Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (1): 78-87.DOI: 10.19852/j.cnki.jtcm.20231204.003
• Original articles • Previous Articles Next Articles
Efficacy of Yisui granule (益髓颗粒) on myelodysplastic syndromes in SKM-1 mouse xenograft model through suppressing Wnt/β-catenin signaling pathway
WU Jieya1, HOU Li1, ZHANG Xiaoyuan1, Elizabeth Gullen2, GAO Chong3( ), WANG Jing1(
), WANG Jing1( )
)
- 1 Department of Oncology and Hematology, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China
2 Department of Pharmacology, Yale Medical School, New Haven, CT 06510, USA
3 Department of Oncology, Beijing Hospital of Traditional Chinese Medicine, Capital Medical University, Beijing 100010, China
-
Received:2022-10-17Accepted:2023-02-21Online:2024-02-15Published:2023-12-04 -
Contact:Dr. GAO Chong, Department of Oncology, Beijing Hospital of Traditional Chinese Medicine, Capital Medical University, Beijing 100010, China. gaochong9356@126.com;Dr. WANG Jing, Department of Oncology and Hematology, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing 100700, China. jwang2936@126.com. Telephone: +86-10-84013145 -
Supported by:Clinical Translational Research of Beijing Municipal Science and Technology Commission, Administrative Commission of Zhongguancun Science Park-funded Project: Study on Mechanisms and Efficacy of Yisui granule Treating Low and Intermediate Risk of Myelodysplastic Syndromes via DNA Demethylation(Z211100002921018);National Natural Science Foundation of China-funded Projects: Study on Molecular Mechanisms of Yisui granule Treating Myelodysplastic Syndromes via Regulating DNA Methylation(81503575);Mechanism Study of Tea Polyphenols activating cGAS-STING Pathway to Inhibit Lung Adenocarcinoma Immune Escape based on Redox Balance(82172760);Golden Bridge Project of Beijing Association for Science and Technology-funded Project: Study on Mechanisms of Yisui granule Treating Low and Intermediate Risk of Myelodysplastic Syndromes via DNA Demethylation(ZZ20059)
Cite this article
WU Jieya, HOU Li, ZHANG Xiaoyuan, Elizabeth Gullen, GAO Chong, WANG Jing. Efficacy of Yisui granule (益髓颗粒) on myelodysplastic syndromes in SKM-1 mouse xenograft model through suppressing Wnt/β-catenin signaling pathway[J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 78-87.
share this article
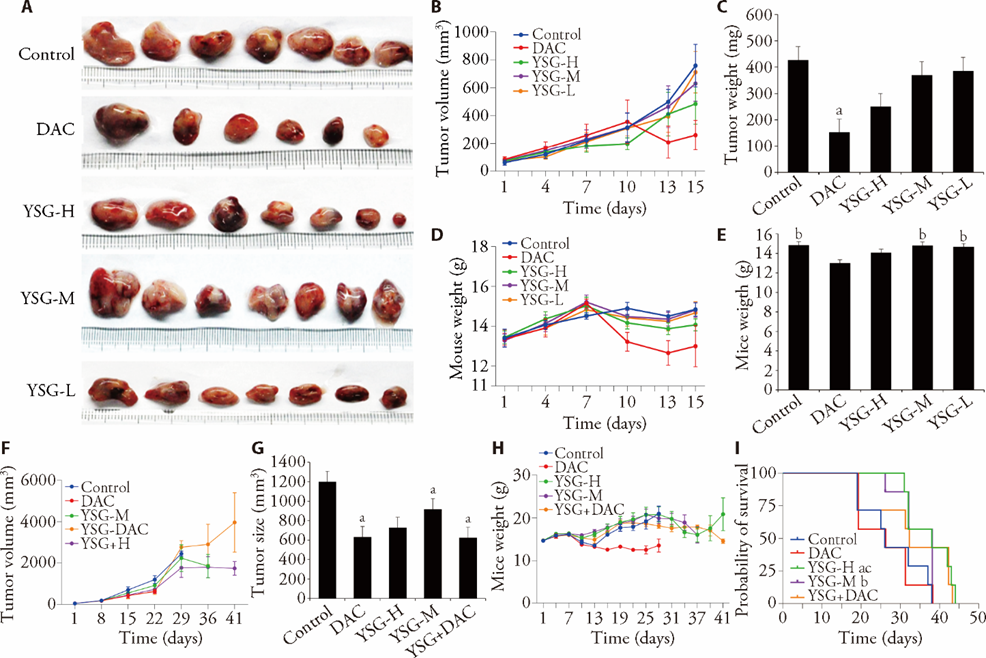
Figure 1 YSG reduced decitabine-induced adverse effects and prolonged OS A: tumor tissues of each group from d15. Except for DAC, n = 6, for other groups, n = 7. B: tumor size of each group during d1 to d15. Two-way ANOVA was applied. Except for DAC, n = 6, for other groups, n = 7. C: tumor weight of each group from d15. Except for DAC, n = 6, for other groups, n = 7. D: effect of high-dosage, medium-dosage, low-dosage YSG, and decitabine on body weight of animals. Except for DAC, n = 6, for other groups, n = 7. E: animal body weight of each group from d15. Except for DAC, n = 6, for other groups, n = 7. F: effect of high-dosage YSG, medium-dosage YSG, and/or decitabine on tumor size in animals from d1 to d41. For all groups, n = 7. G: comparison of tumor size between each group in d22. For Control and YSG+DAC, n = 5. For DAC, n = 4. For YSG-H and YSG-M, n = 7. H: effect of high-dosage YSG, medium-dosage