Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (5): 915-924.DOI: 10.19852/j.cnki.jtcm.20230802.006
Previous Articles Next Articles
Inhibitory effect of berberine on morphine tolerance and hyperalgesia in mice
HAN Shuai1,2, MAO Hua3, JIN Tingting4, YAN Rubing4, WANG Ziyi4, ZHANG Jie5, SHI Jianwen6( ), LIANG Yongxin6(
), LIANG Yongxin6( )
)
- 1 Department of Anesthesiology, Women’s and Children’s Hospital Affiliated to Qingdao University, Qingdao 266034, China
2 Yangzhou University, Yangzhou 225009, China
3 Department of Anesthesiology, Women’s and Children’s Hospital Affiliated to Qingdao University, Qingdao 266034, China
4 Qingdao University, Qingdao 266071, China
5 Department of Anesthesiology, the Affiliated Hospital of Qingdao University, Qingdao 266075, China
6 Department of Anesthesiology, Women’s and Children’s Hospital Affiliated to Qingdao University, Qingdao 266034, China
-
Received:2022-06-11Accepted:2022-10-12Online:2023-10-15Published:2023-08-02 -
Contact:Prof. LIANG Yongxin, Department of Anesthesiology, Women’s and Children’s Hospital Affiliated to Qingdao University, Qingdao 266034, China.liangzi66@hotmail.com ; Prof. SHI Jianwen, Department of Anesthesiology, Women’s and Children’s Hospital Affiliated to Qingdao University, Qingdao 266034, China.javen0523@163.com . Telephone: +86-10-15618135070 -
Supported by:Natural Science Foundation of Shandong Province: Mechanism of the Compound Huanglian Decoction Against Morphine Tolerance Based on Network Pharmacology and Microglia M1/M2 Pattern(ZR2023MH270)
Cite this article
HAN Shuai, MAO Hua, JIN Tingting, YAN Rubing, WANG Ziyi, ZHANG Jie, SHI Jianwen, LIANG Yongxin. Inhibitory effect of berberine on morphine tolerance and hyperalgesia in mice[J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 915-924.
