Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (3): 448-457.DOI: 10.19852/j.cnki.jtcm.20240402.004
• Original Articles • Previous Articles Next Articles
Optimized new Shengmai powder (优化新生脉散方) inhibits myocardial fibrosis in heart failure by regulating the rat sarcoma/rapidly accelerated fibrosarcoma/mitogen-activated protein kinase kinase/extracellular regulated protein kinases signaling pathway
ZHANG Zeyu, JIA Zhuangzhuang, SONG Yuwei, ZHANG Xuan, WANG Ci, WANG Shuai, ZHANG Peipei, REN Qiuan, WANG Xianliang( ), MAO Jingyuan(
), MAO Jingyuan( )
)
- Department of Cardiovascular, the First Teaching Hospital of Tianjin University of Traditional Chinese Medicine, National Clinical Research Center for Chinese Medicine Acupuncture and Moxibustion, Tianjin 300381, China
-
Received:2023-01-16Accepted:2023-05-25Online:2024-04-02Published:2024-04-02 -
Contact:WANG Xianliang,MAO Jingyuan -
About author:Prof. MAO Jingyuan, Department of First Teaching Hospital of Tianjin University of Traditional Chinese Medicine, National Clinical Research Center for Chinese Medicine Acupuncture and Moxibustion, Tianjin 300381, China. jymao@126.com Telephone: +86-13920007825; +86-13820498886
Prof. WANG Xianliang, Department of First Teaching Hospital of Tianjin University of Traditional Chinese Medicine, National Clinical Research Center for Chinese Medicine Acupuncture and Moxibustion, Tianjin 300381, China. xlwang1981@126.com;
-
Supported by:Innovation Team Development Program of the Ministry of Education: Research on the Prevention and Treatment of Cardiovascular Diseases with Traditional Chinese Medicine(IRT-16R54)
Cite this article
ZHANG Zeyu, JIA Zhuangzhuang, SONG Yuwei, ZHANG Xuan, WANG Ci, WANG Shuai, ZHANG Peipei, REN Qiuan, WANG Xianliang, MAO Jingyuan. Optimized new Shengmai powder (优化新生脉散方) inhibits myocardial fibrosis in heart failure by regulating the rat sarcoma/rapidly accelerated fibrosarcoma/mitogen-activated protein kinase kinase/extracellular regulated protein kinases signaling pathway[J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 448-457.
share this article
| Primer name | Sequence (5’-3’) | Size (bp) | |
|---|---|---|---|
| COL Ⅰ | F | GAG AGG TGA ACA AGG TCC CG | 153 |
| R | AAA CCT CTC TCG CCT CTT GC | ||
