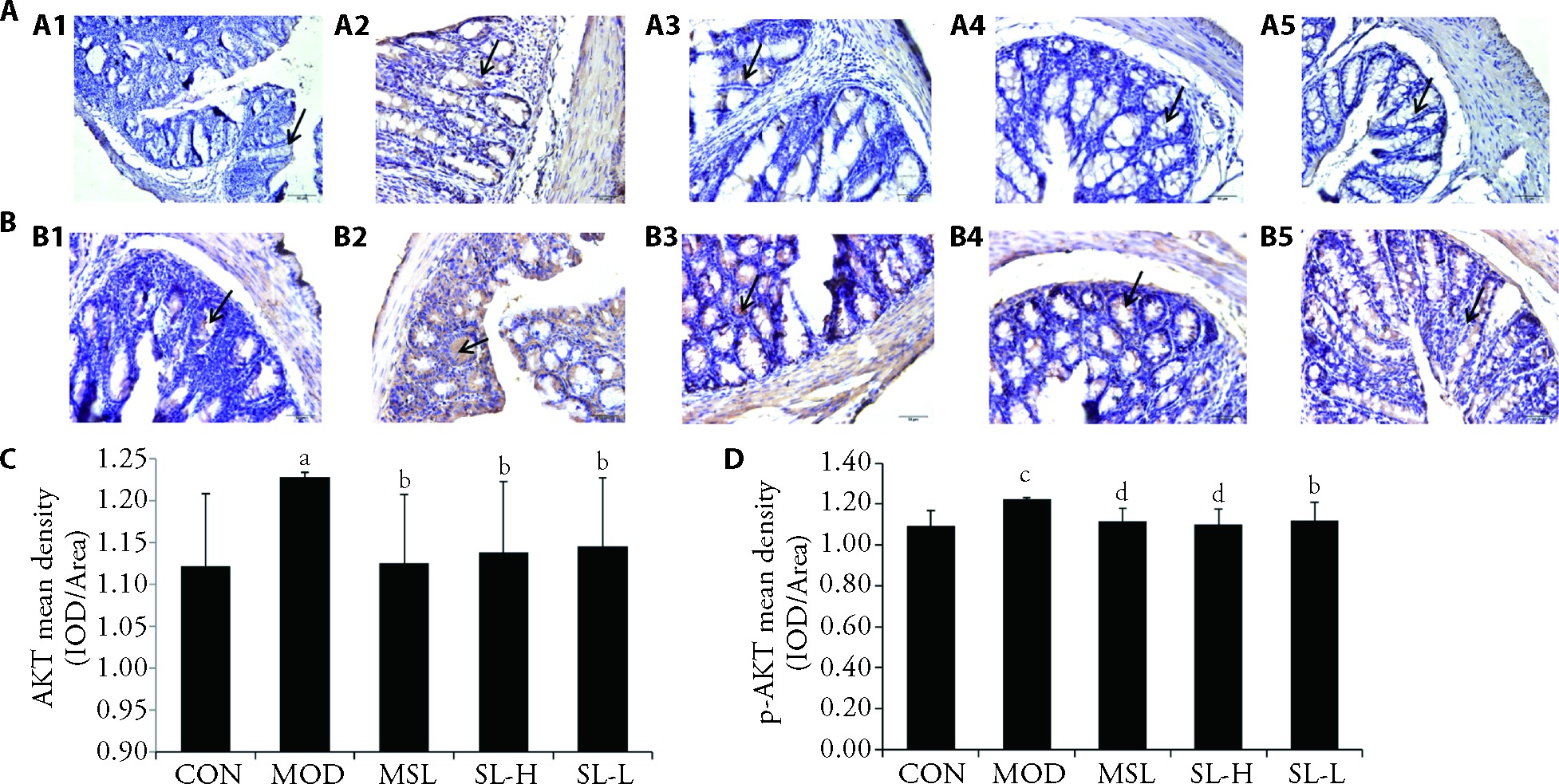
Figure 4 Expression of the AKT and p-AKT protein in the colon A: immunohistochemical staining of AKT protein in mouse colon (× 400). A1: CON; A2: MOD; A3: MSL; A4: SL-H; A5: SL-L; B: immunohistochemical staining of p-AKT protein in mouse colon (× 400). B1: CON; B2: MOD; B3: MSL; B4: SL-H; B5: SL-L; C: average optical density of AKT protein in colon tissue of mice in each group; D: average optical density of p-AKT protein in colon tissue of mice in each group. CON group: normal operation (n = 6); MOD group: 3% DSS without treatment (n = 6); MSL group: 3% DSS rats treated with Mesalazine water solution at a daily dose of 400 mg/kg (n = 6); SL-H group: 3% DSS rats treated with Shuangling extract at a daily dose of 18.34 g/kg for 7 d (n = 6). SL-L group: 3% DSS rats treated with Shuangling extract at a daily dose of 9.17 g/kg for 7 d (n = 6). AKT: AKT serine/threonine kinase; p-AKT: phospho-Akt Serine/threonine kinase; UC: ulcerative colitis; CON: control; MOD: model; MSL: mesalazine; SL-H: Shuangling extract high-dose; SL-L: Shuangling extract low-dose; DSS: dextran sulfate solution. The statistical significances were assessed using a one-way analysis of variance followed by Dunnett's test. All results were presented as mean ± standard deviation. aP < 0.05, cP < 0.01, compared with the CON group; bP < 0.05, dP < 0.01, compared with the MOD group.
| 1. |
Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet 2017; 389: 1756-70.
DOI PMID |
| 2. | Chen F, Yan CK. Recent progress in drug therapy of ulcerative colitis. Shi Jie Hua Ren Xiao Hua Za Zhi 2016; 24: 1840-45. |