share this article

Figure 1 Analgesic effect of BBR in the hot-plate test A: analgesic effect of BBR detected by the hot-plate test. B: effect of BBR on morphine analgesia detected by the hot-plate test. Animals were treated with intraperitoneal BBR 30 min prior to the subcutaneous administration of 5 mg/kg Mor. BBR: berberine, Mor: morphine, % MPAE: the percentage of maximal possible analgesic effect. All values are expressed as the mean ± standard deviation; n = 8 for each group; one-way analysis of variance followed by Dunnett's multiple comparison. aP < 0.01 compared with the control group, bP < 0.01 compared with the morphine group.
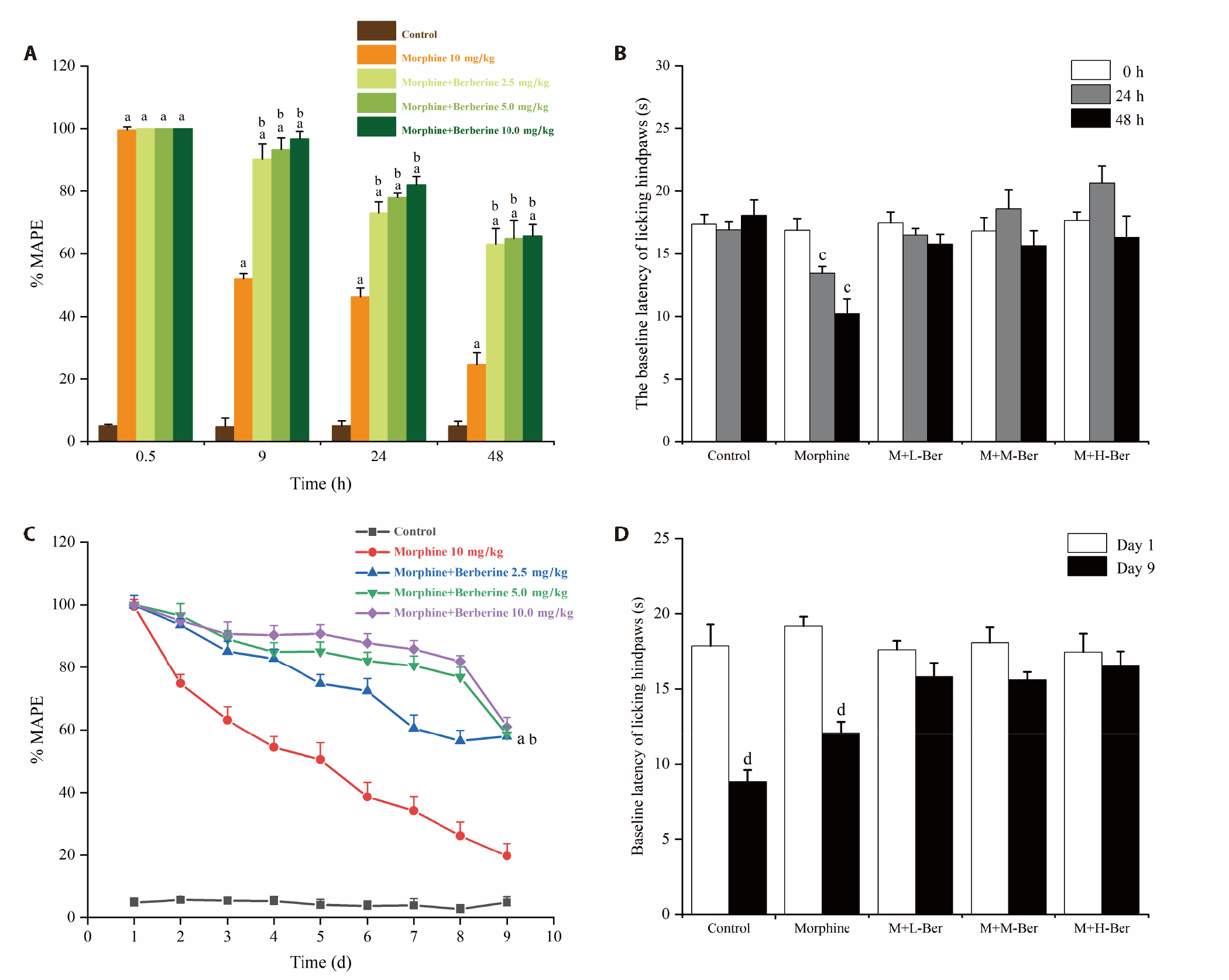
Figure 2 Effect of BBR on morphine-induced acute/ chronic tolerance and hyperalgesia A: the inhibitory effect of BBR on acute morphine-induced tolerance. B: the inhibitory effect of BBR on acute morphine-induced hyperalgesia. C: the inhibitory effect of BBR on morphine-induced chronic tolerance. D: the inhibitory effect of BBR on chronic morphine-induced hyperalgesia. M: morphine 10 mg/kg, L-Ber: berberine 2.5 mg/kg, M-Ber: berberine 5.0 mg/kg, H-Ber: berberine 10 mg/kg, MPAE: maximum possible analgesic effect, BBR: berberine. Data are presented as mean ± standard deviation; n = 8 for each group; one-way analysis of variance followed by Dunnett's multiple comparison (A, B, and C); (D)Two-tailed unpaired student’s t test. aP < 0.01 compared with the control group, bP < 0.01 compared with the morphine group, cP < 0.01 compared with each corresponding group at the 0 h time point, dP < 0.01 compared with each corresponding group on day 1.
