Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (4): 652-659.DOI: 10.19852/j.cnki.jtcm.2024.04.003
• Original articles • Previous Articles Next Articles
Ganoderma Lucidum Spore Oil enhances the effect of cyclophosphamide via inhibiting programmed death-1 and prolongs the survival of H22 tumor-bearing mice
JIANG Zhaojian1, CAI Hongfei1, YUAN Cheng1, CAO Lin1, XU Wendong1, HAN Yaming1, ZHANG Qin1, LI Jing1, WANG Qin2( ), LIU Juyan3,4(
), LIU Juyan3,4( )
)
- 1 Guangzhou HanFang Pharmaceutical Company Limited, National Engineering Research Center of Pharmaceutical Processing Technology of Traditional Chinese Medicine and Drug Innovation, Guangdong Provincial Key Laboratory of Medicinal Lipids, Guangzhou 510240, China
2 Zhongshan Medical College, Sun Yat-sen University, Guangzhou 510080, China
3 Guangzhou Pharmaceutical Holdings Limited, Guangzhou 510130, China
4 National Engineering Research Center of Pharmaceutical Processing Technology of Traditional Chinese Medicine and Drug Innovation, Guangzhou 510240, China
-
Received:2023-02-16Accepted:2023-07-20Online:2024-08-15Published:2024-07-15 -
Contact:WANG Qin, Zhongshan Medical College, Sun Yat-sen University, Guangzhou 510080, China.wangqin6@mail.sysu.edu.cn ; LIU Juyan, Guangzhou Pharmaceutical Holdings Limited, Guangzhou 510130, China.LJYan65@163.com Telephone: + 86-13710583307 -
Supported by:Ministry of Science and Technology of China: Evaluation, Re-examination, Promotion and Demonstration of Complete Sets of Technical Equipment for Efficient and Energy-Saving Extraction and Separation of Traditional Chinese Medicine(2017YFC1703104);Key Laboratory Project of Pharmaceutical Lipids in Guangdong Province : Assessment of Enterprise-Class Key Laboratory of Guangdong Province(2020B1212070024);Guangdong Province Key Areas Research and Development Program Project : Research and Development on Innovative Key Technologies and Intelligent Production Equipment about Processing, Decoction, Extraction and Separation of Traditional Chinese Medicine(2020B1111120002)
Cite this article
JIANG Zhaojian, CAI Hongfei, YUAN Cheng, CAO Lin, XU Wendong, HAN Yaming, ZHANG Qin, LI Jing, WANG Qin, LIU Juyan. Ganoderma Lucidum Spore Oil enhances the effect of cyclophosphamide via inhibiting programmed death-1 and prolongs the survival of H22 tumor-bearing mice[J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 652-659.
share this article
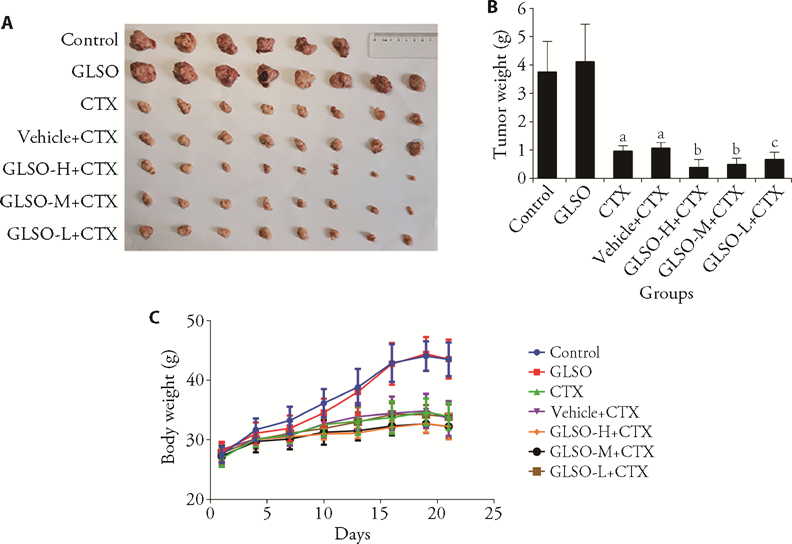