| COL Ⅲ | F | AAG GCT GCA AGA TGG ATG CT | 95 |
| R | GTG CTT ACG TGG GAC AGT CA | ||
| α-SMA | F | CTATGAGGGCTATGCCTTGCC | 122 |
| R | GCTCAGCAGTAGTAACGAAGGA | ||
| c-Fos | F | GTCTTCCTTTGTCTTCACCTACCC | 384 |
| R | CCCTGCCTTCTCTGACTGCT | ||
| β-actin | F | GTCGTACCACTGGCATTGTG | 180 |
| R | TCTCAGCTGTGGTGGTGAAG |
Table 1 Primer sequence table
| Primer name | Sequence (5’-3’) | Size (bp) | |
|---|---|---|---|
| COL Ⅰ | F | GAG AGG TGA ACA AGG TCC CG | 153 |
| R | AAA CCT CTC TCG CCT CTT GC | ||
| COL Ⅲ | F | AAG GCT GCA AGA TGG ATG CT | 95 |
| R | GTG CTT ACG TGG GAC AGT CA | ||
| α-SMA | F | CTATGAGGGCTATGCCTTGCC | 122 |
| R | GCTCAGCAGTAGTAACGAAGGA | ||
| c-Fos | F | GTCTTCCTTTGTCTTCACCTACCC | 384 |
| R | CCCTGCCTTCTCTGACTGCT | ||
| β-actin | F | GTCGTACCACTGGCATTGTG | 180 |
| R | TCTCAGCTGTGGTGGTGAAG |
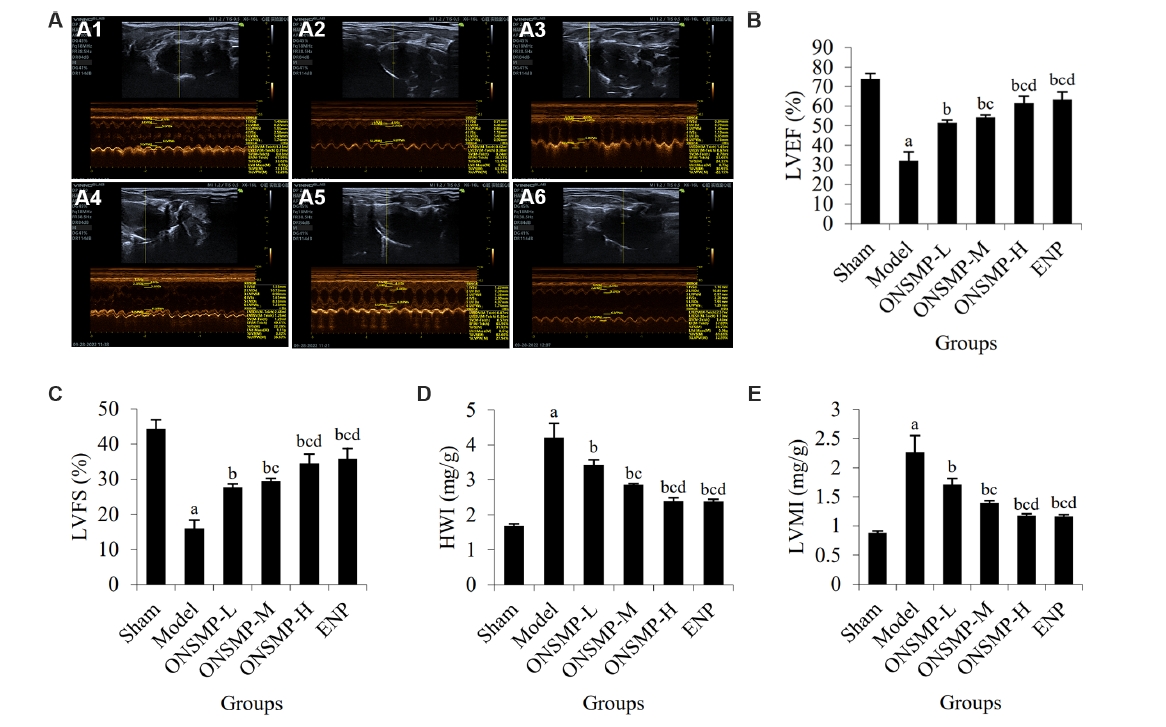
Figure 1 Comparison of cardiac function and organ coefficient in rats of each group A: typical echocardiographic image. A1-A6: the echocardiographic profile of sham (A1), model (A2), ONSMP-L (A3), ONSMP-M (A4), ONSMP-H (A5), and ENP (A6); B: statistic of LVEF of rats in each group; C: Statistic of LVFS of rats in each group; D: Statistic of HMI of rats in each group; E: statistic of LVMI of rats in each group. Sham group: sham operated; model group: ligation of the left anterior descending coronary artery without treatment; ONSMP-L: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 4.05 g crude drug/kg for 4 weeks; ONSMP-M: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 8.1 g crude drug/kg for 4 weeks; ONSMP-H: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 16.2 g crude drug/kg for 4 weeks; ENP: ligation of the left anterior descending coronary artery rats treated with ENP intragastrically at a daily dose of 0.9 g/kg for 4 weeks. ONSMP: Optimized New Shengmai powder; ENP: enalapril; LVEF: left ventricular ejection fractions; LVFS: left ventricular fractional shortening; HWI: heart weight index; LVMI: left ventricular mass index. Data represent the mean ± standard deviation using one-way analysis of variance (n = 6). Compared with the sham group, aP < 0.01; compared with the model group, bP < 0.01; compared with the ONSMP-L group, cP < 0.01; compared with the ONSMP-M group, dP < 0.01.
