Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (4): 734-743.DOI: 10.19852/j.cnki.jtcm.20230404.001
Previous Articles Next Articles
Acupotomy ameliorates subchondral bone absorption and mechanical properties in rabbits with knee osteoarthritis by regulating bone morphogenetic protein 2-Smad1 pathway
Chen Xilin1, GUO Yan2( ), LU Juan3, QIN Luxue1, HU Tingyao1, ZENG Xin1, WANG Xinyue1, ZHANG Anran1, ZHUANG Yuxin1, ZHONG Honggang4, GUO Changqing1(
), LU Juan3, QIN Luxue1, HU Tingyao1, ZENG Xin1, WANG Xinyue1, ZHANG Anran1, ZHUANG Yuxin1, ZHONG Honggang4, GUO Changqing1( )
)
- 1 School of Acupuncture-Moxibustion and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China
2 Acupuncture and Moxibustion Department, Beijing Hospital of Traditional Chinese Medicine affiliated with Capital Medical University, Beijing 100010, China
3 Shenzhen Hospital of Southern Medical University, Shenzhen 518000, China
4 Institute of Bone Injury, China Academy of Chinese Medical Sciences, Beijing 100010, China
-
Received:2022-12-02Accepted:2023-03-13Online:2023-08-15Published:2023-04-04 -
Contact:Dr. GUO Yan, Acupuncture and Moxibustion Department, Beijing Hospital of Traditional Chinese Medicine affiliated with Capital Medical University, Beijing 100010, China. guoyan7777777@126.com. Telephone: +86-10-64286687
Prof. GUO Changqing, School of Acupuncture-Moxibustion and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China. guochangqing66@163.com -
Supported by:Longitudinal Research Development Fund of Beijing University of Chinese Medicine: to Investigate the Effect of Acupotomy on Subchondral Bone Remodeling in Knee Osteoarthritis Based on BMP2-Smad1 Pathway(2020-ZXFZJJ-031);National Natural Science Foundation of China: to Investigate the Mechanism of Acupotomy "Regulating Tendon and Treating Bone" in the Treatment of KOA based on the Vascularization of Hypoxic Cartilage Mediated by HIF-1α/VEGF Pathway(82074523)
Cite this article
Chen Xilin, GUO Yan, LU Juan, QIN Luxue, HU Tingyao, ZENG Xin, WANG Xinyue, ZHANG Anran, ZHUANG Yuxin, ZHONG Honggang, GUO Changqing. Acupotomy ameliorates subchondral bone absorption and mechanical properties in rabbits with knee osteoarthritis by regulating bone morphogenetic protein 2-Smad1 pathway[J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 734-743.
share this article
| Gene | Primer sequences |
|---|---|
| BMP2 | Forward: 5'-CCCCTTAATTTGCAGAA ACAGTA-3' |
| Reverse: 5'-GATCCCTGGTTTAGGAAAGAAGA-3' | |
| Smad1 | Forward: 5'-CCTTCCAGATGCCAGATGTT-3' |
| Reverse: 5'-CCATCCACCAACACA CTCGT-3' | |
| β-actin | Forward: 5'-TTGTCCCCCAACTTGAGATGTA-3' |
| Reverse: 5'-GCACTTTTATTGAACTGGTCTCGT-3' |
Table 1 Primers used in this study
| Gene | Primer sequences |
|---|---|
| BMP2 | Forward: 5'-CCCCTTAATTTGCAGAA ACAGTA-3' |
| Reverse: 5'-GATCCCTGGTTTAGGAAAGAAGA-3' | |
| Smad1 | Forward: 5'-CCTTCCAGATGCCAGATGTT-3' |
| Reverse: 5'-CCATCCACCAACACA CTCGT-3' | |
| β-actin | Forward: 5'-TTGTCCCCCAACTTGAGATGTA-3' |
| Reverse: 5'-GCACTTTTATTGAACTGGTCTCGT-3' |
| Item | Group | n | Before intervention | After intervention |
|---|---|---|---|---|
| PROM | Control | 9 | 141 (2) | 141 (2.5) |
| KOA | 9 | 40 (9.5)a | 50 (18)a | |
| Acupotomy | 9 | 40.00 (14)a | 90 (27)ac | |
| EA | 9 | 38 (5)a | 98 (15)ac | |
| Lequesne MG | Control | 9 | 0 (0) | 0 (0) |