YSG, and/or decitabine on body weight of animals from d1 to d41. For all groups, n = 7. I: the Kaplan-Meier survival graph represents the effect of high-dosage YSG, medium-dosage YSG, and/or decitabine on OS. For all groups, n = 7. Control: control group (0.9% NaCl oral administration, 0.2 mL/10 g, 14 d); DAC: decitabine group (decitabine intraperitoneal injection, 0.5 mg/kg per time per day, 5 d); YSG-H: high-dosage YSG group (YSG solution oral administration, 69 g/kg per day, 14 d); YSG-M: medium-dosage YSG group (YSG solution oral administration, 34.5 g/kg per day, 14 d); YSG-L: low dosage YSG group (YSG solution oral administration, 17.25 g/kg per day, 14 d); YSG+DAC: medium-dosage YSG and decitabine combination group (YSG solution oral administration, 34.5g/kg per day, d1-d14 + decitabine, 0.5 mg/kg per day, intraperitoneal injection, d1-d5). YSG: Yisui granule; OS: overall survival; ANOVA: analysis of variance. One-way ANOVA, two-way ANOVA, and Kaplan-Meier survival analysis were applied. The data measured in this research were expressed as mean ± standard deviation. Compared with the Control, aP < 0.05; compared with the DAC, bP < 0.05, cP < 0.01.

Figure 2 YSG negatively regulated DNMT1 in a dose-dependent manner, demethylated the sFRP5 gene, and up-regulated the expression of the sFRP5 protein A: effect of high-dosage, medium-dosage, low-dosage YSG and decitabine on DNMT1 protein expression determined by western blotting, n = 3. B: effect of high-dosage, medium-dosage, low-dosage YSG and decitabine on DNMT1 mRNA expression determined by real-time PCR, n = 3. C: Western blot analysis of the effect of high-dosage, medium-dosage low-dosage YSG and decitabine on DNMT1 and sFRP5 with β-actin as loading control among different groups. Cropped blots are used in this figure, and they have been run under the same experimental conditions. n = 3. D: effect of high-dosage, medium-dosage, low-dosage YSG, and decitabine on sFRP5 pair 1 gene determined by bisulfite amplicon sequencing, n = 3. E: effect of high-dosage, medium-dosage, low-dosage YSG, and decitabine on the level of methylation of the sFRP5 pair 1 gene methylation level, n = 3. F: effect of high-dosage, medium-dosage, low-dosage YSG, and decitabine on sFRP5 protein expression determined by western blotting. n = 3. G: effect of high-dosage, medium-dosage, low-dosage YSG, and decitabine on sFRP5 mRNA expression determined by real-time PCR. n = 3. Control: control group (0.9% NaCl oral administration, 0.2 mL/10 g, 14 d); DAC: decitabine group (decitabine intraperitoneal injection, 0.5 mg/kg per time per day, 5 d); YSG-H: high-dosage YSG group (YSG solution oral administration, 69 g/kg per day, 14 d); YSG-M: medium-dosage YSG group (YSG solution oral administration, 34.5 g/kg per day, 14 d); YSG-L: low dosage YSG group (YSG solution oral administration, 17.25 g/kg per day, 14 d). YSG: Yisui granule; DNMT1: DNA methyltransferase 1; sFRP5: secreted frizzled related protein 5; PCR: polymerase chain reaction; ANOVA: analysis of variance. One-way ANOVA and two-way ANOVA were performed. The data measured in this research were expressed as mean ± standard deviation. Compared with the Control, aP < 0.001, bP < 0.01, and cP < 0.05; compared with the DAC, dP < 0.05.