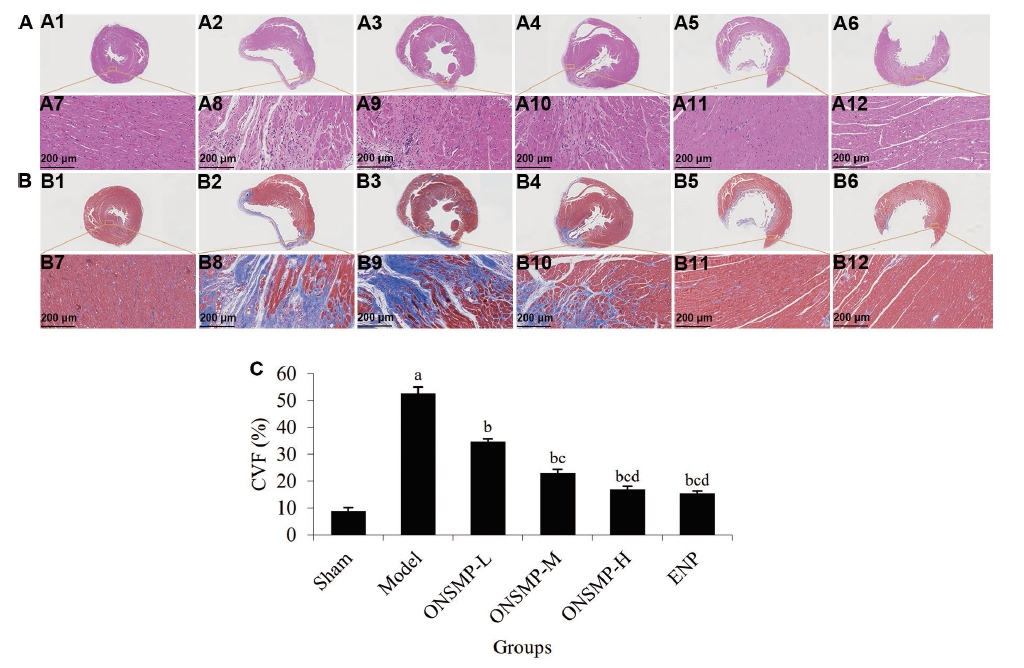
Figure 2 Pathological section results of myocardial tissue of rats in each group A: typical myocardial HE-stained section image. A1-A6: HE-stained section images of sham (A1), model (A2), ONSMP-L (A3), ONSMP-M (A4), ONSMP-H (A5), and ENP (A6) (× 1); A7-A12: HE-stained section images of sham (A7), model (A8), ONSMP-L (A9), ONSMP-M (A10), ONSMP-H (A11), and ENP (A12) (× 200). B: Typical myocardial masson-stained section image. B1-B6: masson-stained section images of sham (B1), model (B2), ONSMP-L (B3), ONSMP-M (B4), ONSMP-H (B5), and ENP (B6) (× 1); B7-B12: masson-stained section images of sham (B7), model (B8), ONSMP-L (B9), ONSMP-M (B10), ONSMP-H (B11), and ENP (B12) (× 200). C: statistics of CVF of rats in each group. Sham group: sham operated; model group: ligation of the left anterior descending coronary artery without treatment; ONSMP-L: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 4.05 g crude drug/kg for 4 weeks; ONSMP-M: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 8.1 g crude drug/kg for 4 weeks; ONSMP-H: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 16.2 g crude drug/kg for 4 weeks; ENP: ligation of the left anterior descending coronary artery rats treated with ENP intragastrically at a daily dose of 0.9 g/kg for 4 weeks. ONSMP: Optimized New Shengmai powder; ENP: enalapril; CVF: collagen volume fraction. Data represent the mean ± standard deviation using one-way analysis of variance (n = 3). Compared with the sham group, aP < 0.01; compared with the model group, bP < 0.01; compared with the ONSMP-L group, cP < 0.01; compared with the ONSMP-M group, dP < 0.01.