Figure 1 Effect of GLSO and CTX on tumor growth and body weight of H22 tumor-bearing mice in different groups A: size of tumors in each group; B: weight of tumors in each group; C: body weight of mice in each group. Control group: mice were given normal saline; GLSO group: mice were given 4.68 g/kg GLSO and normal saline; CTX group: mice were given normal saline and 50 mg/kg CTX; Vehicle + CTX: mice were given vehicle and 50 mg/kg CTX; GLSO-H + CTX: mice were given 4.68 g/kg GLSO and 50 mg/kg CTX; GLSO-M + CTX: mice were given 2.496 g/kg GLSO and 50 mg/kg CTX; GLSO-L + CTX: mice were given 0.312 g/kg GLSO and 50 mg/kg CTX. GLSO: Ganoderma Lucidum Spore Oil; CTX: cyclophosphamide. Data are presented as the mean ± standard deviation using one-way analysis of variance (n = 6-8). aP < 0.001 vs Control group; bP < 0.001, cP < 0.01 vs Vehicle + CTX group.
| Group | n | Average survival time (days) | Increased life span (%) | Median survival time (days) |
|---|---|---|---|---|
| Control | 24 | 32.5±2.7a | - | 28.0±3.1a |
| GLSO | 22 | 35.9±3.4 | 10.23 | 33.0±2.4 |
| Vehicle+CTX | 25 | 41.1±2.8 | 26.37 | 45.0±4.0 |
| GLSO-H+CTX | 24 | 44.6±3.9 | 37.15 | 49.0±12.3 |
| GLSO-M+CTX | 25 | 54.0±3.8b | 65.83 | 62.0±5.8b |
| GLSO-L+CTX | 25 | 54.7±3.9a | 68.04 | 62.0±10.8a |
Table 1 Survival of mice in each group ($\bar{x}±s$)
| Group | n | Average survival time (days) | Increased life span (%) | Median survival time (days) |
|---|---|---|---|---|
| Control | 24 | 32.5±2.7a | - | 28.0±3.1a |
| GLSO | 22 | 35.9±3.4 | 10.23 | 33.0±2.4 |
| Vehicle+CTX | 25 | 41.1±2.8 | 26.37 | 45.0±4.0 |
| GLSO-H+CTX | 24 | 44.6±3.9 | 37.15 | 49.0±12.3 |
| GLSO-M+CTX | 25 | 54.0±3.8b | 65.83 | 62.0±5.8b |
| GLSO-L+CTX | 25 | 54.7±3.9a | 68.04 | 62.0±10.8a |
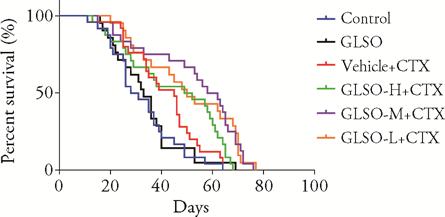
Figure 2 Effect of GLSO on the survival of H22 tumor-bearing mice treated with CTX in different groups Control group: mice were given normal saline; GLSO group: mice were given 4.68 g/kg GLSO and normal saline; Vehicle + CTX: mice were given vehicle and 50 mg/kg CTX; GLSO-H + CTX: mice were given 4.68 g/kg GLSO and 50 mg/kg CTX; GLSO-M + CTX: mice were given 2.496 g/kg GLSO and 50 mg/kg CTX; GLSO-L + CTX: mice were given 0.312 g/kg GLSO and 50 mg/kg CTX. GLSO: Ganoderma Lucidum Spore Oil; CTX: cyclophos-phamide. Kaplan-Meier survival analysis was employed to compare the difference of survival rates between groups (n = 22-25).