| KOA | 9 | 8 (1)b | 8 (0.5)b | |
| Acupotomy | 9 | 8 (0.5)b | 4 (1.5)bd | |
| EA | 9 | 7 (0.5)b | 4 (1)bd |
Table 2 Behavioral assessment of rabbits
| Item | Group | n | Before intervention | After intervention |
|---|---|---|---|---|
| PROM | Control | 9 | 141 (2) | 141 (2.5) |
| KOA | 9 | 40 (9.5)a | 50 (18)a | |
| Acupotomy | 9 | 40.00 (14)a | 90 (27)ac | |
| EA | 9 | 38 (5)a | 98 (15)ac | |
| Lequesne MG | Control | 9 | 0 (0) | 0 (0) |
| KOA | 9 | 8 (1)b | 8 (0.5)b | |
| Acupotomy | 9 | 8 (0.5)b | 4 (1.5)bd | |
| EA | 9 | 7 (0.5)b | 4 (1)bd |

Figure 1 Cartilage degeneration under optical microscope A: the knee articular cartilage degeneration stained with Safranin O-Fast Green (× 200); A1: control group; A2: KOA group; A3: acupotomy group; A4: EA group; B: analysis of the Mankin score; C: analysis of the thickness of total articular cartilage. Data were mean ± standard deviation (n = 6). Control group: received no modeling and no intervention; KOA group: modeling for 6 weeks followed by no intervention; acupotomy group: modeling for 6 weeks followed by acupotomy intervention for 3 weeks; EA group: modeling for 6 weeks followed by electroacupuncture intervention for 3 weeks. TAC: total articular cartilage; KOA: knee osteoarthritis; EA: electroacupuncture. Compared with the control group, aP < 0.01, cP < 0.05; compared with the KOA group, bP < 0.01; compared with the EA group, dP < 0.01.

Figure 2 Subchondral bone remodeling under optical microscope A: the knee articular subchondral bone stained with Safranin O-Fast Green (×40); A1: control group; A2: KOA group; A3: acupotomy group; A4: EA group; B: the thickness of Subchondral bone plate; C: bone volume fraction. Data were mean ± standard deviation (n = 6). Control group: received no modeling and no intervention; KOA group: modeling for 6 weeks followed by no intervention; Acupotomy group: modeling for 6 weeks followed by acupotomy intervention for 3 weeks; EA group: modeling for 6 weeks followed by electroacupuncture intervention for 3 weeks. SBP: subchondral bone plate; BV/TV: bone volume/tissue volume; KOA: knee osteoarthritis; EA: electroacupuncture. Compared with the control group, aP < 0.01; compared with the KOA group, bP < 0.01; compared with the EA group, cP < 0.05, dP < 0.01.

Figure 3 Micro-CT scan and bone parameters analysis of subchondral bone A: Micro-CT scan of subchondral bone cylinder samples; A1-A4 shown transverse section; A5-A8 shown coronal section; A9-A12 shown 3D reconstruction; A1, A5, A9: control group; A2, A6, A10: KOA group; A3, A7, A11: acupotomy group; A4, A8, A12: EA group; B: trabecular thickness; C: trabecular separation/spacing; D: bone volume fraction. Data were expressed as mean ± standard deviation (n = 6). Control group: received no modeling and no intervention; KOA group: modeling for 6 weeks followed by no intervention; Acupotomy group: modeling for 6 weeks followed by acupotomy intervention for 3 weeks; EA group: modeling for 6 weeks followed by electroacupuncture intervention for 3 weeks. Tb. Th: the thickness of trabecular bone; Tb. Sp: the separation of trabecular bone; BV/TV: bone volume/tissue volume; KOA: knee osteoarthritis; EA: electroacupuncture. Compared with the control group, aP < 0.05, cP < 0.01; compared with the KOA group, bP < 0.01, dP < 0.05.