| Group | n | COL Ⅰ | COL Ⅲ |
|---|---|---|---|
| Sham | 6 | 34.5±1.4 | 20.4±1.6 |
| Model | 6 | 44.3±1.3a | 27.1±1.3a |
| ONSMP-L | 6 | 40.5±1.7c | 24.7±1.5b |
| ONSMP-M | 6 | 39.5±1.8c | 23.1±1.1c |
| ONSMP-H | 6 | 36.1±1.8cef | 22.4±0.8cd |
Table 2 Content of COL Ⅰ and COL Ⅲ in myocardial tissue of rats in each group (μg/L, $\bar{x} \pm s$)
| Group | n | COL Ⅰ | COL Ⅲ |
|---|---|---|---|
| Sham | 6 | 34.5±1.4 | 20.4±1.6 |
| Model | 6 | 44.3±1.3a | 27.1±1.3a |
| ONSMP-L | 6 | 40.5±1.7c | 24.7±1.5b |
| ONSMP-M | 6 | 39.5±1.8c | 23.1±1.1c |
| ONSMP-H | 6 | 36.1±1.8cef | 22.4±0.8cd |
| Group | n | COL Ⅰ/β-actin | COL Ⅲ/β-actin | α-SMA/β-actin | c-Fos/β-actin |
|---|---|---|---|---|---|
| Sham | 3 | 1.04±0.29 | 1.04±0.28 | 1.01±0.15 | 1.08±0.40 |
| Model | 3 | 7.69±0.50a | 7.03±0.74a | 7.36±1.62a | 6.45±0.70a |
| ONSMP-L | 3 | 4.85±0.42b | 4.12±0.22b | 5.02±0.70b | 2.92±0.16b |
| ONSMP-M | 3 | 2.73±0.33bc | 2.19±0.30bc | 3.26±0.68bc | 2.05±0.33bc |
| ONSMP-H | 3 | 1.32±0.11bcd | 1.30±0.05bcd | 1.79±0.32bcd | 1.35±0.05bcd |
Table 3 mRNA of COL Ⅰ, COL Ⅲ, α-SMA, and c-Fos in myocardial tissue of rats in each group ($\bar{x} \pm s$)
| Group | n | COL Ⅰ/β-actin | COL Ⅲ/β-actin | α-SMA/β-actin | c-Fos/β-actin |
|---|---|---|---|---|---|
| Sham | 3 | 1.04±0.29 | 1.04±0.28 | 1.01±0.15 | 1.08±0.40 |
| Model | 3 | 7.69±0.50a | 7.03±0.74a | 7.36±1.62a | 6.45±0.70a |
| ONSMP-L | 3 | 4.85±0.42b | 4.12±0.22b | 5.02±0.70b | 2.92±0.16b |
| ONSMP-M | 3 | 2.73±0.33bc | 2.19±0.30bc | 3.26±0.68bc | 2.05±0.33bc |
| ONSMP-H | 3 | 1.32±0.11bcd | 1.30±0.05bcd | 1.79±0.32bcd | 1.35±0.05bcd |
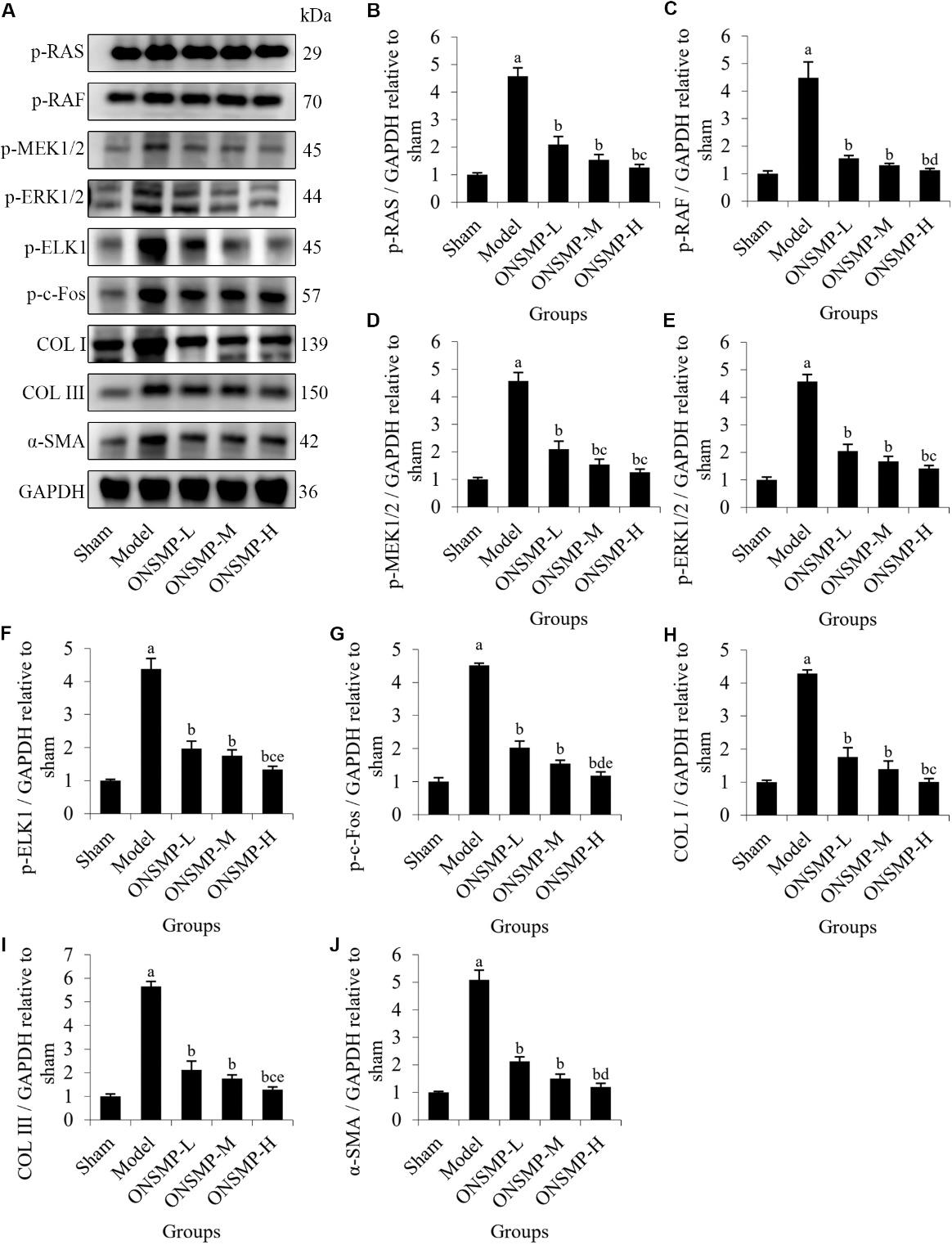
Figure 3 ONSMP inhibited the RAS/RAF/MEK/ERK pathway and downstream protein expression A: Western blotting representative images of p-RAS, p-RAF, p-MEK1/2, p-ERK1/2, p-ELK1, p-c-Fos, COL I, COL Ⅲ, and α-SMA. B: protein expression levels of p-RAS; C: protein expression levels of p-RAF; D: protein expression levels of p-MEK1/2; E: protein expression levels of p-ERK1/2; F: protein expression levels of p-ELK1; G: protein expression levels of p-c-Fos; H: protein expression levels of COL I; I: protein expression levels of COL Ⅲ; J: protein expression levels of α-SMA. Sham group: sham operated; model group: ligation