Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (6): 932-939.DOI: 10.19852/j.cnki.jtcm.20220815.004
• Research Articles • Previous Articles Next Articles
Shikonin alleviates collagen-induced arthritis mice by inhibiting M1 macrophage polarization
HE Lianhua1, LUAN Huijie2, QIN Qingxia2, HE Juan2, CHEN Jian2, HU Yiping2, CAI Yueming2, SUN Desheng3, SHI Yu3( ), WANG Qingwen2(
), WANG Qingwen2( )
)
- 1 Department of Rheumatism and Immunology, Peking University Shenzhen Hospital, Shenzhen 518036, China; Shenzhen China Resources Sanjiu Pharmaceutical Trading Co., Ltd., Shenzhen 518029, China
2 Department of Rheumatism and Immunology, Peking University Shenzhen Hospital, Shenzhen 518036, China. The Key Laboratory of Inflammatory and Immunology Diseases, Shenzhen 518036, China
3 Department of ultrasound, Peking University Shenzhen Hospital, Shenzhen 518036, China
-
Received:2021-11-13Accepted:2022-01-21Online:2022-12-15Published:2022-08-15 -
Contact:SHI Yu,WANG Qingwen -
About author:Prof. WANG Qingwen, Department of Rheumatism and Immunology, Peking University Shenzhen Hospital, Shenzhen 518036, China. wqw_sw@163.com,Telephone: +86-13688802429; +86-13823578405
SHI Yu, Department of Rheumatism and Immunology, Peking University Shenzhen Hospital, Shenzhen 518036, China. shiyu@pkuszh.com;
-
Supported by:Role and Mechanism of Ermiao San in the Regulation of Th cell Differentiation by PKC-ɵ in the Treatment of RA(20211333);Role and Mechanism of CD4+T Cell Smad7 Loss in the Pathogenesis of Rheumatoid Arthritis(81974253);Significance of Regulating Glycolytic Metabolism by HK-2 in Dendritic Cell Migration and RA Pathogenesis(81901641);Research on Key Technologies of Intelligent Robot Based on Artificial Intelligence Image Data(GJHZ20200731095208025);Virtual Reality Clinical Application Public Service Platform(XMHT20190104001);Biological Markers in Idiopathic Inflammatory Myopathy and Clinical Significance of Tumorigenesis(SZXJ2017046);Experimental Study on Treatment of Rabbit Rheumatoid Arthritis with Lipidated Methotrexate Microbubbles Combined with Controlled Release by Ultrasonic Irradiation(2018A030313281)
Cite this article
HE Lianhua, LUAN Huijie, QIN Qingxia, HE Juan, CHEN Jian, HU Yiping, CAI Yueming, SUN Desheng, SHI Yu, WANG Qingwen. Shikonin alleviates collagen-induced arthritis mice by inhibiting M1 macrophage polarization[J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 932-939.
share this article

Figure 1 SKN inhibits M1 macrophage polarization in the joints of CIA mice (via immunofluorescence, ×40) A, C: photomicrographs of iNOS+ M1 macrophage immunofluorescence stained synovial membrane tissues in knee joints of control, CIA, 4 mg/kg SKN-treated, and 0.5 mg/kg MTX-treated CIA mice are shown, respectively;the timing of the dosage for 23 d from day 21 after primary immunization. A1-A3: the F4/80+ cells, iNOS+ cells, iNOS+ F4/80+ cells in control group; A4-A5: the F4/80+ cells, iNOS+ cells, iNOS+ F4/80+ cells in CIA group; A6-A9: the F4/80+ cells, iNOS+ cells, iNOS+ F4/80+ cells in SKN group; A10-A12: the the F4/80+ cells, iNOS+ cells, iNOS+ F4/80+ cells in MTX group; B, C: photomicrographs of CD68+ M1 macrophage immunofluorescence-stained ankle joints of control, CIA, SKN-treated, and MTX-treated CIA mice are shown, respectively. B1-B3: the F4/80+ cells, CD68+ cells, CD68+ F4/80+ cells in control group; B4-B5: the F4/80+ cells, CD68+ cells, CD68+ F4/80+ cells in CIA group; B6-B9: the F4/80+ cells, CD68+ cells, CD68+ F4/80+ cells in SKN group; B10-B12: the the F4/80+ cells, CD68+ cells, CD68+ F4/80+ cells in MTX group. All data are present as mean ± standard error of mean (n = 6). aP < 0.001, compared to the control group; bP < 0.01, and cP < 0.05, compared to the CIA group. SKN: shikonin; CIA: collagen-induced arthritis; iNOS: nitric oxide synthase; MTX: methotrexate.

