Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (1): 78-86.DOI: 10.19852/j.cnki.jtcm.20220727.001
• Original articles • Previous Articles Next Articles
Chinese herbal compound Jinsiwei (金思维) improves synaptic plasticity in mice with sporadic Alzheimer’s disease induced by streptozotocin
TIAN Meijing1, HE Yannan1, ZHENG Mingcui1, QIN Gaofeng3, GONG Zhuoyan1, HUANG Shuaiyang2( ), WANG Pengwen1(
), WANG Pengwen1( )
)
- 1 Key Laboratory of Chinese Internal Medicine of Ministry of Education and Beijing, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China
2 Department of Respiratory Medicine, the Third Affiliated Hospital of Beijing University of Chinese Medicine, Beijing 100029, China
3 Department of Traditional Chinese Medicine, Affiliated Hospital of Binzhou Medical College, Shandong 256603, China
-
Received:2021-12-12Accepted:2022-03-26Online:2023-02-15Published:2023-01-10 -
Contact:HUANG Shuaiyang,WANG Pengwen -
About author:Dr. HUANG Shuaiyang, Department of Respiratory Medicine, the Third Affiliated Hospital of Beijing University of Chinese Medicine, Beijing 100029, China. 1138507954@qq.com. Telephone:+86-10-84013195; +86-18813110509
Prof. WANG Pengwen, Key Laboratory of Chinese Internal Medicine of Ministry of Education and Beijing, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China. pw_wang@163.com;
-
Supported by:The Mechanism of Shenzhiling in Early Intervention on AD via Functional Brain Circuit PI3K/Akt-MTOR Pathway(81573927);The Scientific Research and Graduate Training Project of Beijing Municipal Commission of Education (2016), and Young Teachers Project of Beijing University of Chinese Medicine(2019-BUCMXJKY018)
Cite this article
TIAN Meijing, HE Yannan, ZHENG Mingcui, QIN Gaofeng, GONG Zhuoyan, HUANG Shuaiyang, WANG Pengwen. Chinese herbal compound Jinsiwei (金思维) improves synaptic plasticity in mice with sporadic Alzheimer’s disease induced by streptozotocin[J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 78-86.
share this article
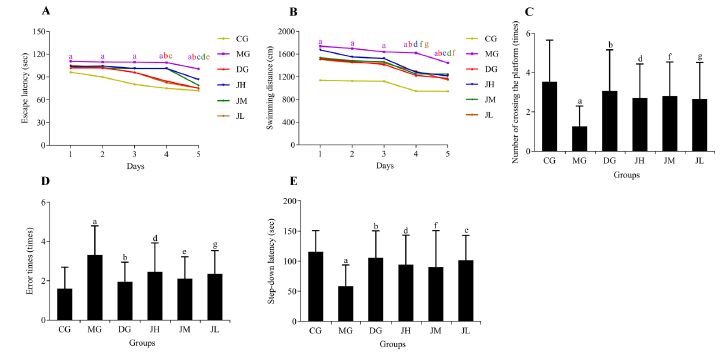
Figure 1 Efficacy of Jinsiwei on Morris Water Maze test and step-down passive avoidance test results in STZ-induced SD mice A: escape latency; B: swimming distance; C: number of crossing the platform; D: error times; E: step-down latency. CG: control group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); MG: model group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); DG: donepezil group (mice were administered with 0.92 mg/kg donepezil via oral gavage once a day for 3 months); JH: Jinsiwei high-dose group (mice were administered with 20 mg/kg Jinsiwei via oral gavage once a day for 3 months); JM: Jinsiwei medium-dose group (mice were administered with 10 mg/kg Jinsiwei via oral gavage once a day for 3 months); JL: Jinsiwei low-dose group (mice were administered with 5 mg/kg Jinsiwei via oral gavage once a day for 3 months). STZ: streptozotocin; SAD: sporadic Alzheimer’s disease. Results are shown as mean ± standard deviation (n = 15). aP < 0.01, model group vs control group; bP < 0.01, donepezil group vs model group; dP < 0.05, Jinsiwei high-dose group vs model group; eP < 0.01, fP < 0.05, Jinsiwei medium-dose group vs model group; cP < 0.01, gP < 0.05, Jinsiwei low-dose group vs model group.