| 3. | Zeng SM, Lin WH, Luo PJ, Wei J, Liang Q, Huang GH. Progress in Traditional Chinese Medicine treatment of ulcerative colitis. Liaoning Zhong Yi Za Zhi 2020; 47: 209-12. |
| 4. | Wang YZ, Gao S, Li LJ, Zhang L, Sun Y, Bao FM. Research advances on bioactivity and pharmacological effects of chicoric acid. Zhong Guo Xin Yao Za Zhi 2020; 29: 1729-33. |
| 5. | You X, Xiong DG, Guo ZB, Shang YJ. Advancement of a variety of chemical constituents and pharmacological mechanisms of poriacocos. Anhui Nong Ye Ke Xue 2015; 43: 106-9. |
| 6. | Wang XL, Peng YH, Sun MY, Shi Y. The knowledge of disease cause of Traditional Chinese Medicine to ulcerative colitis. Liaoning Zhong Yi Za Zhi 2007; 34: 572-3. |
| 7. | Fang WY, Zhong YB, Liu DY, Zhao HM. Progress in clinical research of Bupiyichang pill in treating ulcerative colitis. Jiangxi Zhong Yi Yao Da Xue Xue Bao 2021; 33: 116-20. |
| 8. | Ru J, Li P, Wang J, et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Chem Inform 2014; 6: 13. |
| 9. |
Ahmed SS, Ramakrishnan V. Systems biological approach of molecular descriptors connectivity: optimal descriptors for oral bioavailability prediction. PLoS One 2012; 7: e40654.
DOI URL |
| 10. | Zong Y, Dong HL, Chen T, Yan S, Jiang GR. Mechanism of herb pair containing Scutellariae Radix and Coptidis Rhizoma on treatment of type 2 diabetes mellitus based on network pharmacology. Zhong Cao Yao 2019; 50: 888-94. |
| 11. |
Daina A, Michielin O, Zoete V. Swiss Target Prediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res 2019; 47: W357-64.
DOI URL |
| 12. |
UniProt Consortium. UniProt: a worldwide hub of protein knowledge. Nucleic Acids Res 2019; 47: D506-15.