| Group | n | Maximum load (N) | Elasticity modulus (MPa) |
|---|---|---|---|
| Control | 6 | 117±21 | 265±42 |
| KOA | 6 | 33±10a | 100±24a |
| Acupotomy | 6 | 107±27bc | 226±45bc |
| EA | 6 | 76±32ab | 181±24ab |
Table 3 Mechanical properties of subchondral bone ($\bar{x}±s$)
| Group | n | Maximum load (N) | Elasticity modulus (MPa) |
|---|---|---|---|
| Control | 6 | 117±21 | 265±42 |
| KOA | 6 | 33±10a | 100±24a |
| Acupotomy | 6 | 107±27bc | 226±45bc |
| EA | 6 | 76±32ab | 181±24ab |
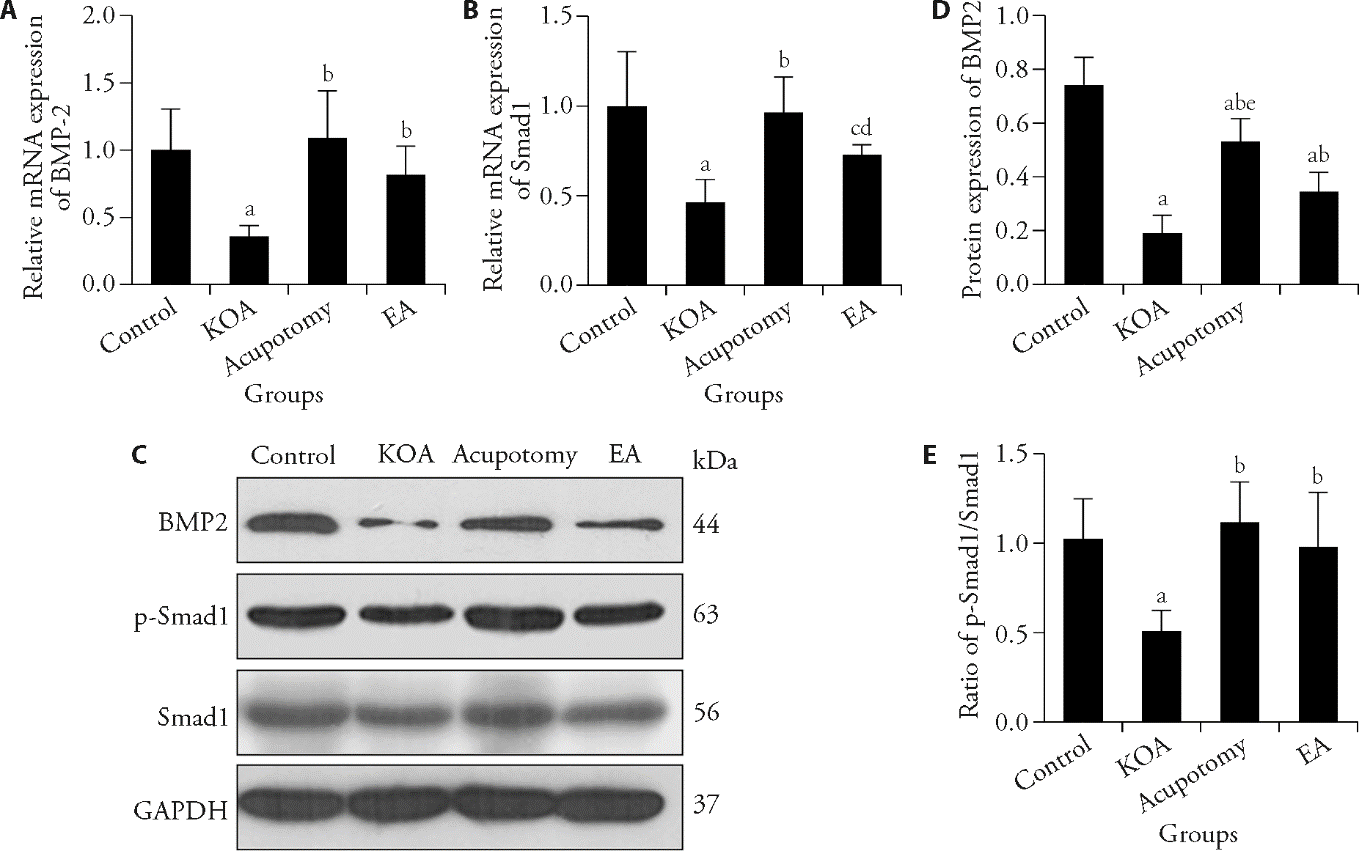
Figure 4 mRNA and protein expression of BMP2-Smad1 in subchondral bone A-B: mRNA expression of BMP2 and Smad1; C-E: Western blot of BMP2, p-Smad1, Smad1. Data were mean ± standard deviation (n = 6). Control group: received no modeling and no intervention; KOA group: modeling for 6 weeks followed by no intervention; Acupotomy group: modeling for 6 weeks followed by acupotomy intervention for 3 weeks; EA group: modeling for 6 weeks followed by electroacupuncture intervention for 3 weeks. BMP2: bone morphogenetic protein 2; KOA: knee osteoarthritis; EA: electroacupuncture. Compared with the control group, aP < 0.01, cP < 0.05; compared with the KOA group, bP < 0.01, dP < 0.05; compared with the EA group, eP < 0.01.
| 1. | Moore N, Salvo F, Duong M, Gulmez SE. Does paracetamol still have a future in osteoarthritis? Lancet 2016; 387: 2065-66. |
| 2. |
Hu W, Chen Y, Dou C, Dong S. Microenvironment in subchondral bone: predominant regulator for the treatment of osteoarthritis. Ann Rheum Dis 2020: 80: 413-22.
DOI PMID |
| 3. |
Felson DT. Osteoarthritis as a disease of mechanics. Osteoarthritis Cartilage 2013; 21: 10-5.
DOI URL |
| 4. | Burr DB, Gallant MA. Bone remodelling in osteoarthritis. Nat Rev Rheumatol 2012; 8: 665-73. |