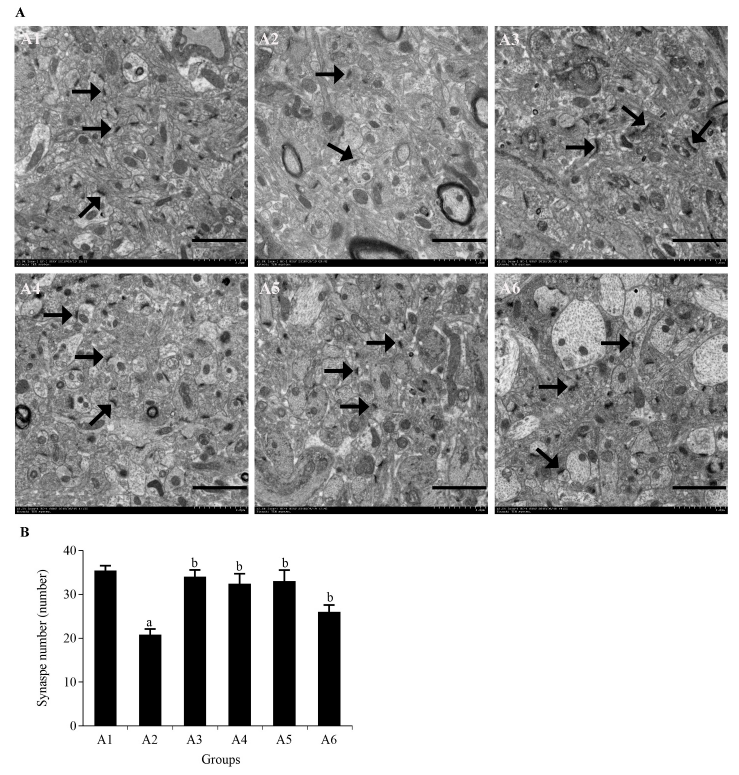
Figure 2 Efficacy of Jinsiwei on the number of synapses in STZ-induced SAD mice A: ultrastructure of synapses in the hippocampal CA1 of mice were observed under transmission electron microscopy at ×2.0 k; B: number of synapses in hippocampal CA1 regions; →: synapses; scale bar = 2 μm. A1: control group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); A2: model group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); A3: donepezil group (mice were administered with 0.92 mg/kg donepezil via oral gavage once a day for 3 months); A4: Jinsiwei high-dose group (mice were administered with 20 mg/kg Jinsiwei via oral gavage once a day for 3 months); A5: Jinsiwei medium-dose group (mice were administered with 10 mg/kg Jinsiwei via oral gavage once a day for 3 months); A6: Jinsiwei low-dose group (mice were administered with 5 mg/kg Jinsiwei via oral gavage once a day for 3 months); STZ: streptozotocin; SAD: sporadic Alzheimer’s disease. Results are shown as mean ± standard deviation (n = 3). aP < 0.01 vs control group, bP < 0.01, cP < 0.05 vs model group.

Figure 3 Efficacy of Jinsiwei on the distribution of positive cells and protein expression of drebrin and cofilin in the hippocampal CA1 region of STZ-induced SAD mice A, B: distribution and positive cells of drebrin in the hippocampal CA1 region of mice (immunohistochemical staining, ×20); C, D: distribution and positive cells of cofilin in the hippocampal CA1 region of mice (immunohistochemical staining, ×20); E: protein expression of drebrin; F: protein expression of cofilin; →: positive cells of cofilin; scale bar = 50 μm. A1, C1, CG: control group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); A2, C2, MG: model group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); A3, C3, DG: donepezil group (mice were administered with 0.92 mg/kg donepezil via oral gavage once a day for 3 months); A4, C4, JH: Jinsiwei high-dose group (mice were administered with 20 mg/kg Jinsiwei via oral gavage once a day for 3 months); A5, C5, JM: Jinsiwei medium-dose group (mice were administered with 10 mg/kg Jinsiwei via oral gavage once a day for 3 months); A6, C6, JL: Jinsiwei low-dose group (mice were administered with 5 mg/kg Jinsiwei via oral gavage once a day for 3 months); STZ: streptozotocin; SAD: sporadic Alzheimer’s disease. Results are shown as mean ± standard deviation (n = 6). aP < 0.01 vs control group; bP < 0.01, cP < 0.05 vs model group.