| sFRP5 Gene Methylation | DAC | YSG-H | YSG-M | YSG-L |
|---|---|---|---|---|
| Pair 1 | ↓ | ↓↓ | ↓↓ | - |
| Pair 2 | ↓↓ | - | ↓ | - |
| Total | ↓↓ | - | ↓ | - |
Table 1 Comparison between high-dosage, medium-dosage, low-dosage YSG and decitabine and Control about sFRP5 gene in pair1, pair2 and total
| sFRP5 Gene Methylation | DAC | YSG-H | YSG-M | YSG-L |
|---|---|---|---|---|
| Pair 1 | ↓ | ↓↓ | ↓↓ | - |
| Pair 2 | ↓↓ | - | ↓ | - |
| Total | ↓↓ | - | ↓ | - |
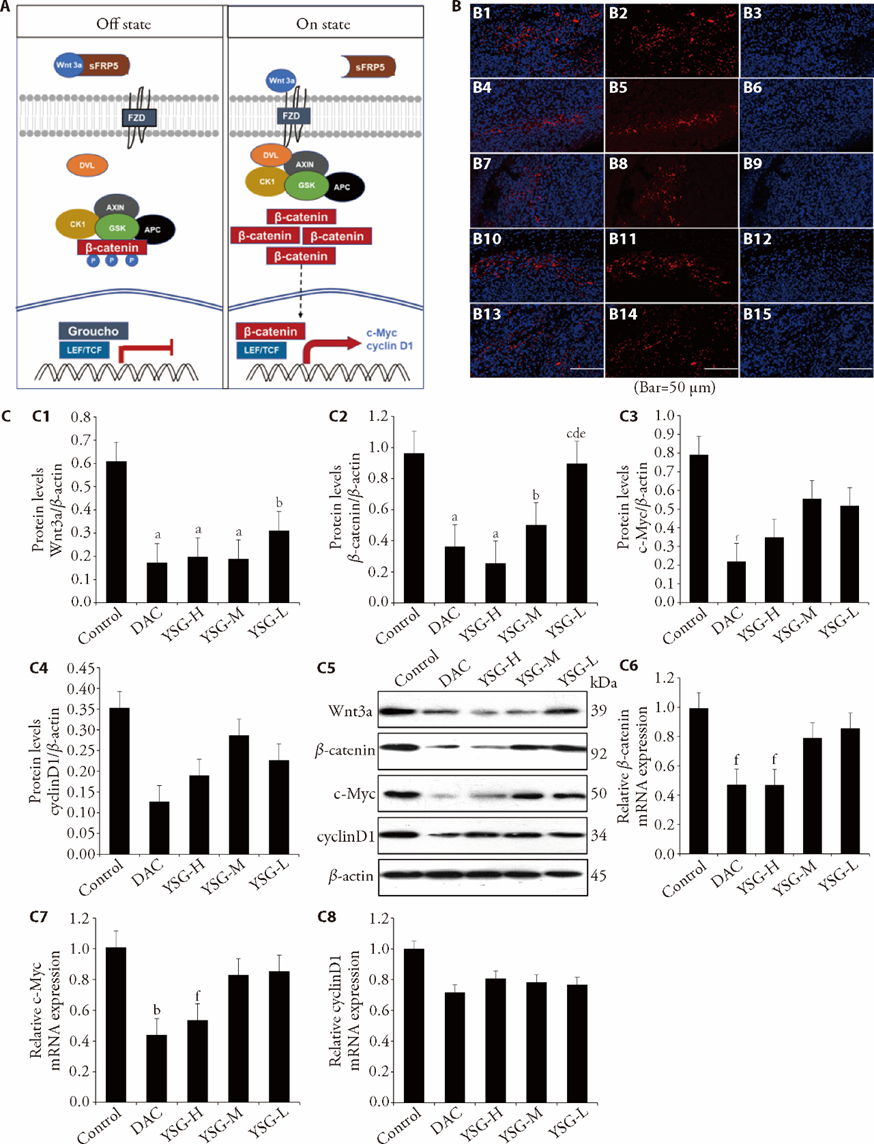
Figure 3 YSG negatively regulated protein and mRNA expressions in the Wnt/β-catenin signaling pathway A: Wnt/β-catenin signaling mechanism. In the "Off state" β-catenin forms a macromolecular complex with AXIN, APC, CK1, and GSK (destruction complex). Phosphorylation by CK1 and GSK prepares β-catenin to be ubiquitinated and degraded. In "On state", the interaction of the Wnt3a ligand with receptor sFRP5 and DVL is recruited to the plasma membrane. the nucleus. Then, β-catenin is no longer phosphorylated and ubiquitinated because the destruction complex disassembles. The β-catenin accumulated in the cytoplasm is translocated to the nucleus. It displaces the co-repressor Groucho and interacts with TCF to activate gene expressions, including c-Myc and cyclinD1. B: down-regulation effect of high-dosage YSG, medium-dosage YSG and/or decitabine on the immunofluorescence signal of β-catenin. The right lanes are stained with DAPI to capture the nuclear orientation (blue). The middle lanes are probed with anti-β-catenin antibodies to examine the localization of β-catenin (red). The left lanes represent a merge of DAPI and anti-β-catenin staining. B1: merge of Control; B2: β-catenin of Control; B3: DAPI of Control; B4: merge of DAC; B5: β-catenin of DAC; B6: DAPI of DAC; B7: merge of YSG-H; B8: β-catenin of YSG-H; B9: DAPI of YSG-H; B10: merge of YSG-M; B11: β-catenin