| 5. | Sims NA, Martin TJ. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. Bonekey Rep 2014; 3: 481. |
| 6. |
Bei MJ, Tian FM, Xiao YP, et al. Raloxifene retards cartilage degradation and improves subchondral bone micro-architecture in ovariectomized rats with patella baja-induced- patellofemoral joint osteoarthritis. Osteoarthritis Cartilage 2020; 28: 344-55.
DOI URL |
| 7. |
Hayes KN, Giannakeas V, Wong A. Bisphosphonate use is protective of radiographic knee osteoarthritis progression among those with low disease severity and being non-overweight: data from the osteoarthritis initiative. J Bone Miner Res 2020; 35: 2318-26.
DOI URL |
| 8. |
Zhu D, Deng X, Han XF, et al. Wedelolactone enhances osteoblastogenesis through ERK- and JNK-mediated BMP2 expression and Smad/1/5/8 phosphorylation. Molecules 2018; 23: 561.
DOI URL |
| 9. |
Iijima H, Ito A, Nagai M, et al. Physiological exercise loading suppresses post-traumatic osteoarthritis progression via an increase in bone morphogenetic proteins expression in an experimental rat knee model. Osteoarthr Cartilage 2016; 25: 964-75.
DOI URL |
| 10. | Guo CQ, Si T, Wen JM, et al. A randomized controlled clinical study on the improvement of pain symptoms of knee osteoarthritis with acupotomy. Tianjin Zhong Yi Yao 2012; 29: 35-8. |
| 11. | Wu YEED, Guo CQ, Wang T, et al. Effect of acupotomy on extensor - flexor atrophy and elastic modulus of muscle tension in rabbit model of mid - stage knee osteoarthritis. Hunan Zhong Yi Yao Da Xue Xue Bao 2019; 39: 1248-53. |
| 12. | Ma SN, Xie ZG, Guo Y, et al. Effect of acupotomy on FAK-PI3K signaling pathways in KOA rabbit articular cartilages. Evid Based Complement Alternat Med 2017; 2017: 4535326. |
| 13. |
Frost HM. Wolff's Law and bone's structural adaptations to mechanical usage: an overview for clinicians. Angle Orthod 1994; 64: 175-88.