Figure 4 Efficacy of Jinsiwei on the distribution of positive cells and protein expression of syn and NR2B in the hippocampal CA1 region of STZ-induced SAD mice A, B: distribution and positive cells of syn in the hippocampal CA1 region of mice (immunohistochemical staining, ×20); C, D: distribution and positive cells of NR2B in the hippocampal CA1 region of mice (immunohistochemical staining, ×20); E: protein expression of syn; F: protein expression of NR2B; →: positive cells of syn; scale bar = 50 μm. A1, C1, CG: control group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); A2, C2, MG: model group (mice were administered with an equal volume of 0.5% carboxymethyl cellulose via oral gavage once a day for 3 months); A3, C3, DG: donepezil group (mice were administered with 0.92 mg/kg donepezil via oral gavage once a day for 3 months); A4, C4, JH: Jinsiwei high-dose group (mice were administered with 20 mg/kg Jinsiwei via oral gavage once a day for 3 months); A5, C5, JM: Jinsiwei medium-dose group (mice were administered with 10 mg/kg Jinsiwei via oral gavage once a day for 3 months); A6, C6, JL: Jinsiwei low-dose group (mice were administered with 5 mg/kg Jinsiwei via oral gavage once a day for 3 months). NR2B: N-methyl D-aspartate receptor subtype 2B; STZ: streptozotocin; SAD: sporadic Alzheimer’s disease. Results are shown as mean ± standard deviation (n = 6). aP < 0.01 vs control group; bP < 0.01, cP < 0.05 vs model group.
| 1 |
Lane CA, Hardy J, Schott JM. Alzheimer’s disease. Eur J Neurol 2018; 25: 59-70.
DOI PMID |
| 2 | Chen Y, Fu AKY, Ip NY. Synaptic dysfunction in Alzheimer’s disease: mechanisms and therapeutic strategies. Pharmacol Ther 2019; 195: 186-98. |
| 3 | Guo L, Tian J, Du H. Mitochondrial dysfunction and synaptic transmission failure in Alzheimer’s disease. J Alzheimers Dis 2017; 57: 1071-86. |
| 4 | Zablah BY, Merovitch N, Jia ZP. The role of ADF/Cofilin in synaptic physiology and Alzheimer’s disease. Front Cell Dev Biol 2020; 8: 594998. |
| 5 | Meng Y, Ding L, Zhang HY, et al. An Aβ3-10-KLH vaccine reduced Alzheimer’s disease-like pathology and had a sustained effect in Tg-APPswe/PSEN1dE9 mice. Brain Res 2017; 1673: 72-7. |
| 6 | Chang L, Zhang YL, Liu JP, et al. Differential regulation of N-Methyl-D-Aspartate receptor subunits is an early event in the actions of soluble amyloid-β1-40 oligomers on hippocampal neurons. J Alzheimers Dis 2016; 51: 197-212. |
| 7 |
Sasaki-Hamada S, Ikeda M, Oka JI. Glucagon-like peptide-2 rescues memory impairments and neuropathological changes in a mouse model of dementia induced by the intracerebroventricular administration of streptozotocin. Sci Rep 2019; 9: 13723.
DOI PMID |
| 8 | Salkovic-Petrisic M, Knezovic A, Hoyer S, et al. What have we learned from the streptozotocin-induced animal model of sporadic Alzheimer’s disease, about the therapeutic strategies in Alzh-eimer’s research. J Neural Transm (Vienna) 2013; 120: 233-52. |
| 9 | Kamat PK. Streptozotocin induced Alzheimer’s disease like changes and the underlying neural degeneration and regeneration mechanism. Neural Regen Res 2015; 10: 1050-2. |
| 10 | Shi J, Tian JZ, Zhang XK. A combination extract of ginseng, epimedium, polygala, and tuber curcumae increases synaptophysin expression in APPV717I transgenic mice. J Chin Med 2012; 7: 13. |
| 11 | Tian JZ, Shi J, Miao Y. A preliminary study of GEPT capsule in the treatment of amnestic mild cognitive impairment. J Alzheimers Dement 2009; 5: 225. |
| 12 | Liu ZH, Qin GF, Mana LL, et al. GAPT regulates cholinergic dysfunction and oxidative stress in the brains of learning and memory impairment mice induced by scopolamine. Brain Behav 2020; 10: e01602. |
| 13 | Grieb P. Intracerebroventricular streptozotocin injections as a model of Alzheimer’s disease: in search of a relevant mechanism. Mol Neurobiol 2016; 53: 1741-52. |
| 14 | Wang YH, Wang PW, Chen F, et al. Potential synaptic plasticity-based Shenzhiling oral liquid for a SAD mouse model. Brain Behav 2019; 9: e01385. |
| 15 | Qin GF, Wang YH, Liu ZH, et al. Shenzhiling oral solution promotes myelin repair through PI3K/Akt-mTOR pathway in stz-induced SAD mice. 3 Biotech 2021; 11: 361. |
| 16 | Mana LL, Feng HL, Dong YF, et al. Effect of Chinese herbal compound GAPT on the early brain glucose metabolism of APP/PS1 transgenic mice. Int J Immunopathol Pharmacol 2019; 33: 1-13. |
| 17 |
Hunter S, Arendt T, Brayne C. The senescence hypothesis of disease progression in alzheimer disease: an integrated matrix of disease pathways for FAD and SAD. Mol Neurobiol 2013; 48: 556-70.
