Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (1): 160-171.DOI: 10.19852/j.cnki.jtcm.2026.01.015
• Original Articles • Previous Articles Next Articles
Exploring the mechanism of the Lianshi Jianpi formula (莲实健脾方) in treating impaired glucose tolerance: a network pharmacology, molecular docking, and experimental validation study
JIANG Mingqian1,2, WANG Tong3, HUANG Bowei2, QIU Chen1, LIANG Yanbin4, YE Binhua1( )
)
- 1 Department of Endocrinology and Metabolism, People's Hospital Affiliated to Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China
2 First Clinical Medicine College, Fujian University of Traditional Chinese Medicine, Fuzhou 350122, China
3 College of Traditional Chinese Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou 350122, China
4 Department of Clinical Nutrition, People's Hospital Affiliated to Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China
-
Received:2025-04-19Accepted:2025-10-09Online:2026-02-15Published:2026-01-28 -
Contact:Prof. YE Binhua, Department of Endocrinology and Metabolism, People's Hospital Affiliated to Fujian University of Traditional Chinese Medicine, Fuzhou 350004, Fujian, China.Ybinhua2022@163.com ;Telephone: +86-13275911822 -
Supported by:Natural Science Foundation of Fujian Province Project: To Investigate the Mechanism of Lianshi Jianpi Formula In Improving Impaired Glucose Tolerance via the Fibroblast Growth Factor 21-Mediated Adenosine 5‘-monophosphate-Activated Protein Kinases Energy Metabolism Pathway(2023J01841);ZHOU Guoying Fujian Province Famous Traditional Chinese Medicine Inheritance Workstation Construction Project(Min Zhong Yi [2023] 56)
Cite this article
JIANG Mingqian, WANG Tong, HUANG Bowei, QIU Chen, LIANG Yanbin, YE Binhua. Exploring the mechanism of the Lianshi Jianpi formula (莲实健脾方) in treating impaired glucose tolerance: a network pharmacology, molecular docking, and experimental validation study[J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 160-171.
share this article

Figure 1 Herb-compound-target network and GO biological process analysis A: "Herbs-active compounds-hub therapeutic target proteins" network of LSJPF for IGT. Ellipses represent the hub therapeutic targets, V-shaped nodes represent herbs, and diamond-shaped nodes represent active compounds. Edges represent the relationships between herbs, active compounds, and hub therapeutic target proteins; B: GO enrichment analysis of the 12 hub targets: Top 20 biological process terms with the most significant P-values. IGT: impaired glucose tolerance; LSJPF: Lianshi Jianpi formula; GO: gene ontology. SLC6A3: sodium-dependent dopamine transporter; PRKACA: cAMP-dependent protein kinase catalytic subunit alpha; AKT1: RAC-alpha serine/threonine-protein kinase; MMP2: matrix metallopeptidase 2; ALB: albumin; CASP3: caspase-3; CAV1: caveolin-1; PTGS2: prostaglandin-endoperoxide synthase 2; JUN: jun proto-oncogene, AP-1 transcription factor subunit; HSP90AB1: heat shock protein 90 kDa alpha B1; BCL2: B-cell lymphoma 2 family protein; CTNNB1: catenin beta-1.

Figure 2 KEGG pathway enrichment analyses and molecular docking analysis A: sankey-dot plot of KEGG enrichment analysis; B: molecular docking diagrams; B1: AKT1 and beta-sitosterol molecular docking; B2: BCL2 and beta-sitosterol molecular docking; B3: HSP90AB1 and beta-sitosterol molecular docking. SLC6A3: sodium-dependent dopamine transporter; PRKACA: cAMP-dependent protein kinase catalytic subunit alpha; AKT1: RAC-alpha serine/threonine-protein kinase; MMP2: matrix metallopeptidase 2; ALB: albumin; CASP3: caspase-3; CAV1: caveolin-1; PTGS2: prostaglandin-endoperoxide synthase 2; JUN: jun proto-oncogene, AP-1 transcription factor subunit; HSP90AB1: heat shock protein 90 kDa alpha B1; BCL2: B-cell lymphoma 2 family protein; CTNNB1: catenin beta-1; PI3K: phosphatidylinositol-3-kinase.