| Group | n | Thymus index | Spleen index | Liver index |
|---|---|---|---|---|
| Control | 6 | 0.226±0.045 | 4.711±0.638 | 42.531±10.011 |
| GLSO | 8 | 0.248±0.074 | 4.656±0.387 | 49.983±6.393 |
| CTX | 8 | 0.160±0.030a | 5.484±1.766 | 55.010±4.888a |
| Vehicle+CTX | 8 | 0.167±0.018a | 5.096±1.048 | 53.687±5.359d |
| GLSO-H+CTX | 8 | 0.232±0.040b | 5.292±0.784 | 52.048±6.397 |
| GLSO-M+CTX | 8 | 0.204±0.026b | 5.511±0.960 | 53.738±4.913 |
| GLSO-L+CTX | 8 | 0.246±0.046c | 4.763±0.399 | 51.727±6.517 |
Table 2 Thymus, spleen and liver indices in each group (mg/g, $\bar{x}±s$)
| Group | n | Thymus index | Spleen index | Liver index |
|---|---|---|---|---|
| Control | 6 | 0.226±0.045 | 4.711±0.638 | 42.531±10.011 |
| GLSO | 8 | 0.248±0.074 | 4.656±0.387 | 49.983±6.393 |
| CTX | 8 | 0.160±0.030a | 5.484±1.766 | 55.010±4.888a |
| Vehicle+CTX | 8 | 0.167±0.018a | 5.096±1.048 | 53.687±5.359d |
| GLSO-H+CTX | 8 | 0.232±0.040b | 5.292±0.784 | 52.048±6.397 |
| GLSO-M+CTX | 8 | 0.204±0.026b | 5.511±0.960 | 53.738±4.913 |
| GLSO-L+CTX | 8 | 0.246±0.046c | 4.763±0.399 | 51.727±6.517 |
| Group | n | UREA (mmol/L) | CRE (μmol/L) | ALT (U/L) | AST (U/L) |
|---|---|---|---|---|---|
| Control | 7 | 6.8±1.0 | 16.0±1.4 | 194.9±18.7 | 1514.1±171.1 |
| GLSO | 7 | 5.3±1.3a | 14.0±2.2 | 195.0±79.1 | 1462.4±452.5 |
| CTX | 7 | 6.8±1.2 | 15.3±2.3 | 74.7±10.2b | 383.1±70.9b |
| Vehicle+CTX | 7 | 6.3±0.8 | 15.6±1.7 | 72.1±9.1b | 348.7±73.5b |
| GLSO-H+CTX | 7 | 5.4±0.5c | 15.3±2.8 | 73.9±12.7 | 383.0±58.1 |
| GLSO-M+CTX | 7 | 5.2±0.8c | 14.4±1.7 | 72.4±4.3 | 408.4±103.5 |
| GLSO-L+CTX | 7 | 5.8±1.4 | 14.1±2.3 | 69.0±12.7 | 366.9±60.1 |
Table 3 Plasma level of UREA, CRE, ALT, AST in each group ($\bar{x}±s$)
| Group | n | UREA (mmol/L) | CRE (μmol/L) | ALT (U/L) | AST (U/L) |
|---|---|---|---|---|---|
| Control | 7 | 6.8±1.0 | 16.0±1.4 | 194.9±18.7 | 1514.1±171.1 |
| GLSO | 7 | 5.3±1.3a | 14.0±2.2 | 195.0±79.1 | 1462.4±452.5 |
| CTX | 7 | 6.8±1.2 | 15.3±2.3 | 74.7±10.2b | 383.1±70.9b |
| Vehicle+CTX | 7 | 6.3±0.8 | 15.6±1.7 | 72.1±9.1b | 348.7±73.5b |