of the left anterior descending coronary artery without treatment; ONSMP-L: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 4.05 g crude drug/kg for 4 weeks; ONSMP-M: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 8.1 g crude drug/kg for 4 weeks; ONSMP-H: ligation of the left anterior descending coronary artery rats treated with ONSMP intragastrically at a daily dose of 16.2 g crude drug/kg for 4 weeks; ENP: ligation of the left anterior descending coronary artery rats treated with ENP intragastrically at a daily dose of 0.9 g/kg for 4 weeks. p-RAS: phosphor-rat sarcoma; p-RAF: phosphor-rapidly accelerated fibrosarcoma; p-MEK1/2: phosphor-mitogen-activated protein kinase kinase 1/2; p-ERK1/2: phosphor-extracellular regulated protein kinases; p-ELK1: phosphor-ETS-like-1 transcription factor; p-c-Fos: phosphor-c-Fos proto-oncogene; COL I: collagen I; COL Ⅲ: collagen Ⅲ; α-SMA: α-smooth muscle actin; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; ONSMP: optimized New Shengmai powder; ENP: enalapril. Data represent the mean ± standard deviation using one-way analysis of variance (n = 3). Compared with the sham group, aP < 0.01; compared with the model group, bP < 0.01; compared with the ONSMP-L group, cP < 0.05, dP < 0.01; compared with the ONSMP-M group, eP < 0.05.
| 1. |
McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2021; 42: 3599-726.
DOI PMID |
| 2. | Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics-2022 update: a report from the american heart association. Circulation 2022; 145: e153-639. |
| 3. | González A, Schelbert EB, Díez J, Butler J. Myocardial interstitial fibrosis in heart failure: biological and translational perspectives. J Am Coll Cardiol 2018; 71:1696-706. |
| 4. |
Marunouchi T, Nakashima M, Ebitani S, et al. Hsp90 inhibitor attenuates the development of pathophysiological cardiac fibrosis in mouse hypertrophy via suppression of the calcineurin-NFAT and c-Raf-Erk pathways. J Cardiovasc Pharmacol 2021; 77: 822-9.
DOI PMID |
| 5. |
Chen M, Li H, Wang G, Shen X, Zhao S, Su W. Atorvastatin prevents advanced glycation end products (AGEs)-induced cardiac fibrosis via activating peroxisome proliferator-activated receptor gamma (PPAR-γ). Metabolism 2016; 65: 441-53.