DOI PMID |
| 14. | Hugle T, Geurts J. What drives osteoarthritis? -synovial versus subchondral bone pathology. Rheumatology (Oxford) 2017; 56: 1461-71. |
| 15. | Wang T, Guo Y, Shi XW, et al. Acupotomy contributes to suppressing subchondral bone resorption in KOA rabbits by regulating the OPG/RANKL signaling pathway. Evid Based Complement Alternat Med 2021; 2021: 8168657. |
| 16. |
Videman T. Experimental osteoarthritis in the rabbit: comparison of different periods of repeated immobilization. Acta Orthop Scand 1982; 53: 339-47.
PMID |
| 17. | Lequesne MG, Mery C, Samson M, et al. Indexes of severity for osteoarthritis of the hip and knee. Validation--value in comparison with other assessment tests. Scand J Rheumatol 1988; 17: 241. |
| 18. | Guo CQ. Acupotomology. Beijing: China Press of Traditional Chinese Medicine, 2017: 199-204. |
| 19. | Li ZR. Experimental acupuncture science. Beijing: China Press of Traditional Chinese Medicine, 2007: 314-30. |
| 20. |
Nagira K, Ikuta Y, Shinohara M, et al. Histological scoring system for subchondral bone changes in murine models of joint aging and osteoarthritis. Sci Rep 2020; 10: 10077.
DOI PMID |
| 21. |
Felson DT. Developments in the clinical understanding of osteoarthritis. Arthritis Res Ther 2009; 11: 203.
DOI PMID |
| 22. |
O'Neill TW, Felson DT. Mechanisms of osteoarthritis (OA) pain. Curr Osteoporos Rep 2018; 16: 611-6.
DOI PMID |
| 23. |
Zaki S, Blaker CL, Little CB. OA foundations-experimental models of osteoarthritis. Osteoarthritis Cartilage 2022; 30: 357-80.
DOI URL |
| 24. |
Christiansen BA, Guilak F, Lockwood KA, et al. Non-invasive mouse models of post-traumatic osteoarthritis. Osteoarthritis Cartilage 2015; 23: 1627-38.
DOI URL |
| 25. |
Kikuchi T, Sakuta T, Yamaguchi T. Intra-articular injection of collagenase induces experimental osteoarthritis in mature rabbits. Osteoarthritis Cartilage 1998; 6: 177-86.
DOI URL |
| 26. |
Beuningen HM, Glansbeek HL, Kraan PM, Berg WB. Osteoarthritis-like changes in the murine knee joint resulting from intra-articular transforming growth factor-beta injections. Osteoarthritis Cartilage 2000; 8: 25-33.
DOI URL |
| 27. |
Shi X, Yu W, Wang T, et al. Electroacupuncture alleviates cartilage degradation: improvement in cartilage biomechanics via pain relief and potentiation of muscle function in a rabbit model of knee osteoarthritis. Biomed Pharmacother 2020; 123: 109724.
DOI URL |
| 28. |
Takagi S, Omori G, Koga H, et al. Quadriceps muscle weakness is related to increased risk of radiographic knee OA but not its progression in both women and men: the Matsudai knee osteoarthritis survey. Knee Surg Sports Traumatol Arthrosc 2018; 26: 2607-14.
DOI |
| 29. |
Stewart HL, Kawcak CE. The importance of subchondral bone in the pathophysiology of osteoarthritis. Front Vet Sci 2018; 5: 178.
DOI PMID |
| 30. |
Norrdin RW, Kawcak CE, Capwell BA, McIlwraith CW. Subchondral bone failure in an equine model of overload arthrosis. Bone 1998; 22: 133-9.
PMID |
| 31. | McIlwraith CW, Frisbie DD, Kawcak CE, Weeren RV. Joint disease in the horse. Saunders: Saunders/Elsevier, 2016: 408. |
| 32. |
Kawcak CE, McIlwraith CW, Norrdin RW, Park RD, James SP. The role of subchondral bone in joint disease: a review. Equine Vet J 2001; 33: 120-6.