| GLSO-H+CTX | 7 | 5.4±0.5c | 15.3±2.8 | 73.9±12.7 | 383.0±58.1 |
| GLSO-M+CTX | 7 | 5.2±0.8c | 14.4±1.7 | 72.4±4.3 | 408.4±103.5 |
| GLSO-L+CTX | 7 | 5.8±1.4 | 14.1±2.3 | 69.0±12.7 | 366.9±60.1 |
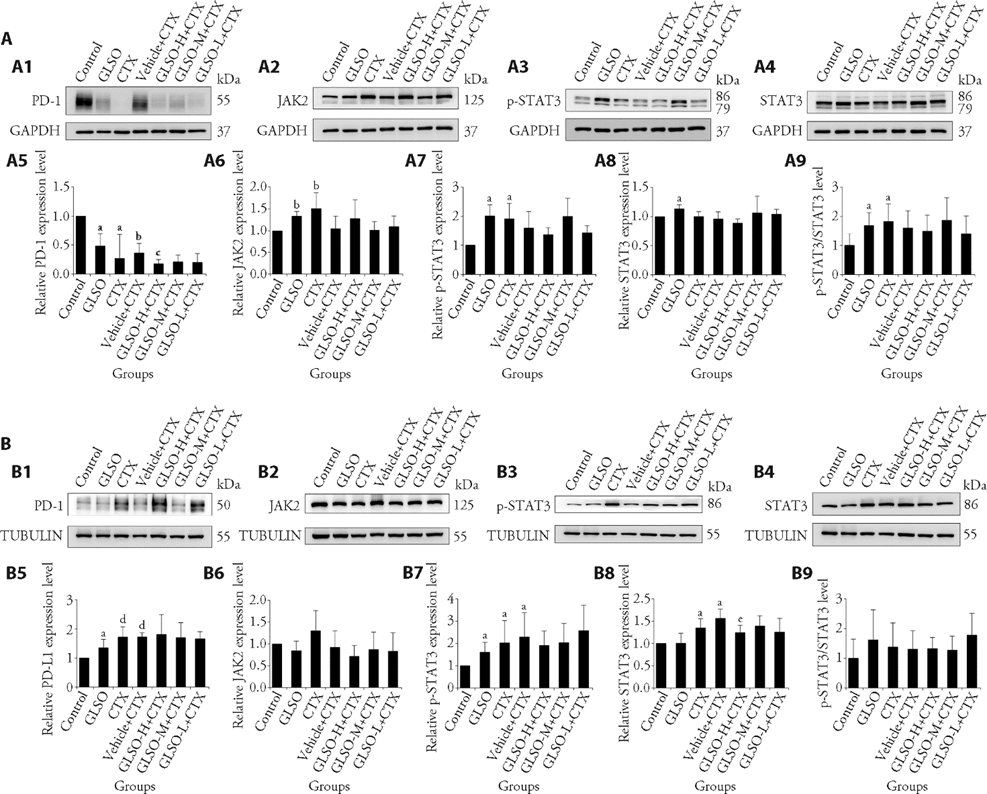
Figure 3 Effect of GLSO and CTX on the protein expression level of PD-1, JAK2, p-STAT3 and STAT3 in spleen and PD-L1, JAK2, p-STAT3 and STAT3 in tumor A: PD-1, JAK2, p-STAT3, STAT3, and pSTAT3/STAT3 level in spleen; A1: PD-1 expression was detected by Western blot analysis; A2: JAK2 expression was detected by Western blot analysis; A3: p-STAT3 expression was detected by Western blot analysis; A4: STAT3 expression was detected by Western blot analysis; A5: Western blot analysis of PD-1 expression; A6: Western blot analysis of JAK2 expression; A7: Western blot analysis of p-STAT3 expression; A8: Western blot analysis of STAT3 expression; A9: Western blot analysis of p-STAT3/STAT3 level; B: PD-L1, JAK2, p-STAT3, STAT3, and pSTAT3/STAT3 level in tumor; B1: PD-L1 expression was detected by Western blot analysis; B2: JAK2 expression was detected by Western blot analysis; B3: p-STAT3 expression was detected by Western blot analysis; B4: STAT3 expression was detected by Western blot analysis; B5: Western blot analysis of PD-L1 expression; B6: Western blot analysis of JAK2 expression; B7: Western blot analysis of p-STAT3 expression; B8: Western blot analysis of STAT3 expression; B9: Western blot analysis of p-STAT3/STAT3 level. Control group: mice were given normal saline; GLSO group: mice were given 4.68 g/kg GLSO and normal saline; CTX group: mice were given normal saline and 50 mg/kg CTX; Vehicle + CTX: mice were given vehicle and 50 mg/kg CTX; GLSO-H + CTX: mice were given 4.68 g/kg GLSO and 50 mg/kg CTX; GLSO-M + CTX: mice were given 2.496 g/kg GLSO and 50 mg/kg CTX; GLSO-L + CTX: mice were given 0.312 g/kg GLSO and 50 mg/kg CTX. GLSO: Ganoderma Lucidum Spore Oil; CTX: cyclophosphamide; PD-1: programmed death-1; PD-L1: programmed death ligand 1; JAK2: janus