of YSG-M; B12: DAPI of YSG-M; B13: merge of YSG-L; B14: β-catenin of YSG-L; B15: DAPI of YSG-L. C: effect of high-dosage, medium-dosage, low-dosage YSG, and decitabine on protein and mRNA expressions of Wnt3a, β-catenin, c-Myc, and cyclyinD1. C1: western blot result of Wnt3a protein; C2: western blot result of β-catenin protein; C3: Western blot result of c-Myc protein; C4: western blot result of cyclinD1 protein; C5: western blot analysis of Wnt3a, β-catenin, c-Myc, cyclinD1 and β-actin proteins; C6: mRNA of β-catenin; C7: mRNA of c-Myc; C8: mRNA of cyclyinD1. Control: control group (0.9% NaCl oral administration, 0.2 mL/10 g, 14 d); DAC: decitabine group (decitabine intraperitoneal injection, 0.5 mg/kg per time per day, 5 d); YSG-H: high-dosage YSG group (YSG solution oral administration, 69 g/kg per day, 14 d); YSG-M: medium-dosage YSG group (YSG solution oral administration, 34.5 g/kg per day, 14 d); YSG-L: low dosage YSG group (YSG solution oral administration, 17.25 g/kg per day, 14 d). YSG: Yisui granule; APC: adenomatous polyposis coli; CK1: casein kinase 1; sFRP5: secreted frizzled related protein 5; DVL: dishevelled; TCF: T-cell factor; DAPI; 4',6-diamidino-2-phenylindole; PCR: polymerase chain reaction; ANOVA: analysis of variance. Using Western blotting and real-time PCR, collected data were analyzed by one-way ANOVA, n = 3. The data measured in this research were expressed as mean ± standard deviation. Compared with the Control, aP < 0.001, bP < 0.01, and fP < 0.05; compared with the DAC, cP < 0.01; compared with the YSG-H, dP < 0.001; compared with the YSG-M, eP < 0.01.
| 1. |
Cabezón M, Malinverni R, Bargay J, et al. Different methylation signatures at diagnosis in patients with high-risk myelodysplastic syndromes and secondary acute myeloid leukemia predict azacitidine response and longer survival. Clin Epigenetics 2021; 13: 1-14.