DOI |
| 13. | Amberger JS, Hamosh A. Searching online mendelian inheritance in man (OMIM): a knowledgebase of human genes and genetic phenotypes. Curr Protoc Bioinformatics 2017; 58: 1.2. 1-12. |
| 14. | Wishart DS, Feunang YD, Guo AC, et al. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res 2018; 46: D1074-82. |
| 15. | Wang Y, Zhang S, Li F, et al. Therapeutic target database 2020: enriched resource for facilitating research and early development of targeted therapeutics. Nucleic Acids Res 2020; 48: D1031-41. |
| 16. |
Szklarczyk D, Gable AL, Lyon D, et al. STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res 2019; 47: D607-13.
DOI |
| 17. |
Jiao X, Sherman BT, Huang da W, et al. DAVID-WS: a stateful web service to facilitate gene/protein list analysis. Bioinformatics 2012; 28: 1805-6.
DOI PMID |
| 18. |
Huang da W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 2009; 4: 44-57.
DOI PMID |
| 19. |
Su M, Guo C, Liu M, Liang X, Yang B. Therapeutic targets of vitamin C on liver injury and associated biological mechanisms: a study of network pharmacology. Int Immunopharmacol 2019; 66: 383-7.
DOI PMID |
| 20. |
Du Z, Zhou X, Ling Y, Zhang Z, Su Z. AgriGO: a GO analysis toolkit for the agricultural community. Nucleic Acids Res 2010; 38: W64-70.
DOI URL |
| 21. |
Kanehisa M, Furumichi M, Tanabe M, Sato Y, Morishima K. KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res 2017; 45: D353-61.
DOI URL |
| 22. |
Kim S, Chen J, Cheng T, et al. PubChem in 2021: new data content and improved web interfaces. Nucleic Acids Res 2021; 49: D1388-95.
DOI PMID |
| 23. |
Morris GM, Huey R, Lindstrom W, et al. AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 2009; 30: 2785-91.
DOI PMID |
| 24. |
Adasme MF, Linnemann KL, Bolz SN, et al. PLIP 2021: expanding the scope of the protein-ligand interaction profiler to DNA and RNA. Nucleic Acids Res 2021; 49: W530-4.
DOI PMID |
| 25. |
Kozlovskii I, Popov P. Spatiotemporal identification of druggable binding sites using deep learning. Commun Biol 2020; 3: 618.
DOI PMID |
| 26. | Li AA, Jiang T, Zhan M, et al. Pharmacological mechanism of Shenling Baizhu San in the treatment of knee osteoarthritis based on network pharmacology and molecular docking. Zhong Guo Zu Zhi Gong Cheng Yan Jiu 2022; 26: 205-12. |
| 27. | Song X, Zhang Y, Dai E, Wang L, Du H. Prediction of triptolide targets in rheumatoid arthritis using network pharmacology and molecular docking. IntImmunopharmacol 2020; 80: 106179. |
| 28. | Jian GH, Su BZ, Zhou WJ, Xiong H. Application of network pharmacology and molecular docking to elucidate the potential mechanism of Eucommiaulmoides-Radix Achyranthis Bidentatae against osteoarthritis. Bio Data Min 2020; 13: 12. |
| 29. |
Ye M, Luo G, Ye D, et al. Network pharmacology, molecular docking integrated surface plasmon resonance technology reveals the mechanism of Toujie Quwen granules against coronavirus disease 2019 pneumonia. Phytomedicine 2021; 85: 153401.
DOI URL |
| 30. |
Murthy SN, Cooper HS, Shim H, Shah RS, Ibrahim SA, Sedergran DJ. Treatment of dextran sulfate sodium-induced murine colitis by intracoloniccyclosporin. Dig Dis Sci 1993; 38: 1722-34.
DOI URL |
| 31. |
Bian M, Wang J, Wang Y, et al. Chicory ameliorates hyperuricemia via modulating gut microbiota and alleviating LPS/TLR4 axis in quail. Biomed Pharmacother 2020; 131: 110719.
DOI URL |
| 32. |
Boirivant M, Fuss IJ, Ferroni L, De Pascale M, Strober W. Oral administration of recombinant cholera toxin subunit B inhibits IL-12-mediated murine experimental (trinitrobenzene sulfonic acid) colitis. J Immunol 2001; 166: 3522-32.