DOI URL |
| 6. | García-Martín A, Navarrete C, Garrido-Rodríguez M, et al. EHP-101 alleviates angiotensin Ⅱ-induced fibrosis and inflammation in mice. Biomed Pharmacother 2021; 142: 112007. |
| 7. | Mao JY, Zhu MJ. Expert consensus on Traditional Chinese Medicine diagnosis and treatment of chronic heart failure. Zhong Yi Za Zi 2014; 55: 1258-60. |
| 8. | Wang XL, Yuan Y, Mao JY, et al. A single-case crossover randomized controlled study of optimized new Shengmai powder combined with Western medicine in the treatment of chronic heart failure. Zhong Yi Za Zi 2015; 56: 1849-53. |
| 9. | Pang L, Mao JY, Wan CJ, Wang S. Study on the protective effect of New Shengmai powder optimized prescription on adriamycin-induced myocardial injury in mice. Jilin Zhong Yi Yao 2012; 32: 712-5. |
| 10. | Zhang ZY, Wang XL, Wang S, Jia ZZ, Yan HF, Mao JY. Based on network pharmacology and molecular docking technology to explore the mechanism of optimized new Shengmai powder in anti-myocardial fibrosis in chronic heart failure. Henan Zhong Yi 2022; 42: 1032-9. |
| 11. |
Frangogiannis NG. Cardiac fibrosis. Cardiovasc Res 2021; 117: 1450-88.
DOI PMID |
| 12. |
Piek A, Silljé HHW, de Boer RA. The vicious cycle of arrhythmia and myocardial fibrosis. Eur J Heart Fail 2019; 21: 492-4.
DOI PMID |
| 13. |
Park S, Nguyen NB, Pezhouman A, Ardehali R. Cardiac fibrosis: potential therapeutic targets. Transl Res 2019; 209: 121-37.
DOI PMID |
| 14. | Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the management of heart failure: a report of the american college of cardiology/american heart association joint committee on clinical practice guidelines. Circulation 2022; 145: e895-1032. |
| 15. |
Talman V, Ruskoaho H. Cardiac fibrosis in myocardial infarction-from repair and remodeling to regeneration. Cell Tissue Res 2016; 365: 563-81.
DOI PMID |
| 16. |
Zhou R, Han B, Xia C, Zhuang X. Membrane-associated periodic skeleton is a signaling platform for RTK transactivation in neurons. Science 2019; 365: 929-34.
DOI PMID |
| 17. |
Jain R, Watson U, Vasudevan L, Saini DK. ERK activation pathways downstream of GPCRs. Int Rev Cell Mol Biol 2018; 338: 79-109.
DOI PMID |
| 18. |
Chen Z, Oh D, Dubey AK, et al. EGFR family and Src family kinase interactions: mechanics matters. Curr Opin Cell Biol 2018; 51: 97-102.
DOI URL |
| 19. | Hu M, He W, Gao P, et al. Virus-induced accumulation of intracellular bile acids activates the TGR5-β-arrestin-SRC axis to enable innate antiviral immunity. Cell Res 2019; 29: 193-205. |
| 20. | Smith JS, Pack TF, Inoue A, et al. Noncanonical scaffolding of G(αi) and β-arrestin by G protein-coupled receptors. Science 2021;371: eaay1833. |
| 21. | Arkun Y, Yasem Mi. Dynamics and control of the ERK signaling pathway: sensitivity, bistability, and oscillations. PloS One 2018; 13: e195513. |
| 22. |
Xie K, Colgan LA, Dao MT, et al. NF1 is a direct G protein effector essential for opioid signaling to Ras in the striatum. Curr Biol 2016; 26: 2992-3003.
DOI PMID |
| 23. | Rodríguez-Álvarez FJ, Jiménez-Mora E, Caballero M, Gallego B, Chiloeches A, Toro MJ. Somatostatin activates Ras and ERK1/2 via a G protein βγ-subunit-initiated pathway in thyroid cells. Mol Cell Biochem 2016; 411: 253-60. |
| 24. |
Cui N, Li L, Feng Q, Ma HM, Lei D, Zheng PS. Hexokinase 2 promotes cell growth and tumor formation through the Raf/MEK/ERK signaling pathway in cervical cancer. Front Oncol 2020; 10: 581208.
DOI URL |
| 25. |
Cheng Y, Zhu Y, Xu J, et al. PKN2 in colon cancer cells inhibits M2 phenotype polarization of tumor-associated macrophages via regulating DUSP6-ERK1/2 pathway. Mol Cancer 2018; 17: 13.
DOI PMID |
| 26. |
Zhou Y, Shiok TC, Richards AM, Wang P. MicroRNA-101a suppresses fibrotic programming in isolated cardiac fibroblasts and in vivo fibrosis following trans-aortic constriction. J Mol Cell Cardiol 2018; 121: 266-76.