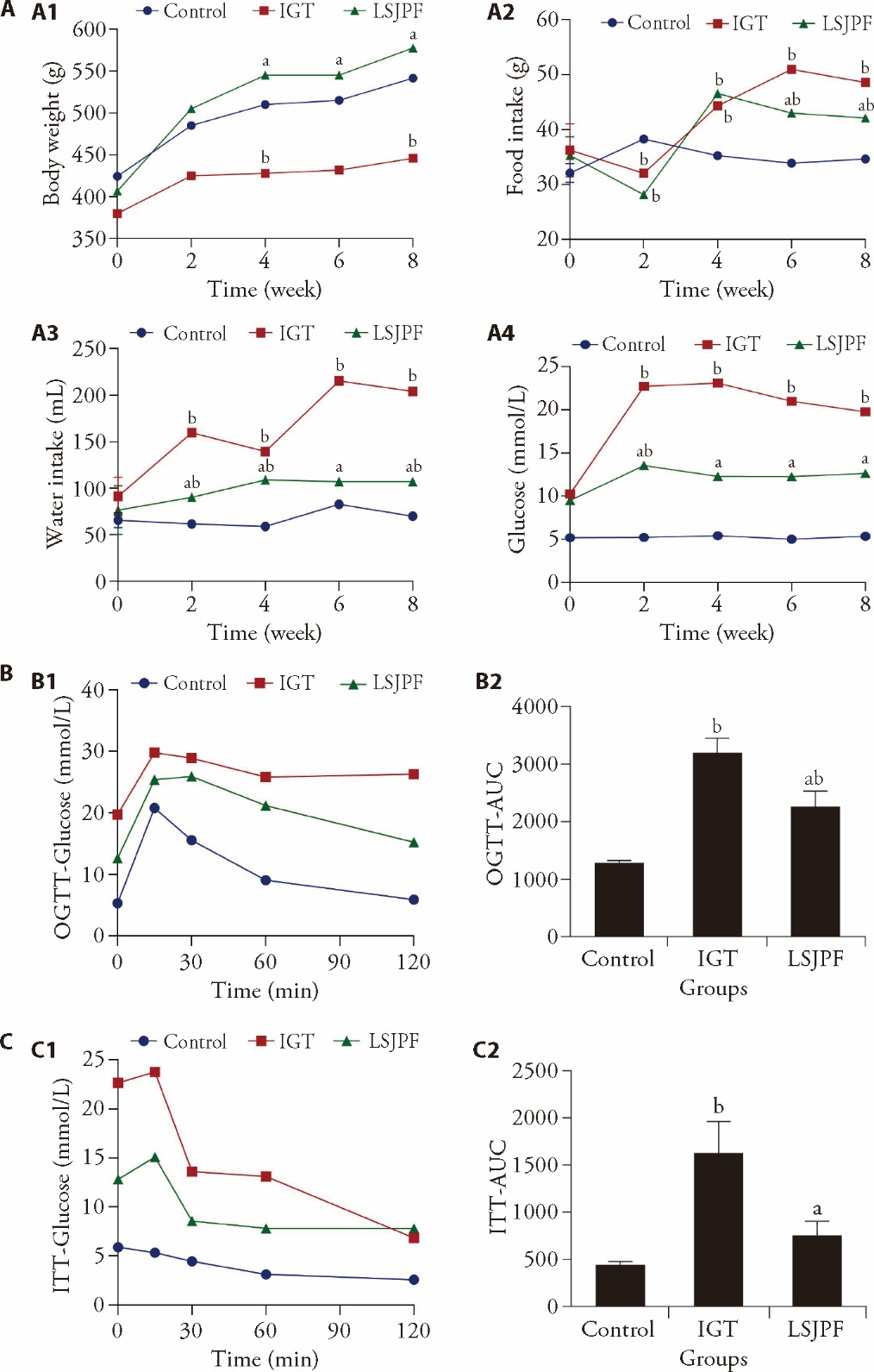
Figure 3 Effects of LSJPF treatment on body weight, food intake, water intake and glucose metabolism in IGT rats A: effects of LSJPF treatment on body weight, food intake, water intake and glucose metabolism in different time points; A1: body weight; A2: food intake; A3: water intake. A4: FPG; B: OGTT results in each group; B1: curve of OGTT at week 8; B2: AUC of OGTT; C: ITT results in each group; C1: Curve of ITT at week 8; C2: AUC of ITT. Control group: normal SD rats fed with standard diet; IGT group: IGT model rats fed with high-fat diet; LSJPF group: IGT model rats fed with 2/3 of the high-fat diet and 1/3 of the LSJPF. IGT: impaired glucose tolerance; LSJPF: Lianshi Jianpi formula; FPG: fasting plasma glucose; AUC: area under the curve; OGTT: oral glucose tolerance test; ITT: insulin tolerance test. Statistical analyses were measured by using one-way analysis of variance to analyse the differences between the groups. Data were shown as mean ± standard error of the mean (n = 8). aP < 0.05, compared with IGT group; bP < 0.05, compared with Control group.