PMID |
| 33. |
Yang J, Liu XX, Fan H, et al. Extracellular vesicles derived from bone marrow mesenchymal stem cells protect against experimental colitis via attenuating colon inflammation, oxidative stress and apoptosis. PLoS One 2015; 10: e0140551.
DOI URL |
| 34. | Liu P, Xu H, Shi Y, Deng L, Chen X. Potential molecular mechanisms of plantain in the treatment of gout and hyperuricemia based on network pharmacology. Evid Based Complement Alternat Med 2020; 2020: 3023127. |
| 35. |
Liang J, Chen S, Chen J, et al. Therapeutic roles of polysaccharides from Dendrobium Officinaleon colitis and its underlying mechanisms. Carbohydr Polym 2018; 185: 159-68.
DOI URL |
| 36. |
Molodecky NA, Soon IS, Rabi DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012; 142: 46-54.
DOI PMID |
| 37. | Peng MX, Xie HC. Research progress on regulating cytokines in the treatment of ulcerative colitis with Traditional Chinese Medicine and its effective ingredients. Hubei Min Zu Xue Yuan Xue Bao 2019; 36: 50-3. |
| 38. |
Wen S, Zhong Z, He L, et al. Network pharmacology dissection of multiscale mechanisms for Jiaoqi powder in treating ulcerative colitis. J Ethnopharmacol 2021; 275: 114109.
DOI URL |
| 39. |
Tatiya-Aphiradee N, Chatuphonprasert W, Jarukamjorn K. Ethanolic Garciniamangostana extract and α-mangostin improve dextran sulfate sodium-induced ulcerative colitis via the suppression of inflammatory and oxidative responses in ICR mice. J Ethnopharmacol 2021; 265: 113384.
DOI URL |
| 40. |
Kou FS, Shi L, Li JX, et al. Clinical evaluation of Traditional Chinese Medicine on mild active ulcerative colitis: a multi-center, randomized, double-blind, controlled trial. Medicine (Baltimore) 2020; 99: e21903.
DOI URL |
| 41. |
Ding P, Liu J, Li Q, et al. Investigation of the active ingredients and mechanism of Hudi enteric-coated capsules in DSS-induced ulcerative colitis mice based on network pharmacology and experimental verification. Drug Des Devel Ther 2021; 15: 4259-73.
DOI URL |
| 42. | Cui SX, Mao YF, Lu WM. Exploring the mechanism of Lianzhi Qingchang Tang in treating ulcerative colitis based on network pharmacology and molecular docking. Hunan Zhong Yi Za Zhi 2022; 38: 160-8. |
| 43. | Kim WS, Song HY, Han JM, Byun EB. GLM, a novel luteolin derivative, attenuates inflammatory responses in dendritic cells: therapeutic potential against ulcerative colitis. Biochem Biophys Res Commun 2019; 518: 87-93. |
| 44. |
Bischoff TA, Kelley CJ, Karchesy Y, Laurantos M, Nguyen-Dinh P, Arefi AG. Antimalarial activity of lactucin and lactucopicrin: sesquiterpene lactones isolated from Cichoriumintybus L. J Ethnopharmacol 2004; 95: 455-7.
DOI URL |
| 45. |
Zhang X, Lan D, Ning S, Ruan L. Anticancer action of lactucopicrin in SKMEL-5 human skin cancer cells is mediated via apoptosis induction, G2/M cell cycle arrest and downregulation of m = TOR/PI3K/AKT signalling pathway. J Buon 2018; 23: 224-8.
PMID |
| 46. |
Venkatesan R, Shim WS, Yeo EJ, Kim SY. Lactucopicrin potentiates neuritogenesis and neurotrophic effects by regulating Ca2+/CaMKII/ATF1 signaling pathway. J Ethnopharmacol 2017; 198: 174-83.
DOI PMID |
| 47. | Wesołowska A, Nikiforuk A, Michalska K, Kisiel W, Chojnacka-Wójcik E. Analgesic and sedative activities of lactucin and some lactucin-like guaianolides in mice. J Ethnopharmacol 2006; 107: 254-8. |
| 48. | He L, Weng H, Li Q, et al. Lactucopicrin inhibits cytoplasmic dynein-mediated NF-κB activation in inflammated macrophages and alleviates atherogenesis in apolipoprotein E-Deficient mice. Mol Nutr Food Res 2021; 65: e2000989. |
| 49. |
Zhang N, Chen X. Potential role of O-GlcNAcylation and involvement of PI3K/Akt1 pathway in the expression of oncogenic phenotypes of gastric cancer cells in vitro. Biotechnol Appl Biochem 2016; 63: 841-51.