kinase 2; p-STAT3: phosphorylated signal transducer and activator of transcription 3; STAT3: signal transducer and activator of transcription 3. Data are presented as the mean ± standard deviation using one-way analysis of variance (n = 5). aP < 0.05, bP < 0.01, dP < 0.001, vs Control group, cP < 0.05, eP < 0.001, vs Vehicle + CTX group.
| 1. | Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021; 71: 209-49. |
| 2. | Li H, Gu L, Zhong Y, et al. Administration of polysaccharide from Panax notoginseng prolonged the survival of H22 tumor-bearing mice. Onco Targets Ther 2016; 9: 3433-41. |
| 3. |
Ikeda M, Morizane C, Ueno M, Okusaka T, Ishii H, Furuse J. Chemotherapy for hepatocellular carcinoma: current status and future perspectives. Jpn J Clin Oncol 2018; 48: 103-14.
DOI PMID |
| 4. |
Emadi A, Jones RJ, Brodsky RA. Cyclophosphamide and cancer: golden anniversary. Nat Rev Clin Oncol 2009; 6: 638-47.
DOI PMID |
| 5. | Ahlmann M, Hempel G. The effect of cyclophosphamide on the immune system: implications for clinical cancer therapy. Cancer Chemother Pharmacol 2016; 78: 661-71. |
| 6. |
Ahmad MF. Ganoderma lucidum: persuasive biologically active constituents and their health endorsement. Biomed Pharmacother 2018; 107: 507-19.
DOI PMID |
| 7. |
Chan SW, Tomlinson B, Chan P, Lam CWK. The beneficial effects of Ganoderma lucidum on cardiovascular and metabolic disease risk. Pharm Biol 2021; 59: 1161-71.
DOI PMID |
| 8. | Cui XY, Cui SY, Zhang J, et al. Extract of Ganoderma lucidum prolongs sleep time in rats. J Ethnopharmacol 2012; 139: 796-800. |
| 9. | Ma HT, Hsieh JF, Chen ST. Anti-diabetic effects of Ganoderma lucidum. Phytochemistry 2015; 114: 109-13. |
| 10. | Cuong VT, Chen W, Shi J, et al. The anti-oxidation and anti-aging effects of Ganoderma lucidum in Caenorhabditis elegans. Exp Gerontol 2018; 117: 99-105. |
| 11. |
Li J, Gu F, Cai C, et al. Purification, structural characterization, and immunomodulatory activity of the polysaccharides from Ganoderma lucidum. Int J Biol Macromol 2020; 143: 806-13.