DOI PMID |
| 18 | Li X, Liu Y, Zhang X, et al. Effect of ginsenoside Rg1 on expressions of phosphory protein tau and N-methyl-D-aspartate receptor subunits NR1 and NR2B in rat brain slice model of Alzheimer’s disease. Zhong Guo Zhong Yao Za Zhi 2010; 35: 3339-43. |
| 19 | Liu SJ, Yang C, Zhang Y, et al. Neuroprotective effect of β-asarone against Alzheimer’s disease: regulation of synaptic plasticity by increased expression of syp and gluR1. Drug Des Devel Ther 2016; 10: 1461-9. |
| 20 | Srivastava P, Dhuriya YK, Kumar V, et al. PI3K/Akt/GSK3β induced CREB activation ameliorates arsenic mediated alterations in NMDA receptors and associated signaling in rat hippocampus: neuroprotective role of curcumin. Neurotoxicology 2018; 67: 190-205. |
| 21 | Shi J, Zhang X, Ni J, et al. The influence of GAPT extraction on synapse loss of APPswe/PS1dE9 transgenic mice via adjusting bcl-2/bax balance. Alzheimers Dement (N Y) 2018; 4: 724-36. |
| 22 | Kozlov S, Afonin A, Evsyukov I, et al. Alzheimer’s disease: as it was in the beginning. Rev Neurosci 2017; 28: 825-43. |
| 23 |
Clark JK, Furgerson M, Crystal JD, et al. Alterations in synaptic plasticity coincide with deficits in spatial working memory in presymptomatic 3xTg-AD mice. Neurobiol Learn Mem 2015; 125: 152-62.
DOI PMID |
| 24 |
Mufson EJ, Binder L, Counts SE, et al. Mild cognitive impairment: pathology and mechanisms. Acta Neuropathol 2012; 123: 13-30.
DOI PMID |
| 25 | Liu Y, Xu YF, Zhang L, et al. Effective expression of Drebrin in hippocampus improves cognitive function and alleviates lesions of Alzheimer’s disease in APP (swe)/PS1 ((ΔE9) mice. CNS Neurosci Ther 2017; 23: 590-604. |
| 26 | Kang DE, Woo JA. Cofilin, a master node regulating cytoskeletal pathogenesis in Alzheimer’s disease. J Alzheimers Dis 2019; 72: S131-44. |
| 27 | Subramanian J, Savage JC, Tremblay ME. Synaptic loss in Alzheimer’s disease: mechanistic insights provided by two-photon in vivo imaging of transgenic mouse models. Front Cell Neurosci 2020; 14: 592607. |
| 28 | Clayton DA, Mesches MH, Alvarez E.A hippocampal NR2B deficit can mimic age-related changes in long-term potentiation and spatial learning in the Fischer 344 rat. J Neurosci 2002; 22: 3628-37. |
| [1] | ZHAO Ming, LUO Yimiao, WANG Huichan, CAO Yu, MA Lina, PEI Hui, LI Hao. Guilingji capsule (龟龄集胶囊) for Alzheimer's disease: secondary analysis of a randomized non-inferiority controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1019-1025. |
| [2] | ZHANG Xiaoying, WANG Ruixuan, WANG Yiqing, XU Fanxing, YAN Tingxu, WU Bo, ZHANG Ming, JIA Ying. Spinosin protects Neuro-2a/APP695 cells from oxidative stress damage by inactivating p38 [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 868-875. |
| [3] | QIAO Lina, SHI Yinan, TAN Lianhong, JIANG Yanshu, YANG Yongsheng. Efficacy of electroacupuncture stimulating Shenmen (HT7), Baihui (GV20), Sanyinjiao (SP6) on spatial learning and memory deficits in rats with insomnia induced by para-chlorophenylalanine: a single acupoint vs combined acupoints [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 704-714. |
| [4] | HUANG Hongye, WANG Bingqian, CHEN Shuijin, FANG Jiayu, WANG Xiaohua, CHEN Lechun, JIANG Yu, ZHANG Huanzhen, CHEN Jincheng, LIN Zhigang. Chinese Tuina remodels the synaptic structure in neuropathic pain rats by downregulating the expression of N-methyl D-aspartate receptor subtype 2B and postsynaptic density protein-95 in the spinal cord dorsal horn [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 715-724. |
