Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 306-315.DOI: 10.19852/j.cnki.jtcm.2026.02.004
• Original Articles • Previous Articles Next Articles
No. 2 Kangxianling decoction (抗纤灵二号方) protects against renal ischemia/reperfusion injury by hypoxia-inducible factor activation
YU Kena1, ZHONG Liping4, LIN Xiaomeng1, CHEN Jian5, HE Liqun2,3( ), CAI Xudong1(
), CAI Xudong1( )
)
- 1
Department of Nephrology ,Ningbo Municipal Hospital of Traditional Chinese Medicine (TCM), Affiliated Hospital of Zhejiang Chinese Medical University Ningbo 315000, China
2Shanghai University of Traditional Chinese Medicine Shanghai 200120, China
3Department of Nephrology ,Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine Shanghai 200013, China
4Department of Nephrology ,Hubei Provincial Hospital of Integrated Traditional Chinese and Western Medicine Wuhan 430000, China
5Department of Traditional Medicine ,Shanghai Integrated Traditional Chinese and Western Medicine Hospital Affiliated to Shanghai University of Traditional Chinese Medicine Shanghai 200082, China
-
Received:2024-11-19Accepted:2025-06-10Online:2026-04-15Published:2026-04-04 -
Contact:Prof. CAI Xudong, Department of Nephrology, Ningbo Municipal Hospital of Traditional Chinese Medicine (TCM), Affiliated Hospital of Zhejiang Chinese Medical University, Ningbo 315000, China, xudongcai1979@hotmail.com; Prof. HE Liqun, Shanghai University of Traditional Chinese Medicine, Shanghai 200120, China; Department of Nephrology, Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 200013, China, heliqun59@163.com. Telephone: +86-574-89085041 -
Supported by:National Natural Science Foundation of China: Research on The Pathogenesis of Renal Micro Syndrome Accumulation in Chronic Renal Failure through Mammalian Target of Rapamycin Signaling Pathway and the Anti-fibrotic Effect of Kangxianling Decoction on Gene Knockout Mice(81373615);Zhejiang Natural Science Foundation of China: Mechanism Study of Wenyangxiaozheng Decoction Alleviating Cellular Senescence and Ameliorating Renal Fibrosis by Regulating the Phosphatidylinositol 3-kinase/Protein Kinase B(LBY24H290002)