DOI PMID |
| 27. |
Ulrich M, Wissenbach U, Thiel G. The super-cooling compound icilin stimulates c-Fos and Egr-1 expression and activity involving TRPM8 channel activation, Ca2+ ion influx and activation of the ternary complex factor ELK1. Biochem Pharmacol 2020; 177: 113936.
DOI URL |
| 28. |
Tallquist MD. Cardiac fibroblast diversity. Annu Rev Physiol 2020; 82: 63-78.
DOI PMID |
| 29. |
Tallquist MD, Molkentin JD. Redefining the identity of cardiac fibroblasts. Nat Rev Cardiol 2017; 14: 484-91.
DOI PMID |
| 30. |
Deng T, Karin M. c-Fos transcriptional activity stimulated by H-Ras-activated protein kinase distinct from JNK and ERK. Nature 1994. 371: 171-5.
DOI |
| 31. | Chen RH, Abate C, Blenis J. Phosphorylation of the c-Fos transrepression domain by mitogen-activated protein kinase and 90-kDa ribosomal S6 kinase. Proc Natl Acad Sci USA 1993; 9: 10952-6. |
| 32. |
Peng B, Zhu H, Ma L, Wang YL, Klausen C, Leung PC. AP-1 transcription factors c-Fos and c-JUN mediate GnRH-induced cadherin-11 expression and trophoblast cell invasion. Endocrinology 2015; 156: 2269-77.
DOI PMID |
| 33. | Hattori Y, Hattori S, Akimoto K, et al. Globular adiponectin activates nuclear factor-kappa B and activating protein-1 and enhances angiotensin Ⅱ-induced proliferation in cardiac fibroblasts. Diabetes 2007; 56: 804-8. |
| 34. |
Kovary K, Bravo R. The Jun and Fos protein families are both required for cell cycle progression in fibroblasts. Mol Cell Biol 1991; 11: 4466-72.
DOI PMID |
| 35. |
Ou W, Ni N, Zuo R, et al. Cyclin D1 is a mediator of gastrointestinal stromal tumor KIT-independence. Oncogene 2019; 38: 6615-29.
DOI |
| 36. |
Rodriguez-Pascual F, Slatter DA. Collagen cross-linking: insights on the evolution of metazoan extracellular matrix. Sci Rep 2016; 6: 37374.
DOI PMID |
| 37. |
Lee W, Mitchell P, Tjian R. Purified transcription factor AP-1 interacts with TPA-inducible enhancer elements. Cell 1987; 49: 741-52.
DOI PMID |
| 38. |
Jia J, Ye T, Cui P, Hua Q, Zeng H, Zhao D. AP-1 transcription factor mediates VEGF-induced endothelial cell migration and proliferation. Microvasc Res 2016; 105: 103-8.
DOI PMID |
| 39. |
Ryseck RP, Bravo R. c-JUN, JUN B, and JUN D differ in their binding affinities to AP-1 and CRE consensus sequences: effect of FOS proteins. Oncogene 1991; 6: 533-42.
PMID |
| 40. |
Kovary K, Bravo R. Existence of different Fos/Jun complexes during the G0-to-G1 transition and during exponential growth in mouse fibroblasts: differential role of Fos proteins. Mol Cell Biol 1992; 12: 5015-23.