| [5] | XIN Zhixiong, LI Jiyuan, XU Yanni, LING Jing, JIANG Min, YU Yutian. Acupuncture on intrauterine growth restriction associated brain injury: case study including use of magnetic resonance imaging [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 602-605. |
| [6] | QIN Xiude, LIU Yu, WU Yanqing, WANG Shuo, WU Haibin, ZHU Jinqiang. Anti-apoptotic efficacy of Qingnao Yizhi formula(清脑益智方) in hypoxia/reoxygenation primary cortical neurons through the promotion of synaptic plasticity by modulating the NogoA-Nogo receptor/Rho-Rho kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2021, 41(1): 59-67. |
| [7] | Wang Xu, Li Zhaolong, Li Chunri, Wang Yue, Yu Song, Ren Lu. Electroacupuncture with Bushen Jiannao improves cognitive deficits in senescence-accelerated mouse prone 8 mice by inhibiting neuroinflammation [J]. Journal of Traditional Chinese Medicine, 2020, 40(5): 812-819. |
| [8] | Ren Beida, Cheng Fafeng, Wang Xueqian, Wan Yuxiang, Ji Wenting, Du Xin, Zhang Shuang, Liu Shuling, Ma Chongyang, Xiong Yiliang, Hao Gaoting, Wang Qingguo. Possible mechanisms underlying treatment of Alzheimer's disease with Traditional Chinese Medicine: active components, potential targets and synthetic pathways of Bulao Elixir [J]. Journal of Traditional Chinese Medicine, 2020, 40(3): 484-496. |
| [9] | Rangachari Balamurugan, Jeong Hwa Kim, Mi-na Jo, Chenglian Xue, Jin kyu Park, Jae Kwon Lee. Bee wax coated water-soluble fraction of bee venom improved altered glucose homeostasis in streptozotocin-induced diabetic rats [J]. Journal of Traditional Chinese Medicine, 2019, 39(06): 842-852. |
| [10] | Chi Huiying, Liu Te, Pan Weidong, Chen Jiulin, Wu Beiling, Yu Zhihua, Chen Chuan. Shen-Zhi-Ling oral solution improves learning and memory ability in Alzheimer's disease mouse model [J]. Journal of Traditional Chinese Medicine, 2019, 39(05): 667-677. |
| [11] | Liu Lijuan, Yu Chen, Liu Junbao. Effect of Cuzhi liquid on learning and memory dysfunction in a mouse model of Alzheimer's disease [J]. Journal of Traditional Chinese Medicine, 2018, 38(06): 890-895. |
| [12] | Yu Chaochao, Wang Li, Kong Lihong, Shen Feng, Ma Chaoyang, Du Yanjun, Zhou Hua. Acupoint combinations used for treatment of Alzheimer's disease: A data mining analysis [J]. Journal of Traditional Chinese Medicine, 2018, 38(06): 943-952. |
| [13] | Wang Jing, Cheng Kai, Qin Zhuo, Wang Yanping, Zhai Lijing, You Min, Wu Juanjiao. Effects of electroacupuncture at Guanyuan(CV 4) or Sanyinjiao (SP 6) on hypothalamus-pituitary-ovary axis and spatial learning and memory in female SAMP8 mice [J]. Journal of Traditional Chinese Medicine, 2017, 37(01): 96-100. |
| [14] | Hu Haiyan, Wang Yiyu, Zhang Yihui, Wang Wenhua, Xu Dongmei, Chen Zhiyu, Zhang Xiaoyan, Mao Dandan. Effect of Qingxinkaiqiao compound on cortical mRNA expression of the apoptosis-related genes Bcl-2, BAX, caspase-3, and Aβ in an Alzheimer's disease rat model [J]. Journal of Traditional Chinese Medicine, 2016, 36(05): 654-662. |
| [15] | Bhesh Raj Sharma, Min Suk Kim, Dong Young Rhyu. Nelumbo Nucifera leaf extract attenuated pancreatic β-cells toxicity induced by interleukin-1β and interferon-γ, and increased insulin secrection of pancreatic β-cells in streptozotocin-induced diabetic rats [J]. Journal of Traditional Chinese Medicine, 2016, 36(01): 71-77. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