| Group | n | HbA1c (%) | Lee’s index | Visceral fat percentage (%) | TG (mmol/L) | HDL-C (mmol/L) | CHOL (mmol/L) | LDL-C (mmol/L) |
|---|---|---|---|---|---|---|---|---|
| Control | 8 | 5.800±0.743 | 294.078±5.751 | 0.273±0.006 | 0.573±0.162 | 0.515±0.092 | 1.965±0.512 | 0.166±0.193 |
| IGT | 8 | 12.171±3.995a | 290.882±13.881 | 0.349±0.275 | 0.946±0.545a | 0.555±0.244 | 3.904±2.003a | 1.570±0.976a |
| LSJPF | 8 | 7.671±3.967b | 301.111±9.263 | 0.319±0.023 | 0.586±0.138b | 0.445±0.065 | 2.213±0.440b | 0.653±0.192b |
Table 1 Effects of LSJPF treatment on HbA1c and lipid metabolism in IGT rats ($\bar{x} \pm s$)
| Group | n | HbA1c (%) | Lee’s index | Visceral fat percentage (%) | TG (mmol/L) | HDL-C (mmol/L) | CHOL (mmol/L) | LDL-C (mmol/L) |
|---|---|---|---|---|---|---|---|---|
| Control | 8 | 5.800±0.743 | 294.078±5.751 | 0.273±0.006 | 0.573±0.162 | 0.515±0.092 | 1.965±0.512 | 0.166±0.193 |
| IGT | 8 | 12.171±3.995a | 290.882±13.881 | 0.349±0.275 | 0.946±0.545a | 0.555±0.244 | 3.904±2.003a | 1.570±0.976a |
| LSJPF | 8 | 7.671±3.967b | 301.111±9.263 | 0.319±0.023 | 0.586±0.138b | 0.445±0.065 | 2.213±0.440b | 0.653±0.192b |
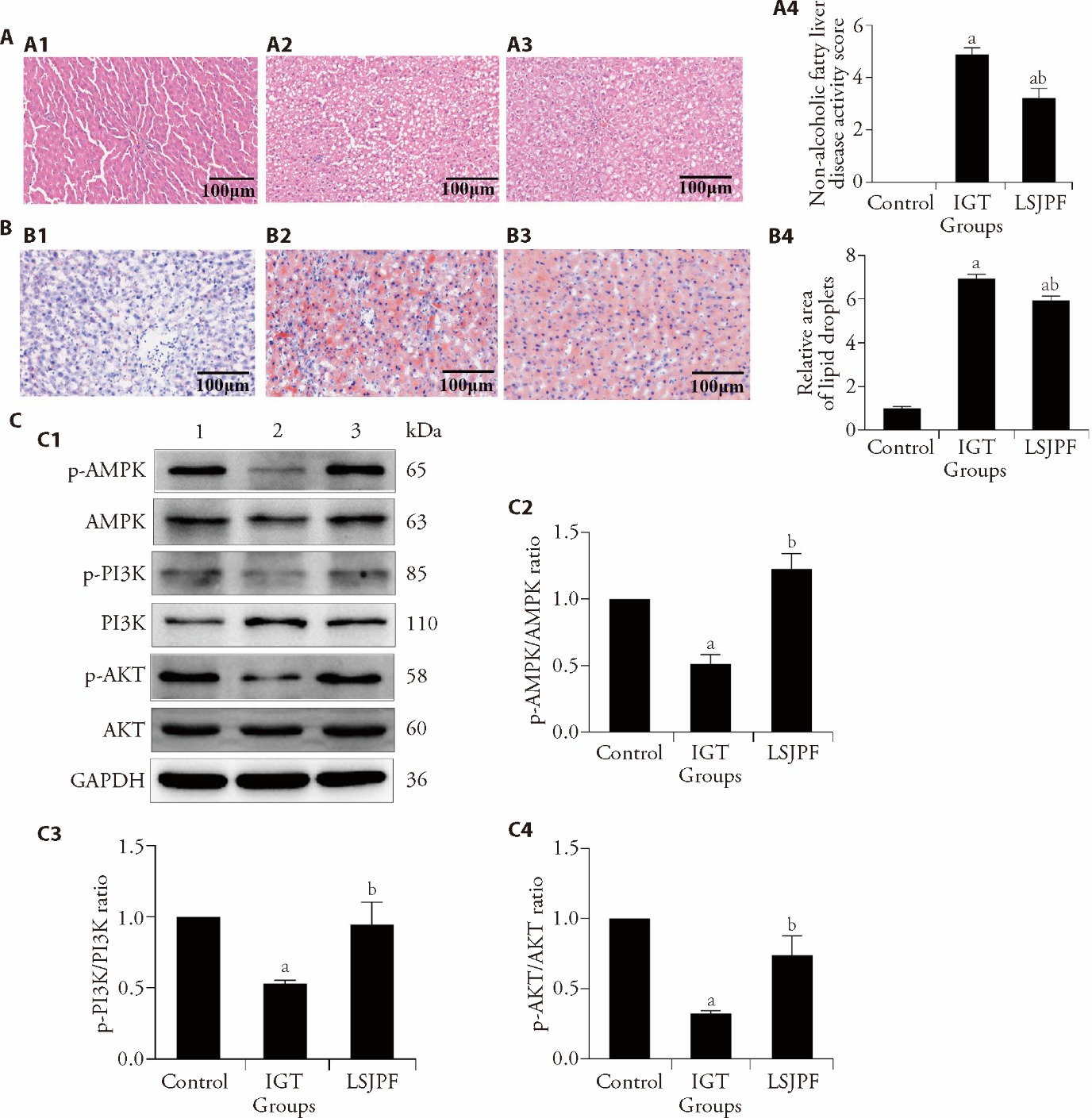