Cite this article
YU Kena, ZHONG Liping, LIN Xiaomeng, CHEN Jian, HE Liqun, CAI Xudong. No. 2 Kangxianling decoction (抗纤灵二号方) protects against renal ischemia/reperfusion injury by hypoxia-inducible factor activation[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 306-315.
share this article
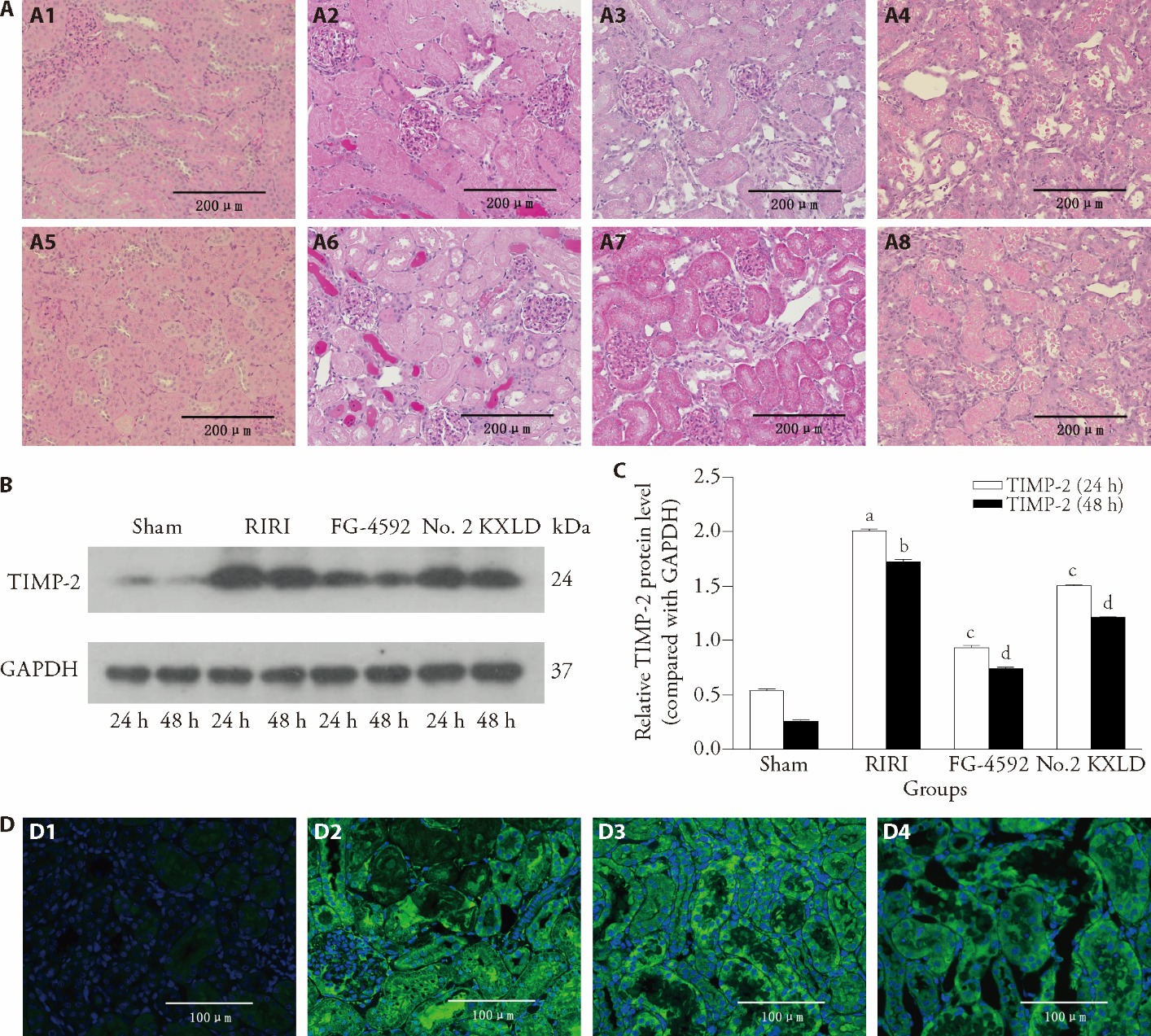
Figure 1 No. 2 KXLD attenuated the tubular injury in RIRI rats A: PAS staining in kidneys; B: Western blot analysis of renal TIMP-2; C: quantitative analysis of TIMP-2 protein. D: immunofluorescence staining of NGAL (green: NGAL; blue: DAPI); A1: sham group at 24 h after I/R; A2: I/R group at 24 h after I/R; A3: FG-4592 group at 24 h after I/R; A4: No. 2 KXLD group at 24 h after I/R; A5: sham group at 48 h after I/R; A6: I/R group at 48 h after I/R; A7: FG-4592 group at 48 h after I/R; A8: No. 2 KXLD group at 48 h after I/R; D1: sham group; D2: RIRI group; D3: FG-4592 group; D4: No. 2 KXLD group. Sham group: sham surgery; RIRI: renal I/R; FG-4592: FG-4592 + renal I/R, 10 mg·kg-1·d-1; No. 2 KXLD: No. 2 KXLD + renal I/R, 30 g·kg-1·d-1, i.g. TIMP-2: tissue inhibitor of metalloproteinases-2; NGAL: neutrophil gelatinase-associated lipocalin; PAS: periodic acid-schiff; RIRI: renal ischemia/reperfusion injury; No. 2 KXLD: No. 2 Kangxianling decoction; DAPI: 4',6-diamidino-2-phenylindole; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; RT-qPCR: reverse transcription quantitative polymerase chain reaction. Data were analyzed using a one-way analysis of variance test with a post hotc (least significant difference analysis) for comparison of multiple independent sample groups. Data were expressed as mean ± standard deviation (n = 3). a P < 0.01 vs sham group at 24 h after I/R; b P < 0.01 vs sham group at 48 h after I/R; c P < 0.01 vs RIRI group at 24 h after I/R; d P < 0.01 vs RIRI group at 48 h after I/R.