DOI URL |
| 50. |
Zheng L, Wen XL, Dai YC. Mechanism of Jianpi Qingchang Huashi recipe in treating ulcerative colitis: a study based on network pharmacology and molecular docking. World J Clin Cases 2021; 9: 7653-70.
DOI PMID |
| 51. | Hirano T. IL-6 in inflammation, autoimmunity and cancer. IntImmunol 2021; 33: 127-48. |
| 52. |
Holbrook J, Lara-Reyna S, Jarosz-Griffiths H, McDermott M. Tumour necrosis factor signalling in health and disease. F1000Res 2019; 8: F1000 Faculty Rev-111.
DOI URL |
| 53. |
Kaisho T, Akira S. Toll-like receptors and their signaling mech-anism in innate immunity. Acta Odontol Scand 2001; 59: 124-30.
PMID |
| 54. |
Friedrich M, Pohin M, Powrie F. Cytokine networks in the pathophysiology of inflammatory bowel disease. Immunity 2019; 50: 992-1006.
DOI PMID |
| 55. |
Stucchi A, Reed K, O'Brien M, et al. A new transcription factor that regulates TNF-alpha gene expression, LITAF, is increased in intestinal tissues from patients with CD and UC. Inflamm Bowel Dis 2006; 12: 581-7.
PMID |
| 56. |
Schwanke RC, Marcon R, Meotti FC, et al. Oral administration of the flavonoid myricitrin prevents dextran sulfate sodium-induced experimental colitis in mice through modulation of PI3K/Akt signaling pathway. Mol Nutr Food Res 2013; 57: 1938-49.
DOI PMID |
| 57. |
Baregamian N, Rychahou PG, Hawkins HK, Evers BM, Chung DH. Phosphatidylinositol 3-kinase pathway regulates hypoxia-inducible factor-1 to protect from intestinal injury during necrotizing enterocolitis. Surgery 2007; 142: 295-302.
DOI URL |
| 58. |
Reddy SA, Huang JH, Liao WS. Phosphatidylinositol 3-kinase as a mediator of TNF-induced NF-kappa B activation. J Immunol 2000; 164: 1355-63.
DOI PMID |
| 59. |
Gustin JA, Ozes ON, Akca H, et al. Cell type-specific expression of the Ikappa B kinases determines the significance of phosphatidylinositol 3-kinase/Akt signaling to NF-kappa B activation. J Biol Chem 2004; 279: 1615-20.
DOI PMID |
| 60. |
Cook L, Stahl M, Han X, et al. Suppressive and gut-reparative functions of human type 1 T regulatory cells. Gastroenterology 2019; 157: 1584-98.