Figure 4 Effects of LSJPF treatment on liver pathology and AMPK/PI3K/AKT pathway in IGT rats A: hematoxylin-eosin staining observes liver tissues. A1: control group; A2: IGT group; A3: LSJPF group; A4: non-alcoholic fatty liver disease activity score; B: oil red O staining observes liver tissues. B1: control group; B2: IGT group; B3: LSJPF group; B4: relative area of lipid droplets. C: protein expression in each group; C1: representative immunoblotting images of p-AMPK, AMPK, p-PI3K, PI3K, p-AKT, AKT and GAPDH; 1: Control group; 2: IGT group; 3: LSJPF group; C2: p-AMPK/AMPK ratio; C3: p- PI3K / PI3K ratio; C4: p-AKT/AKT ration; control group: normal SD rats fed with standard diet; IGT group: IGT model rats fed with high-fat diet; LSJPF group: IGT model rats fed with 2/3 of the high-fat diet and 1/3 of the LSJPF. IGT: impaired glucose tolerance; LSJPF: Lianshi Jianpi formula; p-AMPK: phosphorylated Adenosine 5‘-monophosphate-activated protein kinase; AMPK: Adenosine 5‘-monophosphate-activated protein kinase; p-PI3K: phosphorylated phosphatidylinositol 3-kinase; PI3K: phosphatidylinositol 3-kinase; p-AKT: phosphorylated protein kinase B; AKT: protein kinase B; GAPDH: glyceraldehyde-3-phosphate dehydrogenase. Statistical analyses were measured by using one-way analysis of variance to analyze the differences between the groups. Date were shown as mean ± standard error of the mean (n = 3). aP < 0.05, compared with Control group; bP < 0.05 compared with IGT group.
| 1. |
Rooney MR, Fang M, Ogurtsova K, et al. Global prevalence of prediabetes. Diabetes Care 2023; 46: 1388-94.