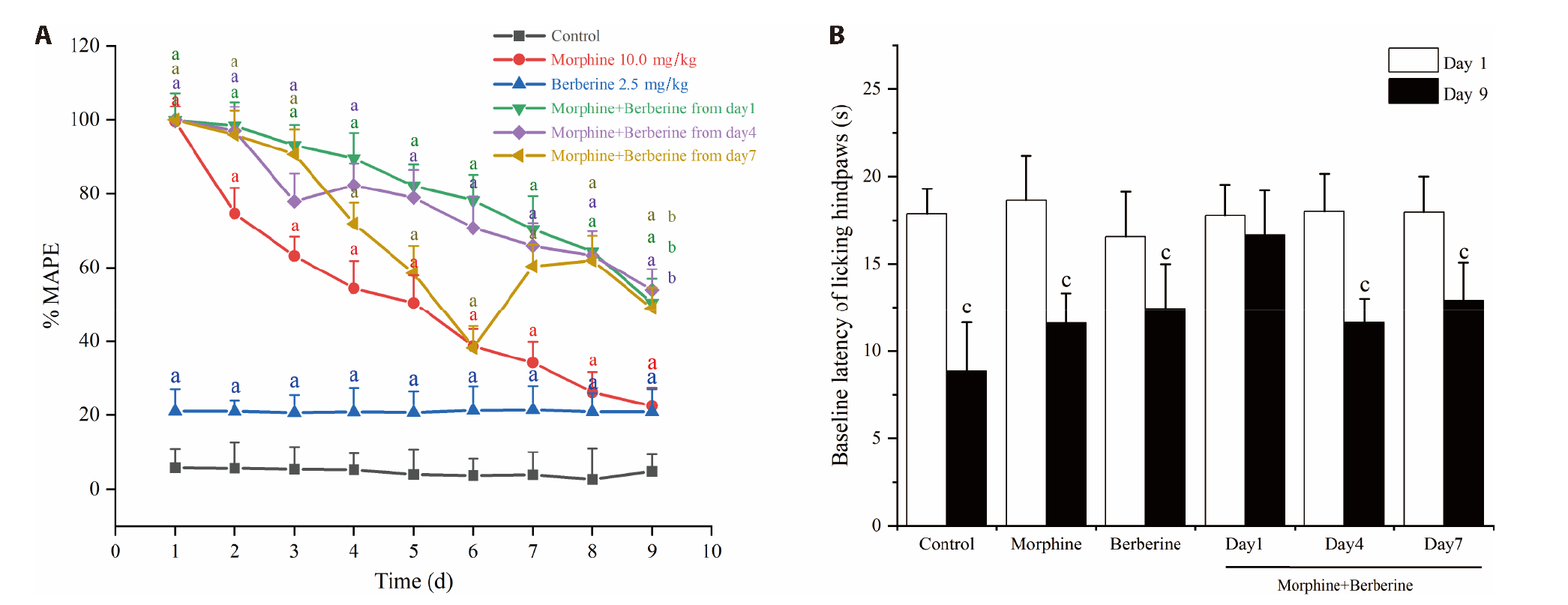
Figure 3 Effect of BBR (2.5 mg/kg) on morphine-induced established tolerance and hyperalgesia A: the inhibitory effect of BBR on morphine-induced established tolerance. B: the inhibitory effect of BBR on established morphine-induced hyperalgesia. Mice were given morphine for 8 days; BBR was co-administered on days 1, 4, and 7. BBR: berberine. All values are expressed as the mean ± standard deviation; n = 8 for each group. For Figure A, aP < 0.01 compared with the control group, bP < 0.01 compared with the morphine group, one-way analysis of variance followed by Dunnett's multiple comparison; for Figure B, cP < 0.01 compared with each corresponding group on day 1, two-tailed unpaired student’s t test.