DOI PMID |
| [1] | YANG Qinjun, YIN Dandan, WANG Hui, GAO Yating, WANG Xinheng, WU Di, TONG Jiabing, WANG Chuanbo, LI Zegeng. Uncovering the action mechanism of Shenqi Tiaoshen formula (参芪调肾方) in the treatment of chronic obstructive pulmonary disease through network pharmacology, molecular docking, and experimental verification [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 770-783. |
| [2] | HU Yuanyuan, LIU Xinguang, ZHAO Peng, WU Jinyan, YAN Xinhua, HOU Runsu, WANG Xiangcheng, YANG Fan, TIAN Xinrong, LI Jiansheng. Integration of serum pharmacochemistry with network pharmacology to reveal the potential mechanism of Yangqing Chenfei formula (养清尘肺方) for the treatment of silicosis [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 784-793. |
| [3] | LUO Shan, YANG Fan, CHEN Yuanchun, ZHAO Ruoxi, LIU Haiye, GAO Fei, MA Wencan, GAO Weijuan, YU Wentao. Sanhua Tang (三化汤) protects against ischemic stroke by preventing blood-brain barrier injury: a network pharmacology and in vivo experiments [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 794-803. |
| [4] | FENG Yanchen, LIU Yali, DANG Xue, LIN Zixuan, ZHANG Yunke, CHE Zhiying, LI Xiang, PAN Xiaolong, LIU Feixiang, ZHENG Pan. Exploring the multicomponent synergy mechanism of Zuogui Wan (左归丸) on postmenopausal osteoporosis by a systems pharmacology strategy [J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 489-495. |
| [5] | WANG Yan, DENG Fanying, LIU Shiqi, WANG Yingli. Network pharmacology and experimental validation to reveal the pharmacological mechanisms of Sini decoction (四逆汤) against renal fibrosis [J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 362-372. |
| [6] | CHANG Fengjin, ZHOU Peng, LI Guoying, ZHANG Weizhi, ZHANG Yanyan, PENG Daiyin, CHEN Guangliang. Taohong Siwu decoction (桃红四物汤) ameliorates atherosclerosis in rats possibly through toll-like receptor 4/myeloid differentiation primary response protein 88/nuclear factor-κB signal pathway [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 103-112. |
| [7] | ZHI Guoguo, SHAO Bingjie, ZHENG Tianyan, JI Shaoxiu, LI Jingwei, DANG Yanni, LIU Feng, WANG Dong. Efficacy of Ganshuang granules (肝爽颗粒) on non-alcoholic fatty liver and underlying mechanism: a network pharmacology and experimental verification [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 122-130. |
| [8] | YANG Ye, CHEN Xiaoyang, YAO Junkai, HU Yueyao, WANG Wei. Efficacy of Danlou tablet (丹蒌片) on myocardial ischemia/ reperfusion injury assessed by network pharmacology and experimental verification [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 131-144. |
| [9] | ZHANG Qi, CHEN Dexuan, ZHU Guixiang, ZHANG Shihu, FENG Xiao, MA Chaoqun, ZHANG Yi. Efficacy of Tounongsan decoction (透脓散方) on pyogenic liver abscess: network pharmacology and clinical trial validation [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 145-155. |
| [10] | REN Hui, ZHAO Lintao, GAO Kai, YANG Yuanyuan, CUI Xiaomin, HU Jing, CHEN Zhiyong, LI Ye. Deciphering the chemical profile and pharmacological mechanism of Jinlingzi powder (金铃子散) against bile reflux gastritis using ultra-high performance liquid chromatography coupled with Q exactive focus mass spectrometry, network pharmacology, and molecular docking [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1209-1218. |
| [11] | YANG Xirui, ZHAO Hui, SHAN Muhammad, DONG Feixue, ZHANG Dandan, WANG Jixue, YUAN Xingxing. Efficacy of bioactive compounds of Chaihu (Radix Bupleuri Chinensis) on glaucomatous optic atrophy through interleukin-6/hypoxia inducible factor-1α signal pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1219-1226. |
| [12] | HAN Huagang, LI Ziqiang, OUYANG Jingfeng, WANG Tianquan, DONG Lingyan, CAO Junling. Mechanism of Lingbao Huxin Dan (灵宝护心丹) in the treatment of bradyarrhythmia complicated with coronary heart disease: a network pharmacology analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1001-1009. |
| [13] | PANG Fengtao, LI Kesong, ZHANG Yi, TANG Xiaopo, ZHOU Xinyao. Efficacy of Lushi Runzao decoction (路氏润燥汤) on ameliorating Sjogren's syndrome: a network pharmacology and experimental verification-based study [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 751-759. |
| [14] | JIA Lihua, KUANG Haodan, XU Yuan. Efficacy of Buzhong Yiqi decoction (补中益气汤) on benign prostatic hyperplasia and its possible mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 533-541. |
| [15] | JIA Lihong, TIE Defu, FAN Zhaohui, CHEN Dan, CHEN Qizhu, CHEN Jun, BO Huaben. Mechanism underlying Fanmugua (Fructus Caricae) leaf multicomponent synergistic therapy for anemia: data mining based on hematopoietic network [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 542-551. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