DOI |
| 2. |
Jones PA, Issa J-PJ, Baylin S. Targeting the cancer epigenome for therapy. Nat Rev Genet 2016; 17: 630-41.
DOI PMID |
| 3. |
Mohammad HP, Barbash O, Creasy CL. Targeting epigenetic modifications in cancer therapy: erasing the roadmap to cancer. Nat Med 2019; 25: 403-18.
DOI PMID |
| 4. |
Zhou JD, Zhang TJ, Xu ZJ, et al. Genome-wide methylation sequencing identifies progression-related epigenetic drivers in myelodysplastic syndromes. Cell Death Dis 2020; 11: 1-15.
DOI |
| 5. |
Reilly B, Tanaka TN, Diep D, et al. DNA methylation identifies genetically and prognostically distinct subtypes of myelodysplastic syndromes. Blood Adv 2019; 3: 2845-58.
DOI PMID |
| 6. |
Jiang Y, Liu L, Wang J, Cao Z, Zhao Z. The Wilms' tumor gene-1 is a prognostic factor in myelodysplastic syndrome: a Meta analysis. Oncotarget 2018; 9: 16205-12.
DOI PMID |
| 7. |
Yang K, Wang X, Zhang H, et al. The evolving roles of canonical WNT signaling in stem cells and tumorigenesis: implications in targeted cancer therapies. Lab Invest 2016; 96: 116-36.
DOI PMID |
| 8. |
Wang H, Fan R, Wang XQ, et al. Methylation of Wnt antagonist genes: a useful prognostic marker for myelodysplastic syndrome. Ann Hematol 2013; 92: 199-209.
DOI PMID |
| 9. |
Reya T, Duncan AW, Ailles L, et al. A role for Wnt signalling in self-renewal of haematopoietic stem cells. Nature 2003; 423: 409-14.
DOI |
| 10. |
Willert K, Brown JD, Danenberg E, et al. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 2003; 423: 448-52.
DOI |
| 11. |
Fenaux P. Myelodysplastic syndromes: from pathogenesis and prognosis to treatment. Semin Hematol 2004; 41: 6-12.
PMID |
| 12. |
Stomper J, Rotondo JC, Greve G, Lubbert M. Hypomethylating agents (HMA) for the treatment of acute myeloid leukemia and myelodysplastic syndromes: mechanisms of resistance and novel HMA-based therapies. Leukemia 2021; 35: 1873-89.
DOI PMID |
| 13. | Gil-Perez A, Montalban-Bravo G. Management of myelodysplastic syndromes after failure of response to hypomethylating agents. Ther Adv Hematol 2019; 10: 1-18. |
| 14. |
Zeidan AM, Hu X, Zhu W, et al. Association of provider experience and clinical outcomes in patients with myelodysplastic syndromes receiving hypomethylating agents. Leuk Lymphoma 2020; 61: 397-408.
DOI URL |
| 15. | Bernal T, Martínez-Camblor P, Sánchez-García J, et al. Effectiveness of azacitidine in unselected high-risk myelodysplastic syndromes: results from the Spanish registry. Leukemia 2015; 29: 1875-81. |
| 16. |
Fenaux P, Mufti GJ, Hellstrom-Lindberg E, et al. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase Ⅲ study. Lancet Oncol 2009; 10: 223-32.
DOI URL |
| 17. |
Zeidan A, Sekeres M, Garcia-Manero G, et al. Comparison of risk stratification tools in predicting outcomes of patients with higher-risk myelodysplastic syndromes treated with azanucleosides. Leukemia 2016; 30: 649-57.
DOI PMID |
| 18. |
Zeidan AM, Stahl M, Sekeres MA, Steensma DP, Komrokji RS, Gore SD. A call for action: increasing enrollment of untreated patients with higher-risk myelodysplastic syndromes in first-line clinical trials. Cancer 2017; 123: 3662-72.