Figure 2 Cell morphology of BMDM A: the morphology of BMDMs at 10 × magnification; B: the morphology of BMDMs at 20× magnification; C: the morphology of BMDMs at 40× magnification; D: DAPI in BMDMs via immunofluorescence; E: F4/80 in BMDMs via immunofluorescence; F: the positive rate of F4/80 in BMDMs via immunofluorescence. BMDM: bone marrow-derived-macrophages; DAPI: diphenylindole.

Figure 3 SKN inhibits M1 polarization induced by LPS and IFN-γ in bone marrow-derived macrophages (via immunofluorescence, ×20) BMDMs were pre-treated with LPS 10 μg/L + IFN-γ 20 μg/L or IL-4 20 μg/L + IL-13 20 μg/L for 1 h, and incubated with or without different concentrations of SKN (0.05, 0.1, and 0.2 μM) for 24 h. A, C: effect of SKN on polarization of iNOS+M1 induced by LPS and IFN-γ.A1-A3: the F4/80+ cells, iNOS+ cells, the iNOS+F4/80+ cells in BMDMs; A4-A6: the F4/80+ cells, iNOS+ cells, the iNOS+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs; A7-A9: the F4/80+ cells, iNOS+ cells, the iNOS+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs with SKN 0.05 μM; A10-A12: the F4/80+ cells, iNOS+ cells, the iNOS+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs with SKN 0.1 μM; A13-A15: the F4/80+ cells, iNOS+ cells, the iNOS+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs with SKN 0.05 μm. ; B, C: effect of SKN on polarization of CD68+M1 induced by LPS and IFN-γ. B1-B3: the F4/80+ cells, CD68+ cells, CD68+F4/80+ cells in BMDMs; B4-B6: the F4/80+ cells, CD68+ cells, CD68+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs; B6-B9: the F4/80+ cells, CD68+ cells, CD68+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs with SKN 0.05 μM; B10-B12: the F4/80+ cells, CD68+ cells, CD68+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs with SKN 0.1 μM; B13-B15: the F4/80+ cells, CD68+ cells, CD68+F4/80+ cells in LPS 10 μg/L + IFN-γ 20 μg/L induced BMDMs with SKN 0.2 μm. All data are expressed as mean ± standard error of mean (n = 12). aP < 0.001, compared with the control group. bP < 0.05, cP < 0.01, and dP < 0.001, compared to the vehicle group. SKN: shikonin; iNOS: nitric oxide synthase; LPS: lipopolysaccharide; IFN: interferon; BMDMs: bone marrow-derived-macrophages.
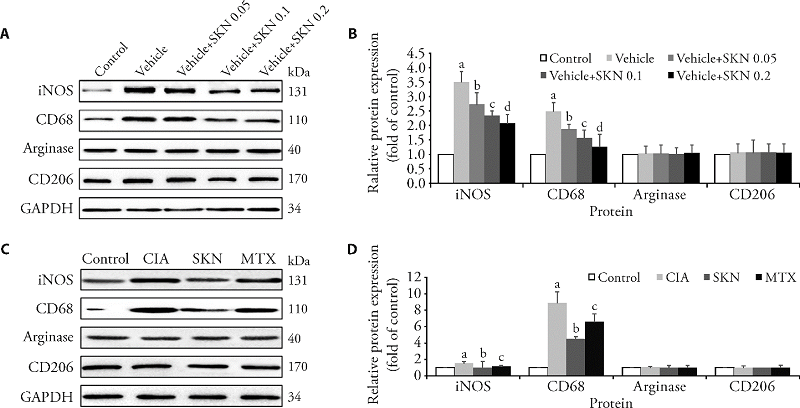
Figure 4 SKN inhibits M1 polarization in CIA mice and LPS + IFN-γ- or IL4 + IL13-induced BMDMs The mice were divided into four groups: the control group (Control), CIA model group (CIA), CIA mice treated with 4 mg/kg SKN group, and CIA mice treated with 0.5 mg/kg MTX group. The timing of the dosage for 23 d from day 21 after primary immunization. BMDMs were pre-treated with LPS 10 μg/L + IFN-γ 20 μg/L or IL-4 20 μg/L + IL-13 20 μg/L for 1 h, and incubated with or without different concentrations of SKN (0.05, 0.1, and 0.2 μM) for 24 h. SKN significantly inhibited the expression of iNOS and CD68, but not arginase and CD206 in LPS + IFN-γ- or IL4 + IL13-induced BMDMs (A and B) (aP < 0.001, compared with the control group. bP < 0.05, cP < 0.01, and dP < 0.001, compared to the vehicle group). Additionally, after SKN and MTX treatment, the expression levels of iNOS and CD68, rather than arginase and CD206, were significantly decreased (C and D). All of the experiments were performed in triplicate. Mean ± standard error of mean was calculated for independent experiments (aP < 0.001, compared to the control group; bP < 0.01, and cP < 0.05, compared to the CIA group.). SKN: shikonin; iNOS: nitric oxide synthase; LPS: lipopolysaccharide; IFN: interferon; IL: interleukin; BMDMs: bone marrow-derived-macrophages; MTX: methotrexate.