Figure 2 Effect of No. 2 KXLD on apoptosis in RIRI rats A: representative images of TUNEL staining at 48 h after I/R (630 × magnification; red: TUNEL; blue: DAPI); B: Western blot analysis of Bcl-2, Bax, caspase-3 in kidneys. C: Western blot analysis of caspase-9, cleaved caspase-9/3 in kidneys. A1: sham group; A2: RIRI group; A3: FG-4592 group; A4: No. 2 KXLD group. Sham group: sham surgery, RIRI: renal I/R, FG-4592: FG-4592 + renal I/R, 10 mg·kg-1·d-1; No. 2 KXLD: No. 2 KXLD + renal I/R, 30 g·kg-1·d-1, i.g. DAPI: 4',6-diamidino-2-phenylindole; RIRI: renal ischemia/reperfusion injury; No. 2 KXLD: No. 2 Kangxianling decoction; Bcl-2: B-cell lymphoma 2; Bax: Bcl-2-associated X protein; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; RT-qPCR: reverse transcription quantitative polymerase chain reaction. Data were expressed as mean ± standard deviation (n = 3). Data were analyzed using a one-way analysis of variance test with a post hotc (least significant difference analysis) for comparison of multiple independent sample groups.

Figure 3 No. 2 KXLD treatment reduced the injury of H/R-induced HK-2 cells by activating HIF activation A: representative images of HK-2 cells in each group (× 200); B: CCK-8 assay was used to analyze the cell viability of HK-2 cells; C: RT-qPCR analyses demonstrated a marked reduction of HIF-1ɑ expression in si-HIF-1ɑ HK-2 cells; D: Western blot analysis of NGAL and TIMP-2 in cells; E: Western blot analysis of HIF-1α, EPO and HO-1 in cells; F: immunofluorescence staining results of KIM-1 in HK-2 cells (400 ×; red: KIM-1; blue: DAPI). A1, F1: control group; A2, F2: H/R group; A3, F3: blank serum group; A4, F4: FG-4592 group; A5, F5: si-HIF-1α + No. 2 KXLD group; A6, F6: No. 2 KXLD group. Control group: normal culture; H/R group: H/R; blank serum group: H/R + blank serum; FG-4592: H/R + FG4592, 10 μg/mL; si- HIF-1α + No. 2 KXLD group: si-HIF-1α + H/R + 25% No. 2 KXLD; No. 2 KXLD group: H/R + 25% No. 2 KXLD. No. 2 KXLD: No. 2 Kangxianling decoction; H/R: hypoxia/reoxygenation; NGAL: neutrophil gelatinase-associated lipocalin; KIM-1: kidney injury molecule-1; TIMP-2: tissue inhibitor of metalloproteinases-2; HIF-1α: hypoxia-inducible factor; EPO: erythropoietin; HO-1: heme oxygenase 1; RT-qPCR: reverse transcription quantitative polymerase chain reaction; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; DAPI: 4',6-diamidino-2-phenylindole; CCK-8: cell counting kit-8. Data were expressed as mean ± standard deviation (n = 3). aP < 0.01 vs control group; bP < 0.01 vs H/R group; cP < 0.01 vs si-NC group.