PMID |
| 33. |
Cabahug-Zuckerman P, Frikha-Benayed D, Majeska RJ, et al. Osteocyte apoptosis caused by hindlimb unloading is required to trigger osteocyte RANKL production and subsequent resorption of cortical and trabecular bone in mice femurs. J Bone Miner Res 2016; 31: 1356-65.
DOI PMID |
| 34. |
Li B, Aspden RM. Mechanical and material properties of the subchondral bone plate from the femoral head of patients with osteoarthritis or osteoporosis. Ann Rheum Dis 1997; 56: 247-54.
PMID |
| 35. |
Hernandez CJ, Lambers FM, Widjaja J, Chapa C, Rimnac CM. Quantitative relationships between microdamage and cancellous bone strength and stiffness. Bone 2014; 66: 205-13.
DOI PMID |
| 36. |
He Z, Chu L, Liu X, et al. Differences in subchondral trabecular bone microstructure and finite element analysis-based biomechanical properties between osteoporosis and osteoarthritis. J Orthop Translat 2020; 24: 39-45.
DOI URL |
| 37. |
Lampropoulou-Adamidou K, Dontas I, Stathopoulos IP, et al. Chondroprotective effect of high-dose zoledronic acid: an experimental study in a rabbit model of osteoarthritis. J Orthop Res 2014; 32: 1646-51.
DOI PMID |
| 38. |
Miao X, Yuan J, Wu J, et al. Bone morphogenetic protein-2 promotes osteoclasts-mediated osteolysis via Smad1 and p65 signaling pathways. Spine (Phila Pa 1976) 2021; 46: E234-42.
DOI URL |
| 39. |
Tao ZS, Zhou WS, Xu HG, Yang M. Aspirin modified strontium-doped beta-tricalcium phosphate can accelerate the healing of femoral metaphyseal defects in ovariectomized rats. Biomed Pharmacother 2020; 132: 110911.
DOI PMID |
| 40. |
Blaney DE, Vitters EL, Lent PL, Loo FA, Berg WB, Kraan PM. Elevated extracellular matrix production and degradation upon bone morphogenetic protein-2 (BMP-2) stimulation point toward a role for BMP-2 in cartilage repair and remodeling. Arthritis Res Ther 2007; 9: R102.
DOI PMID |
| 41. |
Simic P, Culej JB, Orlic I, et al. Systemically administered bone morphogenetic protein-6 restores bone in aged ovariectomized rats by increasing bone formation and suppressing bone resorption. J Biol Chem 2006; 281: 25509-21.
DOI PMID |
| 42. | Betz VM, Keller A, Foehr P, et al. BMP-2 gene activated muscle tissue fragments for osteochondral defect regeneration in the rabbit knee. J Gene Med 2017; 19: 9-10. |
| 43. | An X, Wang T, Zhang W, et al. Chondroprotective effects of combination therapy of acupotomy and human adipose mesenchymal stem cells in knee osteoarthritis rabbits via the GSK3β-Cyclin D1-CDK4/CDK6 signaling pathway. Aging Dis 2020; 11: 1116-32. |
| 44. |
Qin LX, Guo CQ, Zhao RL, et al. Acupotomy inhibits aberrant formation of subchondral bone through regulating osteoprotegerin/receptor activator of nuclear factor-κB ligand pathway in rabbits with knee osteoarthritis induced by modified Videman method. J Tradit Chin Med 2022; 42: 389-99.
DOI PMID |
| 45. |
Fernandez-Martin S, Lopez-Pena M, Munoz F, Permuy M, Gonzalez-Cantalapiedra A. Bisphosphonates as disease-modifying drugs in osteoarthritis preclinical studies: a systematic review from 2000 to 2020. Arthritis Res Ther 2021; 23: 60.
DOI |
| 46. |
McGrory B, Weber K, Lynott JA, et al. The American Academy of orthopaedic surgeons evidence-based clinical practice guideline on surgical management of osteoarthritis of the knee. J Bone Joint Surg Am 2016; 98: 688-92.