DOI PMID |
| 19. |
Gao C, Wang J, Li Y, et al. Incidence and risk of hematologic toxicities with hypomethylating agents in the treatment of myelodysplastic syndromes and acute myeloid leukopenia: a systematic review and Meta-analysis. Medicine 2018; 97:e11860.
DOI URL |
| 20. |
Boumber Y, Kantarjian H, Jorgensen J, et al. A randomized study of decitabine versus conventional care for maintenance therapy in patients with acute myeloid leukemia in complete remission. Leukemia 2012; 26: 2428-31.
DOI PMID |
| 21. |
Dombret H, Seymour JF, Butrym A, et al. International phase 3 study of azacitidine vs conventional care regimens in older patients with newly diagnosed AML with > 30% blasts. Blood 2015; 126: 291-9.
DOI PMID |
| 22. |
Lübbert M, Suciu S, Baila L, et al. Low-dose decitabine versus best supportive care in elderly patients with intermediate-or high-risk myelodysplastic syndrome (MDS) ineligible for intensive chemotherapy: final results of the randomized phase III study of the European Organisation for Research and Treatment of Cancer Leukemia Group and the German MDS Study Group. J Clin Oncol 2011; 29: 1987-96.
DOI URL |
| 23. |
Palmieri R, Paterno G, De Bellis E, et al. Therapeutic choice in older patients with acute myeloid leukemia: a matter of fitness. Cancers 2020; 12: 120.
DOI URL |
| 24. | Li D. Clinic study of the patients infection rate with myelodysplastic syndromes decreased by Yisui granules. Zhong Guo Yi Yao Xue Bao 1998; 13: 27-30. |
| 25. | Hu KW, Sun YL, Le ZS, et al. A clinical study on the treatment of myeloproliferative syndrome by strengthening Qi, nourishing Yin and activating the blood. Beijing Zhong Yi Yao Da Xue Xue Bao 1994; 17: 39-44. |
| 26. | Li R, Pan Y, Wu J, et al. Study on Yisui granules in improving anemia and related symptoms in patients with low- and intermediate-risk type I myelodysplastic syndromes. Beijing Zhong Yi Yao 2021; 40: 456-60. |
| 27. | Ma J. Clinical observation of Yisui granule treating chemotherapy related-anemia. Shanxi Zhong Yi 2016; 32: 13-5, 20. |
| 28. | Chen XY, Wei Y, Su W, et al. Influence of Yisui granules on T-cell subgroups of patients with myelodysplastic syndromes analysed. Zhong Guo Zhong Yi Ji Chu Yi Xue Za Zhi 1995: 32-3. |
| 29. | Wu X, Li D, Chen X, et al. Study on the effect of Yisui granule on CD4+CD25+Foxp3+Treg cell and its related cytokines in spleen of ITP mouse. Dang Dai Yi Xue 2010; 16:1-3. |
| 30. | Shi X. Modern medical laboratory animal science. Beijing: Beijing People's Military Medical Press, 2000: 239-323. |
| 31. |
Yang X, Feng Y, Liu Y, et al. Fuzheng Jiedu Xiaoji formulation inhibits hepatocellular carcinoma progression in patients by targeting the AKT/CyclinD1/p21/p27 pathway. Phytomedicine 2021; 87: 153575.
DOI URL |
| 32. |
Gruenbaum Y, Cedar H, Razin A. Substrate and sequence specificity of a eukaryotic DNA methylase. Nature 1982; 295: 620-2.
DOI |
| 33. | Bhagat TD, Chen S, Bartenstein M, et al. Epigenetically aberrant stroma in MDS propagates disease via Wnt/beta-catenin activation. Cancer Res 2017; 77: 4846-57. |
| 34. |
Cui C, Zhou X, Zhang W, Qu Y, Ke X. Is β-catenin a druggable target for cancer therapy? Trends Biochem Sci 2018; 43: 623-34.
DOI PMID |
| 35. |
Lecarpentier Y, Schussler O, Hébert J-L, Vallée A. Multiple targets of the canonical WNT/β-catenin signaling in cancers. Front Oncol 2019; 9: 1248.