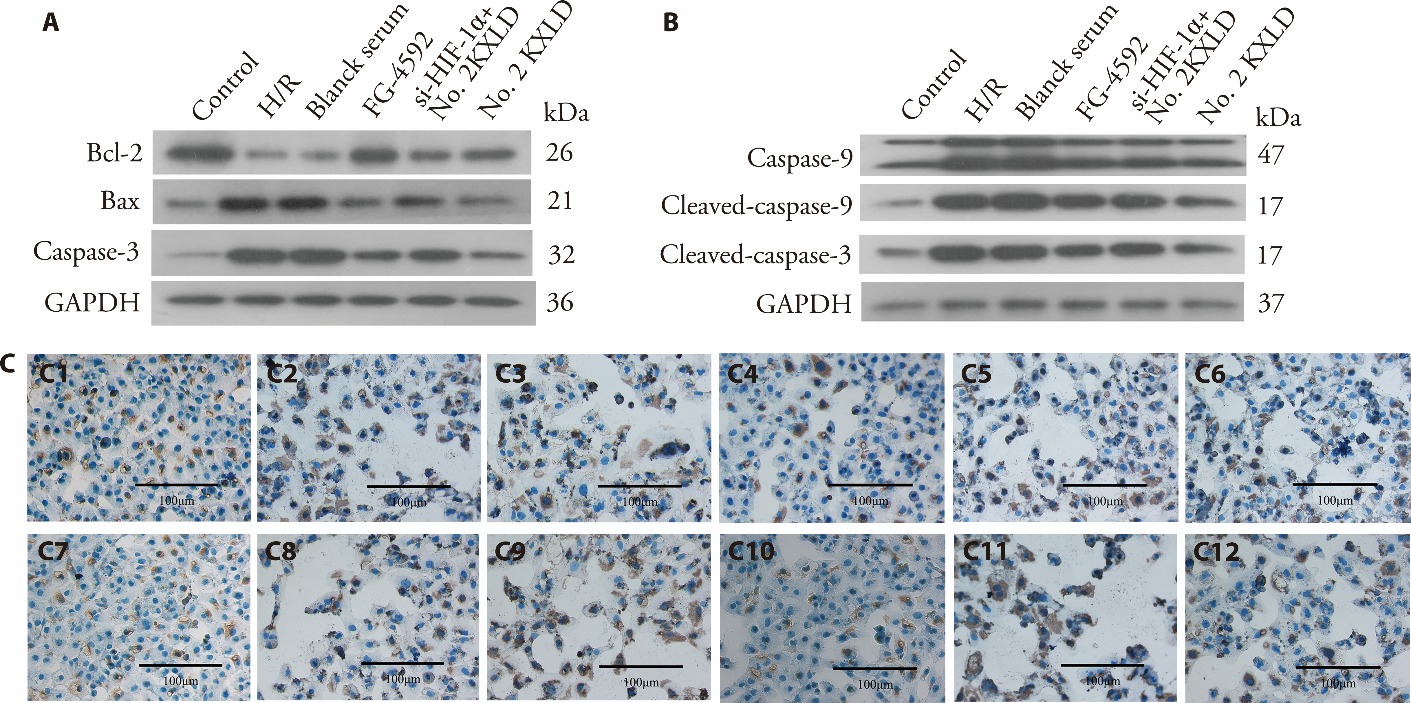
Figure 4 Inflammation and apoptosis were suppressed by No. 2 KXLD in H/R-induced HK-2 cells A: Western blot analysis of Bcl-2, Bax, caspase-3 in cells; B: Western blot analysis of caspase-9, cleaved caspase-9/3 in cells; C: representative immunohistochemistry staining of TNF-α and IL-6 in HK-2 cells (×200); C1: TNF-α in sham group; C2: TNF-α expression in H/R group; C3: TNF-α expression in blank serum group; C4: TNF-α expression in FG-4592 group; C5: TNF-α expression in si-HIF-1α + No. 2 KXLD group; C6: TNF-α expression in No. 2 KXLD group; C7: IL-6 expression in sham group; C8: IL-6 expression in H/R group; C9: IL-6 expression in blank serum group; C10: IL-6 expression in FG-4592 group; C11: IL-6 expression in si-HIF-1α + No. 2 KXLD group; C12: IL-6 expression in No. 2 KXLD group. Control group: normal culture; H/R group: H/R; blank serum group: H/R + blank serum; FG-4592: H/R + FG4592, 10 μg/mL; si-HIF-1α + No. 2 KXLD group: si- HIF-1α + H/R + 25% No. 2 KXLD; No. 2 KXLD group: H/R + 25% No. 2 KXLD. No. 2 KXLD: No. 2 Kangxianling decoction; H/R: hypoxia/reoxygenation; Bcl-2: B-cell lymphoma 2; Bax: Bcl-2-associated X protein; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; RT-qPCR: reverse transcription quantitative polymerase chain reaction. Data were expressed as mean ± standard deviation (n = 3).
| 1. |
Gifford CC, Tang J, Costello A, et al. Negative regulators of TGF-β1 signaling in renal fibrosis; pathological mechanisms and novel therapeutic opportunities. Clin Sci (Lond) 2021; 135: 275-303.