DOI PMID |
| 41. | Humeres C, Shinde AV, Hanna A, et al. Smad7 effects on TGF-β and ErbB2 restrain myofibroblast activation and protect from postinfarction heart failure. J Clin Invest 2022; 13: e146926. |
| [1] | CHANG Junli, ZHAO Fulai, SUN Xingyuan, MA Xiaoping, ZHAO Peng, ZHOU Chujie, SHI Binhao, GU Wenchao, WANG Yongjun, YANG Yanping. Polyphyllin I enhances tumor necrosis factor-related apoptosis-inducing ligand-induced inhibition of human osteosarcoma cell growth via downregulating the Wnt/β-catenin pathway [J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 251-259. |
| [2] | DU Zhongheng, CONG Wenjie, TANG Kejing, ZHENG Qiqi, SONG Zhiwei, CHEN Yong, YANG Su, ZHANG Chunwu, YE Tianshen. Electroacupuncture stimulating Zusanli (ST36), Sanyinjiao (SP6) in mice with collagen-induced arthritis leads to adenosine A2A receptor-mediated alteration of p38α mitogen-activated protein kinase signaling and inhibition of osteoclastogenesis [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1103-1109. |
| [3] | TIAN Xinrong, HOU Runsu, LIU Xinguang, ZHAO Peng, TIAN Yange, LI Jiansheng. Yangqing Chenfei formula (养清尘肺方) alleviates crystalline silica induced pulmonary inflammation and fibrosis by suppressing macrophage polarization [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1126-1139. |
| [4] | CHEN Yunhu, YIN Moqing, FAN Lihua, JIANG Xuechun, ZHANG Tao, ZHU Xingyu, XU Hongfeng. Mirror-like tongue is an important predictor of acute heart failure: a cohort study of acute heart failure in Chinese patients [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1243-1251. |
| [5] | ZHANG Xiaoying, WANG Ruixuan, WANG Yiqing, XU Fanxing, YAN Tingxu, WU Bo, ZHANG Ming, JIA Ying. Spinosin protects Neuro-2a/APP695 cells from oxidative stress damage by inactivating p38 [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 868-875. |
| [6] | ZHU Lingyan, WEI Yihong, WANG Youhua, YANG Jianmei, LI Jiawei, CAO Min, ZHOU Duan. Protective efficacy of Shenge San (参蛤散) on mitochondria in H9c2 cardiomyocytes [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 892-899. |
| [7] | ZHAO Jie, WANG Li, CAO Ai-li, WANG Yun-man, CHI Yang-feng, WANG Yi, WANG Hao, PENG Wen. Huangqi decoction (黄芪汤) attenuates renal interstitial fibrosis via transforming growth factor-β1/mitogen-activated protein kinase signaling pathways in 5/6 nephrectomy mice [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 723-731. |
| [8] | ZHANG Jiaying, WEI Xiangxiang, LI Xuefeng, YUAN Yang, DOU Yinghuan, SHI Yanbin, XIE Ping, ZHOU Mengru, ZHAO Junnan, LI Miao, ZHANG Shuwen, ZHU Rui, TIAN Ying, TAN Hao, TIAN Feifei. Shunxin decoction (顺心组方) improves diastolic function in rats with heart failure with preserved ejection fraction induced by abdominal aorta constriction through cyclic guanosine monophosphate-dependent protein kinase Signaling Pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 764-772. |
| [9] | CHEN Yunhu, FAN Lihua, ZHANG Tao, LIU Xueqian. Effectiveness of Zhuling decoction (猪苓汤) on diuretic resistance in patients with heart failure: a randomized, controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(3): 439-445. |
| [10] | HU Xijiao, LI Shuoxi, YANG Dongxia, GU Na, LIU Jinzhe, WANG Yawen, LIU Li, SUN Yiming. Modified Gexiazhuyu decoction (膈下逐瘀汤加减方) alleviates chronic salpingitis via p38 signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 213-220. |
| [11] | WANG Wei, LI Qingling, MA Qiang, XIA Ran, GAO Bing, WANG Yi, WANG Jing. Effects of moxibustion at bilateral Feishu (BL13) and Xinshu (BL15) combined with benazepril on myocardial cells apoptosis index and apoptosis-related proteins cytochrome c and apoptosis-inducing factor in rats with chronic heart failure [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 227-233. |
| [12] | Jiaru LIN, Li WANG, Bo CHEN, Santao OU, Jianhua QIN, Junming FAN. Shenweifang-containing serum inhibits transforming growth factor-β1-induced myofibroblast differentiation in normal rat kidney interstitial fibroblast cells [J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 39-48. |
| [13] | WANG Shengchun, JIANG Yiming, QIU Lirong, SU Meng. Efficacy of needling acupoints of Guanyuan(CV4), Sanyinjiao(SP6), Zusanli(ST36), Pishu(BL20), Shenshu(BL23), Zigong(EX-CA1) on expression of p38 mitogen-activated protein kinase in ovarian tissue in rats with premature ovarian failure induced by cyclophosp [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 953-958. |
| [14] | CHEN Tzufan, CHEN Chuntzu, HUANG Yuanli, BASKARAN Rathinasamy, TSAI Jeffrey J.P., HU Rouhmei. Ethanolic extract of Puhuang(Pollen Typhae) modulates lipopolysaccharide-induced inflammatory response through inducible nitric oxide synthase/cyclooxygenase-2 signaling in RAW 264.7 macrophages [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 836-844. |
| [15] | WU Mingyun, YU Jianer, BAI Li, XUE Zheng, JIANG Shenhua, LI Liqing, PIAO Xiang, XU Wanchao, WANG Jiani, SHEN Qian. Pingchuan formula(平喘方) improves allergic asthma in mice through inhibiting nuclear factor-kappa B/mitogen-activated protein kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 883-890. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