DOI PMID |
| 36. |
Canaani J. Emerging therapies for the myelodysplastic syndromes. Clin Hematol Int 2020; 2:13.
DOI URL |
| 37. |
Gao X, Wang YY, Li YX, et al. Huganpian, a Traditional Chinese Medicine, inhibits liver cancer growth in vitro and in vivo by inducing autophagy and cell cycle arrest. Biomed Pharmacother 2019; 120: 109469.
DOI URL |
| 38. | Kostroma II, Gritsaev SV, Sidorova ZY, et al. Aberrant methylation of promoter regions of sox7, p15ink4b and WNT pathway antagonist genes in patients with myelodysplastic syndrome. Haematologica 2016; 101: 495-6. |
| 39. | Gao LS, Meng SP. Comparative study between HL-60 reduced by Yisuiling and single nucleus of umbilical cord blood apoptosis. Zhong Hua Zhong Yi Yao Za Zhi 2000; 15: 26-8. |
| 40. | Zhang HJ, Song YH, Sun YL, Chen XY. The effect of Yisui Ling on the proliferation and differentiation of a leukemic megakaryocyte cell line HI-MEG. Zhong Guo Zu Zhi Hua Xue Yu Xi Bao Hua Xue Za Zhi 1996; 5: 483-8. |
| 41. | Zhang HJ, Chen XY, Song YH, Hu KW, Sun YL. Preliminary study on effect of YSL on gene expressions of HI-Meg. Zhong Guo Zhong Yi Ji Chu Yi Xue Za Zhi 1996; 2: 43+8. |
| 42. |
Duffy MJ, O'Grady S, Tang M, Crown J. MYC as a target for cancer treatment. Cancer Treat Rev 2021; 94: 102154.
DOI URL |
| 43. | Li S, Deng G, Su J, et al. A novel all-trans retinoic acid derivative regulates cell cycle and differentiation of myelodysplastic syndrome cells by USO1. Eur J Pharmacol 2021: 174199. |
| 44. | Huang LJ, Dai D, Shen XH. Exploring the action mechanism of astragalus-codonopsis on myelodysplastic syndrome based on network pharmacology. Zhong Yi Lin Chuang Yan Jiu 2021; 13: 1-6. |
| 45. | Gong JM, Zhou YQ, Lin Q. Hydroxysafflor yellow A inhibits ovarian cancer growth through Wnt/β-catenin signaling pathway. Yi Xue Yan Jiu Za Zhi 2019; 48: 131-4. |
| 46. | Wang LY, Sun JL, Su JR. Effect of total flavonoids from caulis spatholepis on human breast cancer cell line MCF-7 and its regulation of Wnt/β-catenin pathway. Zhong Yi Xue Bao 2021; 36: 1512-8. |
| 47. |
Lam W, Ren Y, Guan F, et al. Mechanism based quality control (MBQC) of herbal products: a case study YIV-906 (PHY906). Front Pharmacol 2018; 9: 1324.