DOI PMID |
| 2. |
Martin IV, Borkham-Kamphorst E, Zok S, et al. Platelet-derived growth factor (PDGF)-C neutralization reveals differential roles of PDGF receptors in liver and kidney fibrosis. Am J Pathol 2013; 182: 107-17.
DOI PMID |
| 3. |
Shlipak MG, Day EC. Biomarkers for incident CKD: a new framework for interpreting the literature. Nat Rev Nephrol 2013; 9: 478-83.
DOI PMID |
| 4. |
Wang Y, Zou Z, Jin J, et al. Urinary TIMP-2 and IGFBP7 for the prediction of acute kidney injury following cardiac surgery. BMC Nephrol 2017; 18: 177.
DOI PMID |
| 5. |
Cummings BS, Schnellmann RG. Cisplatin-induced renal cell apoptosis: caspase 3-dependent and -independent pathways. J Pharmacol Exp Ther 2002; 302: 8-17.
DOI PMID |
| 6. |
Wei Q, Dong G, Franklin J, Dong Z, et al. The pathological role of Bax in cisplatin nephrotoxicity. Kidney Int 2007; 72: 53-62.
DOI PMID |
| 7. |
Korsmeyer SJ. Bcl-2 initiates a new category of oncogenes: regulators of cell death. Blood 1992; 80: 879-86.
PMID |
| 8. |
Zhong L, Sarafian T, Kane DJ, et al. Bcl-2 inhibits death of central neural cells induced by multiple agents. Proc Natl Acad Sci U S A 1993; 90: 4533-7.
DOI URL |
| 9. |
Hausenloy DJ, Candilio L, Evans R, et al. Remote ischemic preconditioning and outcomes of cardiac surgery. N Engl J Med 2015; 373: 1408-17.
DOI URL |
| 10. |
Meybohm P, Bein B, Brosteanu O, et al. A multicenter trial of remote ischemic preconditioning for heart surgery. N Engl J Med 2015; 373: 1397-407.
DOI URL |
| 11. |
Hausenloy DJ. Cardioprotection techniques: preconditioning, postconditioning and remote conditioning (basic science). Curr Pharm Des 2013; 19: 4544-63.
DOI URL |
| 12. |
Miao A, Liang J, Yao L, et al. Hypoxia-inducible factor prolyl hydroxylase inhibitor roxadustat (FG-4592) protects against renal ischemia/reperfusion injury by inhibiting inflammation. Ren Fail 2021; 43: 803-10.
DOI PMID |
| 13. |
Zhang M, Dong R, Yuan J, et al. Roxadustat (FG-4592) protects against ischaemia/reperfusion-induced acute kidney injury through inhibiting the mitochondrial damage pathway in mice. Clin Exp Pharmacol Physiol 2022; 49: 311-8.
DOI URL |
| 14. |
Yang YW, Yu XW, Zhang Y, et al. Hypoxia-inducible factor prolyl hydroxylase inhibitor roxadustat (FG-4592) protects against cisplatin-induced acute kidney injury. Clin Sci (Lond) 2018; 132: 825-38.
DOI PMID |
| 15. |
Yang B, Lan S, Dieudé M, et al. Caspase-3 is a pivotal regulator of microvascular rarefaction and renal fibrosis after ischemia-reperfusion injury. J Am Soc Nephrol 2018; 29: 1900-16.
DOI PMID |
| 16. |
Xu X, Kriegel AJ, Liu Y, et al. Delayed ischemic preconditioning contributes to renal protection by upregulation of miR-21. Kidney Int 2012; 82: 1167-75.
DOI PMID |
| 17. |
Brezis M, Rosen S. Hypoxia of the renal medulla--its implications for disease. N Engl J Med 1995; 332: 647-55.
DOI URL |
| 18. |
Nangaku M, Rosenberger R, Heyman SN, Eckardt K. Regulation of hypoxia-inducible factor in kidney disease. Clin Exp Pharmacol Physiol 2013; 40: 148-57.
DOI URL |
| 19. | Fine LG, Bandyopadhay D, Norman JT. Is there a common mechanism for the progression of different types of renal diseases other than proteinuria? Towards the unifying theme of chronic hypoxia. Kidney Int Suppl 2000; 75: S22-6. |
| 20. |
Maxwell P. HIF-1: an oxygen response system with special relevance to the kidney. J Am Soc Nephrol 2003; 14: 2712-22.