DOI PMID |
| 2. |
Ishii H. Cardiovascular events and atherosclerosis in patients with type 2 diabetes and impaired glucose tolerance: what are the medical treatments to prevent cardiovascular events in such patients? J Diabetes Investig 2022; 13: 1114-21.
DOI URL |
| 3. |
Gong Q, Zhang P, Wang J, et al. Morbidity and mortality after lifestyle intervention for people with impaired glucose tolerance: 30-year results of the Da Qing Diabetes Prevention Outcome Study. Lancet Diabetes Endocrinol 2019; 7: 452-61.
DOI URL |
| 4. | Ye BH, Jiang MQ, Qiu C, et al. Clinical observation on Lianshi Jianpi dietary formula on impaired glucose tolerance with failure of transportation due to Spleen Deficiency pattern in 36 cases. Zhong Yi Zha Zhi 2023; 64: 153-8. |
| 5. | Ru J, Li P, Wang J, et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Cheminform 2014; 6: 13. |
| 6. | Fang S, Dong L, Liu L, et al. HERB: a high-throughput experiment- and reference-guided database of Traditional Chinese Medicine. Nucleic Acids Res 2021; 49: D1197-206. |
| 7. |
UniProt: the universal protein knowledgebase. Nucleic Acids Res 2017; 45: D158-69.
DOI URL |
| 8. |
Reese TC, Devineni A, Smith T, Lalami I, Ahn JM, Raj GV. Evaluating physiochemical properties of FDA-approved orally administered drugs. Expert Opin Drug Discov 2024; 19: 225-38.
DOI URL |
| 9. |
Wang X, Liao Z, Zhao G, et al. Curcumin nanocrystals self-stabilized pickering emulsion freeze-dried powder: development, characterization, and suppression of airway inflammation. Int J Biol Macromol 2023; 245: 125493.
DOI URL |
| 10. |
Wang X, Tan Y, Liu F, et al. Pharmacological network analysis of the functions and mechanism of kaempferol from Du Zhong in intervertebral disc degeneration (IDD). J Orthop Translat 2023; 39: 135-46.
DOI URL |
| 11. | Piñero J, Ramírez-Anguita JM, Saüch-Pitarch J, et al. The DisGeNET knowledge platform for disease genomics: 2019 update. Nucleic Acids Res 2020; 48: D845-55. |
| 12. |
Sayers EW, Bolton EE, Brister JR, et al. Database resources of the national center for biotechnology information. Nucleic Acids Res 2022; 50: D20-6.
DOI URL |
| 13. | Stelzer G, Rosen N, Plaschkes I, et al. The GeneCards suite: from gene data mining to disease genome sequence analyses. Curr Protoc Bioinformatics 2016; 54: 1.30. 1-1.30.33. |
| 14. | Szklarczyk D, Gable AL, Nastou KC, et al. The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res 2021; 49: D605-12. |
| 15. |
Oh KK, Gupta H, Min BH, et al. Elucidation of prebiotics, probiotics, postbiotics, and target from gut microbiota to alleviate obesity via network pharmacology study. Cells 2022; 11: 2903.
DOI URL |
| 16. |
Huang P, Yan L, Li Z, et al. Potential shared gene signatures and molecular mechanisms between atherosclerosis and depression: evidence from transcriptome data. Comput Biol Med 2023; 152: 106450.
DOI URL |
| 17. |
Wang L, Zong L, Cao D, Guan M. Unraveling the molecular mechanisms of PFOA in clear cell renal cell carcinoma through network toxicology and molecular docking strategies. Int J Surg 2025; 111: 4842-53.
DOI PMID |
| 18. |
Zhou Y, Zhou B, Pache L, et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun 2019; 10: 1523.
DOI PMID |
| 19. |
Kim S, Chen J, Cheng T, et al. PubChem 2019 update: improved access to chemical data. Nucleic Acids Res 2019; 47: D1102-9.
DOI |
| 20. |
Velankar S, Burley SK, Kurisu G, Hoch JC, Markley JL. The protein data bank archive. Methods Mol Biol 2021; 2305: 3-21.