| Group | n | NOS activity (kU/g protein) | NO content (μmol/g protein) | |
|---|---|---|---|---|
| Normal control | 8 | 6.98±0.27 | 1.22±0.13 | |
| BBR | 8 | 6.78±0.38a | 1.24±0.21a | |
| Mor | 8 | 7.96±0.43b | 1.87±0.17a | |
| Mor+BBR | Day 1 | 8 | 7.18±0.32a | 1.39±0.14a |
| Day 4 | 8 | 7.67±0.31 | 1.47±0.23a,b | |
| Day 7 | 8 | 7.81±0.26b | 1.68±0.14b,c |
Table 1 Effect of Berberine on NOS activity and NO content in the spinal cord of morphine-induced established tolerance mice
| Group | n | NOS activity (kU/g protein) | NO content (μmol/g protein) | |
|---|---|---|---|---|
| Normal control | 8 | 6.98±0.27 | 1.22±0.13 | |
| BBR | 8 | 6.78±0.38a | 1.24±0.21a | |
| Mor | 8 | 7.96±0.43b | 1.87±0.17a | |
| Mor+BBR | Day 1 | 8 | 7.18±0.32a | 1.39±0.14a |
| Day 4 | 8 | 7.67±0.31 | 1.47±0.23a,b | |
| Day 7 | 8 | 7.81±0.26b | 1.68±0.14b,c |

Figure 4 Molecular docking of berberine with the human GluN1-GluN2A, GluN1-GluN2B NMDAR A: crystal structure of the human GluN1-GluN2A NMDAR. B: the binding pocket for BBR in the central vestibule between the GluN1-GluN2A channel gate and selectivity filter. C: BBR binds to the human GluN1-GluN2A NMDAR, two-dimensional pattern diagram. D: crystal structure of the human GluN1-GluN2B NMDAR. E: the binding pocket for BBR in the central vestibule between the GluN1-GluN2B channel gate and selectivity filter. F: BBR binds to the human GluN1-GluN2B NMDAR, two-dimensional pattern diagram. NMDAR: N-methyl-D-aspartate receptor; BBR: berberine.
| 1. |
Ruiz-Quiñonez AK, Espinosa-Riquer ZP, Carranza-Aguilar CJ, et al. Co-administration of morphine and levamisole increases death risk, produces neutropenia and modifies antinociception in mice. Addict Biol 2022; 27: e13166.
DOI URL |
| 2. |
Svensson CK. Opioid-induced hyperalgesia: is it a clinically relevant phenomenon? Int J Pharm Pract 2022; 30: 479-83.
DOI URL |
| 3. | Zhang B, Sun L. Research progress on the mechanism of morphine tolerance. Beijing Da Xue Xue Bao 2020; 42: 239-41. |
| 4. | Christrup LL. Morphine metabolites. Acta Anaesthesiol Scand 1997; 41: 116-22. |
| 5. | Yang LP. Interrelation and intracellular mechanism of heat hyperalgesia and morphine tolerance. Hebei Yi Ke Da Xue Xue Bao 2003; 24: 38-41. |
| 6. | Chou WB. Role of NMDA receptor in pain sensitivity and opioid tolerance and dependence. Anesth Analg 2002:105-8. |
| 7. | Liang HC, Xu YH, Wen Q, et al. Inhibition of Guiyuan tablets on morphine-induced tolerance and hyperalgesia in mice. Zhong Guo Yao Li Xue He Du Li Xue Za Zhi 2014; 28: 813-22. |
| 8. | Zhu SM. The development of research for the mechanisms of morphine-induced enhanced pain sensitivity. Int J Anesth Resus 2006; 27: 320-2. |
| 9. | Chen Y, Wang GL. The role of glutamate energy system in the alteration of spinal plasticity in opioid tolerance. Tianjin Yi Ke Da Xue Xue Bao 2009; 15: 255-61. |
| 10. |
Deng M, Chen SR, Pan HL. Presynaptic NMDA receptors control nociceptive transmission at the spinal cord level in neuropathic pain. Cell Mol Life Sci 2019; 76: 1889-99.