PMID |
| 21. |
Schödel J, Mole DR, Ratcliffe PJ. Pan-genomic binding of hypoxia-inducible transcription factors. Biol Chem 2013; 394: 507-17.
DOI PMID |
| 22. |
Dong F, Zhang X, Wu F, et al. The effects of Kangxianling on renal fibrosis as assessed with a customized gene chip. J Tradit Chin Med 2012; 32: 229-33.
DOI |
| 23. | Ji J, He L. Effect of Kangxianling decoction on expression of TGF-β1/Smads and extracellular matrix deposition. Evid Based Complement Alternat Med 2019; 2019: 5813549. |
| 24. |
Jiang Y, Zhu Y, Zhen T, et al. Transcriptomic analysis of the mechanisms of alleviating renal interstitial fibrosis using the Traditional Chinese Medicine Kangxianling in a rat model. Sci Rep 2020; 10: 10682.
DOI PMID |
| 25. | Liu YM, Zhang Y, He LQ. Experimental study on effect of Kangxianling on rat renal interstitial fibrosis. Zhong Guo Zhong Xi Yi Jie He Za Zhi 2007; 27: 901-4. |
| 26. | Wang Y, Ma Z, Zhong L, Yu K, He L. Effect of Kangxianling recipe on p38MAPK/NF-KBp65 mediated inflammatory factors in 5/6 nephrectomized mice. Evid Based Complement Alternat Med 2017; 37: 365-70. |
| 27. |
Zhang X, Wang Y, Velkov T, et al. T-2 toxin-induced toxicity in neuroblastoma-2a cells involves the generation of reactive oxygen, mitochondrial dysfunction and inhibition of Nrf2/HO-1 pathway. Food Chem Toxicol 2018; 114: 88-97.
DOI PMID |
| 28. |
Patel NS, Sharples E, Cuzzocrea S, et al. Pretreatment with EPO reduces the injury and dysfunction caused by ischemia/reperfusion in the mouse kidney in vivo. Kidney Int 2004; 66: 983-9.
DOI URL |
| 29. |
Haase VH. Hypoxia-inducible factor-prolyl hydroxylase inhibitors in the treatment of anemia of chronic kidney disease. Kidney Int Suppl 2021; 11: 8-25.
DOI PMID |
| 30. |
Kang DH, Park E, Yu E, Lee Y, Yoon Y. Renoprotective effect of erythropoietin (EPO): possibly via an amelioration of renal hypoxia with stimulation of angiogenesis in the kidney. Kidney Int 2005; 67: 1683.
DOI URL |
| 31. |
Pham K, Parikh K, Heinrich EC. Hypoxia and inflammation: insights from high-altitude physiology. Front Physiol 2021; 12: 676782.
DOI URL |
| 32. |
Taylor CT, Colgan SP. Regulation of immunity and inflammation by hypoxia in immunological niches. Nat Rev Immunol 2017; 17: 774-5.
DOI PMID |
| 33. |
Mcgettrick AF, O'neill LA. The role of HIF in immunity and inflammation. Cell Metab 2020; 32: 524-36.
DOI PMID |
| 34. |
Kalogeris T, Baines CP, Krenz M, Korthuis RJ. Ischemia/ reperfusion. Compr Physiol 2016; 7: 113-70.
DOI PMID |
| 35. | Van Uden P, Kenneth NS, Rocha S. Regulation of hypoxia-inducible factor-1alpha by NF-kappa B. Biochem J 2008; 412: 477-84. |
| 36. | Van Uden P, Kenneth NS, Webster R, Müller HA, Mudie S, Rocha S. Evolutionary conserved regulation of HIF-1β by NF-κB. PLoS Genet 2011; 7: e1001285. |
| 37. |
Wang X, Wei L, Li Q, Lai Y. HIF-1α protects osteoblasts from ROS-induced apoptosis. Free Radic Res 2022; 56: 143-53.
DOI URL |
| 38. |
Luo SY, Wang J, Liu C, et al. Hif-1α/Hsf1/Hsp70 signaling pathway regulates redox homeostasis and apoptosis in large yellow croaker (Larimichthys crocea) under environmental hypoxia. Zool Res 2021; 42: 746-60.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