| [1] |
Andújar I, Ríos JL, Giner RM, Recio MC. Pharmacological properties of shikonin - a review of literature since 2002. Planta Med 2013; 79: 1685-97.
DOI URL |
| [2] |
Wang F, Yao X, Zhang Y, Tang J. Synthesis, biological function and evaluation of shikonin in cancer therapy. Fitoterapia 2019; 134: 329-39.
DOI PMID |
| [3] |
Dai Q, Fang J, Zhang FS. Dual role of shikonin in early and late stages of collagen type II arthritis. Mol Biol Rep 2009; 36: 1597-604.
DOI PMID |
| [4] | Lee CC, Wang CN, Lai YT, et al. Shikonin inhibits maturation of bone marrow-derived dendritic cells and suppresses allergic airway inflammation in a murine model of asthma. Br J Pharmacol 2010; 161: 1496-511. |
| [5] |
Kim YO, Hong SJ, Yim SV. The efficacy of shikonin on cartilage protection in a mouse model of rheumatoid arthritis. Korean J Physiol Pharmacol 2010; 14: 199-204.
DOI URL |
| [6] |
Tardito S, Martinelli G, Soldano S, et al. Macrophage M1/M2 polarization and rheumatoid arthritis: a systematic review. Autoimmun Rev 2019; 18: 102397.
DOI URL |
| [7] |
Yuan DP, Gu L, Long J, et al. Shikonin reduces endometriosis by inhibiting RANTES secretion and mononuclear macrophage chemotaxis. Exp Ther Med 2014; 7: 685-90.
DOI URL |
| [8] |
Koike A, Shibano M, Mori H, Kohama K, Fujimori K, Amano F. Simultaneous addition of shikonin and its derivatives with lipopolysaccharide induces rapid macrophage death. Pharm Bull 2016; 39: 969-76.
DOI URL |
| [9] |
Remmers EF, Joe B, Griffiths MM, et al. Modulation of multiple experimental arthritis models by collagen-induced arthritis quantitative trait loci isolated in congenic rat lines: different effects of non-major histocompatibility complex quantitative trait loci in males and females. Arthritis Rheum 2002; 46: 2225-34.
DOI URL |
| [10] | Prado C, Ugalde V, González H, et al. STAT3 activation in combination with NF-KappaB inhibition induces tolerogenic dendritic cells with high therapeutic potential to attenuate |
| [11] | He LH, Qin QX, He J, et al. ErMiao San inhibits angiogenesis in rheumatoid arthritis by suppressing JAK/STAT signaling path-ways. Evid Based Complement Alternat Med 2020; 2020: 1-12. |
| [12] |
He L, Liu C, Sun C, et al. Wu-Tou Decoction inhibits angiogenesis in experimental arthritis by targeting VEGFR2 signaling pathway. Rejuvenation Res 2018; 21: 442-55.
DOI PMID |
| [13] |
Liu CF, Kong XY, Li XB, et al. Wen Luo Yin inhibits angiogenesis in collagen-induced arthritis rat model and in vitro. J Ethnopharmacol 2013; 149: 478-89.
DOI URL |
| [14] |
Liu CF, He LH, Wang JX, et al. Anti-angiogenic effect of shikonin in rheumatoid arthritis by downregulating PI3K/AKT and MAPKs signaling pathways. J Ethnopharmacol 2020; 260: 113039.