DOI PMID |
| 11. |
Liu Y, Luo Z, Liao Z, et al. Effects of excessive activation of N-methyl-D-aspartic acid receptors in neonatal cardiac mitochondrial dysfunction induced by intrauterine hypoxia. Front Cardiovasc Med 2022; 9: 837142.
DOI URL |
| 12. |
Anstötz M, Lee SK, Maccaferri G. Glutamate released by Cajal-Retzius cells impacts specific hippocampal circuits and behaviors. Cell Rep 2022; 39: 110822.
DOI URL |
| 13. |
Frouni I, Huot P. Glutamate modulation for the treatment of levodopa induced dyskinesia: a brief review of the drugs tested in the clinic. Neurodegener Dis Manag 2022; 12: 203-14.
DOI URL |
| 14. |
Zhang Y, Chu JM, Wong GT. Cerebral glutamate regulation and receptor changes in perioperative neuroinflammation and cognitive dysfunction. Biomolecules 2022; 12: 597.
DOI URL |
| 15. |
Grande EM, Raka F, Hoffman S, Adeli K. GLP-2 regulation of dietary fat absorption and intestinal chylomicron production via neuronal nitric oxide synthase (nNOS) signaling. Diabetes 2022; 71: 1388-99.
DOI URL |
| 16. |
Mao J, Price DD, Mayer DJ. Mechanisms of hyperalgesia and morphine tolerance: a current view of their possible interactions. Pain 1995; 62: 259-74.
DOI PMID |
| 17. |
Garzón J, Rodríguez-Muñoz M, Vicente-Sánchez A, Bailón C, Martínez-Murillo R, Sánchez-Blázquez P. RGSZ2 binds to the neural nitric oxide synthase PDZ domain to regulate mu-opioid receptor-mediated potentiation of the N-methyl-D-aspartate receptor calmodulin dependent protein kinase Ⅱ pathway. Antioxid Redox Signal 2011; 15: 873-87.
DOI URL |
| 18. | Yan XF, Zheng JF, Li WD. Studies on the chemical synthesis of natural drugs berberine. You Ji Hua Xue 2021; 41: 2217-27. |
| 19. |
Yang SM, Yu ZJ, Sun WP, et al. The antiviral alkaloid berberine ameliorates neuropathic pain in rats with peripheral nerve injury. Acta Neurol Belg 2020; 120: 557-64.
DOI PMID |
| 20. | Zhou GJ, Yan MZ, Guo G, et al. Ameliorative effect of berberine on neonatally induced type 2 diabetic neuropathy via modulation of BDNF, IGF-1, PPAR-γ, and AMPK expressions. Dose Response 2019; 17: 1559325819862449. |
| 21. |
Chen CQ, Tao CH, Liu ZC, et al. A randomized clinical trial of berberine hydrochloride in patients with diarrhea-predominant irritable bowel syndrome. Phytother Res 2015; 29: 1822-7.
DOI PMID |
| 22. |
Han AM, Heo H, Kwon YK. Berberine promotes axonal regeneration in injured nerves of the peripheral nervous system. J Med Food 2012; 15: 413-7.
DOI PMID |
| 23. |
Peng WH, Lo KL, Lee YH, et al. Berberine produces antidepressant-like effects in the forced swim test and in the tail suspension test in mice. Life Sci 2007; 81: 933-8.
DOI URL |
| 24. |
Hu Y, Ehli EA, Hudziak JJ, et al. Berberine and evodiamine influence serotonin transporter (5-HTT) expression via the 5-HTT-linked polymorphic region. Pharmacogenomics J 2012; 12: 372-8.
DOI PMID |
| 25. |
Chen CQ, Lu ML, Pan QH, et al. Berberine improves intestinal motility and visceral pain in the mouse models mimicking diarrhea-predominant irritable bowel syndrome (IBS-D) symptoms in an opioid-receptor dependent manner. PLoS One 2017; 10: e0145556.