DOI PMID |
| 21. | Yang Y, Chen XY, Yao JK, HU YY, Wang W. Efficacy of Danlou tablet on myocardial ischemia/ reperfusion injury assessed by network pharmacology and experimental verification. J Tradit Chin Med 2024; 44: 131-44. |
| 22. |
Preciado-Saldana AM, Lopez-Diaz JA, Dominguez-Avila JA, et al. Revisiting the high-fat diet/low streptozotocin prediabetic rat model: a bioanalytical adjustment. J Pharmacol Toxicol Methods 2023; 120: 107252.
DOI URL |
| 23. |
Guo C, Zhang X, Yu Y, Wu Y, Xie L, Chang C. Lonicerae Japonicae Flos extract and chlorogenic acid attenuates high-fat-diet- induced prediabetes via CTRPs-AdipoRs-AMPK/PPARalpha axes. Front Nutr 2022; 9: 1007679.
DOI URL |
| 24. |
Brunt EM, Kleiner DE, Wilson LA, Belt P, Neuschwander-Tetri BA, Network NCR. Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: distinct clinicopathologic meanings. Hepatology 2011; 53: 810-20.
DOI PMID |
| 25. |
Camargo FN, Matos SL, Araujo LCC, Carvalho CRO, Amaral AG, Camporez JP. Western diet-fed apoe knockout male mice as an experimental model of non-alcoholic steatohepatitis. Curr Issues Mol Biol 2022; 44: 4692-703.
DOI PMID |
| 26. |
Duan P, Yang K, Su X, et al. HTINet2: herb-target prediction via knowledge graph embedding and residual-like graph neural network. Brief Bioinform 2024; 25: bbae414.
DOI URL |
| 27. |
Liu TT, Zeng KW. Recent advances in target identification technology of natural products. Pharmacol Ther 2025; 269: 108833.
DOI URL |
| 28. |
Sun Y, Li C, Deng X, et al. Target protein identification in live cells and organisms with a non-diffusive proximity tagging system. Elife 2024; 13: RP102667.
DOI URL |
| 29. |
Liu Y, Wang W, Liang B, Zou Z, Zhang A. NLRP3 inflammasome activation and disruption of IRS-1/PI3K/AKT signaling: potential mechanisms of arsenic-induced pancreatic beta cells dysfunction in rats. Ecotoxicol Environ Saf 2025; 289: 117504.
DOI URL |
| 30. |
Wang G, Ge L, Liu T, Zheng Z, Chen L. The therapeutic potential of arctigenin against multiple human diseases: a mechanistic review. Phytomedicine 2023; 110: 154647.
DOI URL |
| 31. | Holm H, Kennback C, Laucyte-Cibulskiene A, Nilsson PM, Jujic A. The impact of prediabetes and diabetes on endothelial function in a large population-based cohort. Blood Press 2024; 33: 2298309. |
| 32. |
Yang J, Shangguan Q, Xie G, Yang M, Sheng G. Sex-specific associations between haemoglobin glycation index and the risk of cardiovascular and all-cause mortality in individuals with pre-diabetes and diabetes: a large prospective cohort study. Diabetes Obes Metab 2024; 26: 2275-83.
DOI URL |
| 33. | Galaviz KI, Narayan KMV, Lobelo F, Weber MB. Lifestyle and the prevention of type 2 diabetes: a status report. Am J Lifestyle Med 2018; 12: 4-20. |
| 34. |
Jiang Q, Li JT, Sun P, Wang LL, Sun LZ, Pang SG. Effects of lifestyle interventions on glucose regulation and diabetes risk in adults with impaired glucose tolerance or prediabetes: a Meta-analysis. Arch Endocrinol Metab 2022; 66: 157-67.
DOI PMID |
| 35. |
Li X, Geng-Ji JJ, Quan YY, et al. Role of potential bioactive metabolites from Traditional Chinese Medicine for type 2 diabetes mellitus: an overview. Front Pharmacol 2022; 13: 1023713.
DOI URL |
| 36. |
Sbraccia P, D'Adamo M, Guglielmi V. Is type 2 diabetes an adiposity-based metabolic disease? From the origin of insulin resistance to the concept of dysfunctional adipose tissue. Eat Weight Disord 2021; 26: 2429-41.