DOI PMID |
| 12. | Jin X, Ruiz Beguerie J, Sze DM, Chan GC. Ganoderma lucidum (Reishi mushroom) for cancer treatment. Cochrane Database Syst Rev 2016; 4: CD007731. |
| 13. |
Zhou D, Zhou F, Ma J, Ge F. Microcapsulation of Ganoderma Lucidum spores oil: evaluation of its fatty acids composition and enhancement of oxidative stability. Ind Crop Prod 2019; 131: 1-7.
DOI |
| 14. | Zhang Y, Cai H, Tao Z, et al. Ganoderma lucidum spore oil (GLSO), a novel antioxidant, extends the average life span in Drosophila melanogaster. Food Sci Hum Well 2021; 10: 38-44. |
| 15. | Alsaab HO, Sau S, Alzhrani R, et al. PD-1 and PD-L 1 checkpoint signaling inhibition for cancer immunotherapy: mechanism, combinations, and clinical outcome. Front Pharmacol 2017; 8: 561. |
| 16. |
Yi M, Zheng X, Niu M, Zhu S, Ge H, Wu K. Combination strategies with PD-1/PD-L1 blockade: current advances and future directions. Mol Cancer 2022; 21: 28.
DOI PMID |
| 17. | National Research Council. Guide for the care and use of laboratory animals:eighth edition. Washington DC: the National Academies Press, 2011: 11-151. |
| 18. | Cor D, Knez Z, Knez Hrncic M. Antitumour, antimicrobial, antioxidant and antiacetylcholinesterase effect of Ganoderma Lucidum terpenoids and polysaccharides: a review. Molecules 2018; 23: 649. |
| 19. |
Brodsky RA, Jones RJ. Aplastic anaemia. Lancet 2005; 365: 1647-56.
DOI PMID |
| 20. | Krishnan C, Kaplin AI, Brodsky RA, et al. Reduction of disease activity and disability with high-dose cyclophosphamide in patients with aggressive multiple sclerosis. Arch Neurol 2008; 65: 1044-51. |
| 21. |
Cheung MC, Agarwal K. Liver abnormalities in the immunosuppressed. Best Pract Res Clin Gastroenterol 2013; 27: 597-618.
DOI PMID |
| 22. |
Wang G, Wang L, Zhou J, Xu X. The possible role of PD-1 protein in ganoderma lucidum-mediated immunomodulation and cancer treatment. Integr Cancer Ther 2019; 18: 1534735419880275.
DOI |
| 23. |
Austin JW, Lu P, Majumder P, Ahmed R, Boss JM. STAT3, STAT4, NFATc1, and CTCF regulate PD-1 through multiple novel regulatory regions in murine T cells. J Immunol 2014; 192: 4876-86.
DOI PMID |
| 24. | Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008; 359: 378-90. |
| 25. | Sun Y, Li Q, Xu JM, et al. A multicenter, single arm phase Ⅱ trial of a small molecule immune-modulator icaritin: safety, overall survival, immune dynamics, and PD-L 1 expression in advanced hepatocellular carcinoma. J Clin Oncol 2018; 36: 4077. |
| [1] | WU Jieya, HOU Li, ZHANG Xiaoyuan, Elizabeth Gullen, GAO Chong, WANG Jing. Efficacy of Yisui granule (益髓颗粒) on myelodysplastic syndromes in SKM-1 mouse xenograft model through suppressing Wnt/β-catenin signaling pathway [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 78-87. |
| [2] | FAN Yifu, ZHAO Hetong, ZHANG Yani, Yani Zifei, DU Juan, LING Changquan. Effectiveness of Jiedu granule (解毒颗粒) on gut microbiota in patients with advanced hepatocellular carcinoma: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 988-996. |
| [3] | XU Qian, QIN Wei, WU Fangzhen, LIN Yao, HONG Liting, CHEN Dan, HU Xuefeng, CAI Jing. Effect of Roucongrong(Herba Cistanches Deserticolae) decoction on the substantia nigra through Wnt/β-catenin signaling pathway in rats with Parkinson's disease induced by 6-hydroxydopamine hydrochloride [J]. Journal of Traditional Chinese Medicine, 2021, 41(5): 762-770. |