DOI PMID |
| 47. |
Tan Q, Cai Z, Li J, et al. Imaging study on acupuncture inhibiting inflammation and bone destruction in knee osteoarthritis induced by monosodium iodoacetate in rat model. J Pain Res 2022; 15: 93-103.
DOI PMID |
| [1] | YANG Yi, YE Huijun, ZHENG Huiling, JIN Lihua. Clinical observation on 90 cases of primary dysmenorrhea treated by buccal acupuncture therapy: a randomized controlled study [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 172-181. |
| [2] | CHEN Ying, SUN Jingqing, LYU Tianli, HONG Jiahui, LIU Yuhan, ZHU Liying, LI Bin, LIU Lu. Effect of acupuncture treatment on nonketotic hyperglycemic hemichorea-hemiballismus: a case report [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 829-833. |
| [3] | QIN Luxue, GUO Changqing, ZHAO Ruili, WANG Tong, WANG Junmei, GUO Yan, ZHANG Wei, HU Tingyao, CHEN Xilin, ZHANG Qian, ZHANG Dian, XU Yue. Acupotomy inhibits aberrant formation of subchondral bone through regulating osteoprotegerin/receptor activator of nuclear factor-κB ligand pathway in rabbits with knee osteoarthritis induced by modified Videman method [J]. Journal of Traditional Chinese Medicine, 2022, 42(3): 389-399. |
| [4] | Fang Ting, Li Qi, Zhou Fanyuan, Liu Fushui, Liu Zhongyong, Zhao Meimei, Chen Mei, You Jianyu, Jin Yuli, Xie Jinmei. Effect and safety of acupotomy in treatment of knee osteoarthritis:a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2020, 40(3): 355-364. |
| [5] | Zhang Wei, Gao Yang, Guo Changqing, Ibrahim Zeyad Ali Khattab, Farid Mokhtari. Effect of acupotomy versus electroacupuncture on ethology and morphology in a rabbit model of knee osteoarthritis [J]. Journal of Traditional Chinese Medicine, 2019, 39(02): 229-236. |
| [6] | Zheng Yu, Lu Luo, Yu Li, Qiaofeng Wu, Shufang Deng, Shouying Lian, Fanrong Liang. Different manual manipulations and electrical parameters exert different therapeutic effects of acupuncture [J]. Journal of Traditional Chinese Medicine, 2014, 34(06): 754-758. |
| [7] | Tao Huang, Lijian Yang, Shuyong Jia, Xiang Mu, Mozheng Wu, Hang Ye, Weizhe Liu, Xinnong Cheng. Capillary blood flow in patients with dysmenorrhea treated with acupuncture [J]. Journal of Traditional Chinese Medicine, 2013, 33(06): 757-760. |
| [8] | Wei He, Yuanyuan Tong, Yingkai Zhao, Li Zhang, Hui Ben, Qingguang Qin, Feng Huang, Peijing Rong. Review of controlled clinical trials on acupuncture versus sham acupuncture in Germany [J]. Journal of Traditional Chinese Medicine, 2013, 33(03): 403-407. |
| [9] | Ji Chen, Yulan Ren, Yong Tang, Zhengjie Li, Fanrong Liang. Acupuncture therapy for angina pectoris:a systematic review [J]. Journal of Traditional Chinese Medicine, 2012, 32(04): 494-501. |
| [10] | Youzhi Sun, Steve An Xue, Zhengyun Zuo. Acupuncture therapy on apoplectic aphasia rehabilitation [J]. Journal of Traditional Chinese Medicine, 2012, 32(03): 314-321. |
| [11] | Xuming Yang, Lingyu Xu, Fei Zhong, Ying Zhu. Data mining-based detection of acupuncture treatment on juvenile myopia [J]. Journal of Traditional Chinese Medicine, 2012, 32(03): 372-376. |
| [12] | Gao, Liang; Chen, Zelin; Tian, Lixin; Li, Zhongzheng; Guo, Yi. Effects of bloodletting puncture at Jing-Well points in distal ends of finger and toe on survival rate and brain edema in cerebral ischemic rats [J]. Journal of Traditional Chinese Medicine, 2012, 32(03): 471-476. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