DOI PMID |
| [1] | FAN Yifu, ZHAO Hetong, ZHANG Yani, Yani Zifei, DU Juan, LING Changquan. Effectiveness of Jiedu granule (解毒颗粒) on gut microbiota in patients with advanced hepatocellular carcinoma: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 988-996. |
| [2] | YAO Nan, CHEN Guocai, LU Yanyan, XU Xuemeng, ZHAO Chuanxi, HUANG Xuejun, LIU Wengang, PENG Sha, WU Huai. Bushen Qiangjin capsule(补肾强筋胶囊) inhibits the Wnt/β-catenin pathway to ameliorate papain-induced knee osteoarthritis in rats [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 935-942. |
| [3] | XU Qian, QIN Wei, WU Fangzhen, LIN Yao, HONG Liting, CHEN Dan, HU Xuefeng, CAI Jing. Effect of Roucongrong(Herba Cistanches Deserticolae) decoction on the substantia nigra through Wnt/β-catenin signaling pathway in rats with Parkinson's disease induced by 6-hydroxydopamine hydrochloride [J]. Journal of Traditional Chinese Medicine, 2021, 41(5): 762-770. |
| [4] | DONG Lei, XU Pei. Danzhi Jiangtang capsule (丹蛭降糖胶囊) alleviate hyperglycemiaand periodontitis via Wnt/β-catenin signaling in diabetic rats [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 608-616. |
| [5] | WANG Dexiu, XU Yonggang, DU Yu, WANG Hongzhi, LI Liu, HU Xiaomei, YANG Xiupeng, MA Rou, ZHOU Qingbing. Arsenic concentration in peripheral blood is correlated with efficacy of a Traditional Chinese Medicine regimen containing realgar for the treatment of myelodysplastic syndromes [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 630-635. |
| [6] | GU Xiaoli, CHEN Menglei, LIU Minghui, ZHANG Zhe, ZHAO Weiwei, CHENG Wenwu. Value of Traditional Chinese Medicine syndrome differentiation in predicting the survival time of patients with advanced cancer [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 636-641. |
| [7] | Wang Lei, Li Xiu, Zhou Rongyao, Shan Yawei. Effect of Bushen Jianpi formula on survival of patients with moderate and advanced hepatocellular carcinoma: a retrospective study [J]. Journal of Traditional Chinese Medicine, 2020, 40(4): 683-689. |
| [8] | Li Yifan, Li Juan, Fan Bifa, Wang Yitong, Jiang Juling, Zhang Zhenhua, Wang Xinxing, Lu Wenping. Efficacy and safety of Yiqi Huoxue Jiedu decoction for the treatment of advanced epithelial ovarian cancer patients: a double-blind randomized controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2020, 40(1): 103-111. |
| [9] | Zheng Lixiang, Zheng Qing, Yu Zhipeng, Wang Jian, Ren Xiaoying, Gong Yan, Yang Xue, Hunag Ping, Weng Meizhi, Liu Hongning, Liu Haizhou. Liuwei Dihuang pill suppresses metastasis by regulating the wnt pathway and disrupting β-catenin/T cell factor interactions in a murine model of triple-negative breast cancer [J]. Journal of Traditional Chinese Medicine, 2019, 39(06): 826-832. |
| [10] | Guan Huiting, Xie Su, Liu Shangyi, Xie Qing, Hou Shengkai, Liu Huarong, Zhang Yunjie, Hu Yaqing, Zhang Chenyu. Effects of Jiazhu decoction in combination with cyclophosphamide on breast cancer in mice [J]. Journal of Traditional Chinese Medicine, 2019, 39(05): 642-648. |
| [11] | Su Tao, Wang Xinning, Li Chunyu, Bai Jingxuan, Chi-Yan Cheng, Fu Xiuqiong, Li Ting, Yu Zhiling. An ethanolic extract of Bailian(Radix Ampelopsis Japonicae):demonstration of colorectal cancer treatment efficacy via inhibition of β-catenin signaling in vitro [J]. Journal of Traditional Chinese Medicine, 2019, 39(03): 339-345. |
| [12] | Li Kai, Liu Rui, Xu Guangzhong, Zhang Weihua, Liu Chen, Zhu Bin, Hua Baojin, Feng Xingzhong. Effect of a modified Banxia Xiexin decoction plus chemotherapy on stage Ⅲ colon cancer [J]. Journal of Traditional Chinese Medicine, 2019, 39(02): 251-257. |
| [13] | Fu Shuping, Yang Li, Hong Hao, Zhang Ronghua. Wnt/β-catenin signaling is involved in the Icariin induced proliferation of bone marrow mesenchymal stem cells [J]. Journal of Traditional Chinese Medicine, 2016, 36(03): 360-368. |
| [14] | Li Qiang, Liu Zhibin, Yang Jiping, Guo Huijun, Xu Liran. Comment on the intervention of Traditional Chinese Medicine on survival rates of patients living with human immunodeficiency virus and acquired immune deficiency syndrome [J]. Journal of Traditional Chinese Medicine, 2016, 36(03): 409-410. |
| [15] | Liu Zhibin, Meng Changhai, Yang Jiping, Chen Xiumin, Jin Yantao. Survival rate:an indicator of the management of acquired immune deficiency syndrome using Traditional Chinese Medicine [J]. Journal of Traditional Chinese Medicine, 2015, 35(04): 473-477. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