| [4] | ZHOU Xingqin, CHANG Yazhou, JI Bin, ZHAO Hongyu, SHEN Chaoyan, CHANG Renan. Efficacy of gecko polysaccharide on suppressed immune response induced by cyclophosphamide in mice [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 539-545. |
| [5] | GU Xiaoli, CHEN Menglei, LIU Minghui, ZHANG Zhe, ZHAO Weiwei, CHENG Wenwu. Value of Traditional Chinese Medicine syndrome differentiation in predicting the survival time of patients with advanced cancer [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 636-641. |
| [6] | Wang Lei, Li Xiu, Zhou Rongyao, Shan Yawei. Effect of Bushen Jianpi formula on survival of patients with moderate and advanced hepatocellular carcinoma: a retrospective study [J]. Journal of Traditional Chinese Medicine, 2020, 40(4): 683-689. |
| [7] | Li Yifan, Li Juan, Fan Bifa, Wang Yitong, Jiang Juling, Zhang Zhenhua, Wang Xinxing, Lu Wenping. Efficacy and safety of Yiqi Huoxue Jiedu decoction for the treatment of advanced epithelial ovarian cancer patients: a double-blind randomized controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2020, 40(1): 103-111. |
| [8] | Guan Huiting, Xie Su, Liu Shangyi, Xie Qing, Hou Shengkai, Liu Huarong, Zhang Yunjie, Hu Yaqing, Zhang Chenyu. Effects of Jiazhu decoction in combination with cyclophosphamide on breast cancer in mice [J]. Journal of Traditional Chinese Medicine, 2019, 39(05): 642-648. |
| [9] | Li Kai, Liu Rui, Xu Guangzhong, Zhang Weihua, Liu Chen, Zhu Bin, Hua Baojin, Feng Xingzhong. Effect of a modified Banxia Xiexin decoction plus chemotherapy on stage Ⅲ colon cancer [J]. Journal of Traditional Chinese Medicine, 2019, 39(02): 251-257. |
| [10] | Li Qiang, Liu Zhibin, Yang Jiping, Guo Huijun, Xu Liran. Comment on the intervention of Traditional Chinese Medicine on survival rates of patients living with human immunodeficiency virus and acquired immune deficiency syndrome [J]. Journal of Traditional Chinese Medicine, 2016, 36(03): 409-410. |
| [11] | Liu Zhibin, Meng Changhai, Yang Jiping, Chen Xiumin, Jin Yantao. Survival rate:an indicator of the management of acquired immune deficiency syndrome using Traditional Chinese Medicine [J]. Journal of Traditional Chinese Medicine, 2015, 35(04): 473-477. |
| [12] | Yantao Jin, Zhibin Liu, Xiumin Chen, Xin Wang, Dan Wang, Ziqiang Jiang, Ying Liu, Jian Wang, Wen Zou, Huijun Guo, Liran Xu. Survival of people living with HIV after treatment with Traditional Chinese Medicine in Henan province of China: a retrospective cohortstudy [J]. Journal of Traditional Chinese Medicine, 2014, 34(04): 430-436. |
| [13] | Zhengrong Huang, Yong Wang, Jiajun Chen, Rongping Wang, Qiang Chen. Effect of Xiaoaiping injection on advanced hepatocellular carcinoma in patients [J]. Journal of Traditional Chinese Medicine, 2013, 33(01): 34-38. |
| [14] | Gao, Liang; Chen, Zelin; Tian, Lixin; Li, Zhongzheng; Guo, Yi. Effects of bloodletting puncture at Jing-Well points in distal ends of finger and toe on survival rate and brain edema in cerebral ischemic rats [J]. Journal of Traditional Chinese Medicine, 2012, 32(03): 471-476. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