DOI PMID |
| 37. |
Lee WH, Najjar SM, Kahn CR, Hinds TD Jr. Hepatic insulin receptor: new views on the mechanisms of liver disease. Metabolism 2023; 145: 155607.
DOI URL |
| 38. | Najjar SM, Perdomo G. Hepatic insulin clearance: mechanism and physiology. Physiology (Bethesda) 2019; 34: 198-215. |
| 39. |
Miao L, Zhang X, Zhang H, et al. Baicalin ameliorates insulin resistance and regulates hepatic glucose metabolism via activating insulin signaling pathway in obese pre-diabetic mice. Phytomedicine 2024; 124: 155296.
DOI URL |
| 40. |
Yang YY, Qi JJ, Jiang SY, Ye L. Esculin ameliorates obesity-induced insulin resistance by improving adipose tissue remodeling and activating the IRS1/PI3K/AKT/GLUT4 pathway. J Ethnopharmacol 2024; 319: 117251.
DOI URL |
| 41. |
Yan J, Wang C, Jin Y, et al. Catalpol ameliorates hepatic insulin resistance in type 2 diabetes through acting on AMPK/NOX4/PI3K/AKT pathway. Pharmacol Res 2018; 130: 466-80.
DOI PMID |
| 42. |
Wang D, Kang X, Zhang L, et al. TRIB2-mediated modulation of AMPK promotes hepatic insulin resistance. Diabetes 2024; 73: 1199-214.
DOI PMID |
| 43. |
Lin SC, Hardie DG. AMPK: sensing glucose as well as cellular energy status. Cell Metab 2018; 27: 299-313.
DOI URL |
| 44. |
Entezari M, Hashemi D, Taheriazam A, et al. AMPK signaling in diabetes mellitus, insulin resistance and diabetic complications: a pre-clinical and clinical investigation. Biomed Pharmacother 2022; 146: 112563.
DOI URL |
| 45. |
Schultze SM, Hemmings BA, Niessen M, Tschopp O. PI3K/AKT, MAPK and AMPK signalling: protein kinases in glucose homeostasis. Expert Rev Mol Med 2012; 14: e1.
DOI URL |
| 46. |
Fan X, Jiao G, Pang T, et al. Ameliorative effects of mangiferin derivative TPX on insulin resistance via PI3K/AKT and AMPK signaling pathways in human HepG2 and HL-7702 hepatocytes. Phytomedicine 2023; 114: 154740.
DOI URL |
| 47. |
Zhao H, Zhai BW, Zhang MY, et al. Phlorizin from Lithocarpus litseifolius [Hance] Chun ameliorates FFA-induced insulin resistance by regulating AMPK/PI3K/AKT signaling pathway. Phytomedicine 2024; 130: 155743.
DOI URL |
| 48. |
Babu S, Jayaraman S. An update on beta-sitosterol: a potential herbal nutraceutical for diabetic management. Biomed Pharmacother 2020; 131: 110702.
DOI URL |
| 49. |
Babu S, Krishnan M, Rajagopal P, et al. Beta-sitosterol attenuates insulin resistance in adipose tissue via IRS-1/Akt mediated insulin signaling in high fat diet and sucrose induced type-2 diabetic rats. Eur J Pharmacol 2020; 873: 173004.
DOI URL |
| 50. |
Jayaraman S, Devarajan N, Rajagopal P, et al. Beta-sitosterol circumvents obesity induced inflammation and insulin resistance by down-regulating IKK beta/NF-kappa B and JNK signaling pathway in adipocytes of type 2 diabetic rats. Molecules 2021; 26: 2101.
DOI URL |
| 51. |
Khan Z, Nath N, Rauf A, et al. Multifunctional roles and pharmacological potential of beta-sitosterol: emerging evidence toward clinical applications. Chem Biol Interact 2022; 365: 110117.
DOI URL |
| 52. |
Ponnulakshmi R, Shyamaladevi B, Vijayalakshmi P, Selvaraj J. In silico and in vivo analysis to identify the antidiabetic activity of beta sitosterol in adipose tissue of high fat diet and sucrose induced type-2 diabetic experimental rats. Toxicol Mech Methods 2019; 29: 276-90.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