DOI URL |
| 26. |
Kim HJ. Berberine ameliorates allodynia induced by chronic constriction injury of the sciatic nerve in rats. J Med Food 2015; 18: 909-15.
DOI PMID |
| 27. |
Habtemariam S. Berberine pharmacology and the gut microbiota: a hidden therapeutic link. Pharmacol Res 2020; 155: 104722.
DOI URL |
| 28. | Tao Z, Zhao HP, Wang RL, et al. The role of berberine on TLR4 expression and microglial activation. Sheng Ming Ke Xue Yi Qi 2019; 17: 66-70. |
| 29. |
Suski M, Bujak-Gizycka B, Madej J, et al. Co-administration of dextromethorphan and morphine: reduction of post-operative pain and lack of influence on morphine metabolism. Basic Clin Pharmacol Toxicol 2010; 107: 680-4.
DOI URL |
| 30. |
Säwe J. High-dose morphine and methadone in cancer patients. Clinical pharmacokinetic considerations of oral treatment. Clin Pharmacokinet 1986; 11: 87-106.
PMID |
| 31. |
Staahl C, Upton R, Foster DJ, et al. Pharmacokinetic-pharmacodynamic modeling of morphine and oxycodone concentrations and analgesic effect in a multimodal experimental pain model. J Clin Pharmacol 2008; 48: 619-61.
DOI PMID |
| 32. |
Zelcer N, van de Wetering K, Hillebrand M, et al. Mice lacking multidrug resistance protein 3 show altered morphine pharmacokinetics and morphine-6-glucuronide antinociception. Proc Natl Acad Sci USA 2005; 102: 7274-9.
PMID |
| 33. |
Lim SY, Cengiz P. Opioid tolerance and opioid-induced hyperalgesia: is TrkB modulation a potential pharmacological solution? Neuropharmacology 2022; 220: 109260.
DOI URL |
| 34. | Yan XB, Huang XL, Huang D. NMDA receptors and NOS are involved in the formation of morphine tolerance in bone cancer pain mice. Zhong Nan Da Xue Xue Bao 2010; 35: 458-63. |
| 35. | Li WX, Deng XM, Yan XQ, et al. Inhibition of morphine tolerance and dependence by LY274614, a competitive NMDA receptor antagonist. Zhong Hua Ma Zui Xue Za Zhi 2001; 2: 28-31. |
| 36. | Ma XQ, Xu T, Jiang W. Recent advances in the relationship between NMDA receptor and mGluR5 in pain research. Zhong Hua Teng Tong Yi Xue Za Zhi 2014; 20: 181-3. |
| 37. | Chen Y, Wang GL. The role of glutamate energy system in the alteration of spinal plasticity in opioid tolerance. Tianjin Yi Ke Da Xue Xue Bao 2001; 15: 155-61. |
| 38. | Fu L, Guo RX, Mo LQ, et al. Role of c-Jun of spinal cord in NMDA receptor subunit NR2B mediated morphine antinociceptive tolerance in rats. Zhong Guo Yao Li Xue Tong Bao 2011; 27: 248-52. |
| 39. | Li JP, Guo RX, Liao XX, et al. Role of the spinal NMDA receptor NR2B subunit in inhibition of morphine tolerance by agmatine. Zhong Guo Lin Chuang Jie Pou Xue Za Zhi 2009; 27: 452-4. |
| 40. |
Bouhassira D. Neuropathic pain: Definition, assessment and epidemiology. Rev Neurol 2019; 175: 16-25.
DOI URL |
| 41. |
Qiu S, Li XY, Zhuo M. Post-translational modification of NMDA receptor GluN2B subunit and its roles in chronic pain and memory. Semin Cell Dev Biol 2011; 22: 521-9.
DOI PMID |
| 42. | Yu XR, Huang YG, Luo AL. Prevention of nociceptive behaviours in chronic constrictive injury rats by ifenprodil pretreatment. Zhong Hua Teng Tong Yi Xue Za Zhi 2007; 13: 342-4. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