DOI URL |
| [15] |
He LH, Luan HJ, He J, et al. Shikonin attenuates rheumatoid arthritis by targeting SOCS1/JAK/STAT signaling pathway of fibroblast like synoviocytes. Chin Med 2021; 16: 96.
DOI URL |
| [16] | Ying W, Cheruku PS, Bazer FW, Safe SH, Zhou B. Investigation of macrophage polarization using bone marrow derived macrophages. J Vis Exp 2013; 50323. |
| [17] |
Burke B, Giannoudis A, Corke KP, et al. Hypoxia-induced gene ex-pression in human macrophages: implications for ischemic tissues and hypoxia-regulated gene therapy. Am J Pathol 2003; 163: 1233-43.
DOI URL |
| [18] | Viatte S, Plant D, Raychaudhuri S. Genetics and epigenetics of rheumatoid arthritis. Nature reviews. Rheumatol 2013; 9: 141-53. |
| [19] |
McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med 2011; 365: 2205-19.
DOI URL |
| [20] |
Alvaro-Gracia JM, Zvaifler NJ, Firestein GS. Cytokines in chronic inflammatory arthritis. V. Mutual antagonism between interferon-gamma and tumor necrosis factor-alpha on HLA-DR expression, proliferation, collagenase production, and granu-locyte macro-phage colony-stimulating factor production by rh-eumatoid arthritis synoviocytes. J Clin Invest 1990; 86: 1790-8.
PMID |
| [21] |
Athanasou NA, Quinn J, Heryet A, Puddle B, Woods CG, McGee JO. The immunohistology of synovial lining cells in normal and inflamed synovium. J Pathol 1988; 155: 133-42.
PMID |
| [22] |
Bondeson J, Blom AB, Wainwright S, Hughes C, Caterson B, van den Berg WB. The role of synovial macrophages and macrophage-produced mediators in driving inflammatory and destructive responses in osteoarthritis. Arthritis Rheum 2010; 62: 647-57.
DOI URL |
| [23] |
Danning CL, Illei GG, Hitchon C, Greer MR, Boumpas DT, McInnes IB. Macrophage-derived cytokine and nuclear factor kappaB p65 expression in synovial membrane and skin of patients with psoriatic arthritis. Arthritis Rheum 2000; 43: 1244-56.
DOI URL |
| [24] |
Brennan FM, McInnes IB. Evidence that cytokines play a role in rheumatoid arthritis. J Clin Invest 2008; 118: 3537-45.
DOI PMID |
| [25] |
Feldmann M, Brennan FM, Maini RN. Role of cytokines in rheumatoid arthritis. Annu Rev Immunol 1996; 14: 397-440.
PMID |
| [26] |
Kishimoto T. Interleukin-6: from basic science to medicine-40 years in immunology. Annu Rev Immunol 2005; 23: 1-21.
DOI URL |
| [27] |
Choy EH, Panayi GS. Cytokine pathways and joint inflammation in rheumatoid arthritis. N Engl J Med 2001; 344: 907-16.
DOI URL |
| [28] |
Bresnihan B, Gerlag DM, Rooney T, et al. Synovial macrophages as a biomarker of response to therapeutic intervention in rheumatoid arthritis: standardization and consistency across centers. J Rheumatol 2007; 34: 620-2.
PMID |
| [29] |
Mulherin D, Fitzgerald O, Bresnihan B. Synovial tissue macrophage populations and articular damage in rheumatoid arthritis. Arthritis Rheum 1996; 39: 115-24.
DOI URL |
| [30] |
Haringman JJ, Gerlag DM, Zwinderman AH, et al. Synovial tissue macrophages: a sensitive biomarker for response to treatment in pa-tients with rheumatoid arthritis. Ann Rheum Dis 2005; 64: 834-8.
PMID |
| [31] |
Gordon S. The macrophage: past, present and future. Eur J Immunol 2007; 37 Suppl 1: S9-17.
DOI URL |
| [32] | Falconer J, Murphy AN, Young SP, et al. Review: synovial cell metabolism and chronic inflammation in rheumatoid arthritis. Arthritis Rheum 2018; 70: 984-99. |
| [33] |
Mantovani A, Sica A, Sozzani S, AllavenaP, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol 2004; 25: 677-86.
DOI PMID |
| [34] | Hamilton JA. Colony-stimulating factors in inflammation and autoimmunity. Nature reviews. Immunology 2008; 8: 533-44. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

