Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (3): 554-563.DOI: 10.19852/j.cnki.jtcm.20240408.001
• Original articles • Previous Articles Next Articles
Proteomics analysis of coronary atherosclerotic heart disease with different Traditional Chinese Medicine syndrome types before and after percutaneous coronary intervention
WANG Zhibo1, LI Ying1, WANG Daoping2, MA Bo1, MIAO Lan1, REN Junguo1, LIU Jinghua3( ), LIU Jianxun1(
), LIU Jianxun1( )
)
- 1 Beijing Key Laboratory of Pharmacology of Chinese Materia Region, Institute of Basic Medical Sciences, Xiyuan Hospital, China Academy of Chinese Medical Sciences, National Clinical Research Center of Cardiovascular Disease of Traditional Chinese Medicine, Beijing 100000, China
2 the National Key Facility for Crop Gene Resources and Genetic Improvement, Institute of Crop Science, Chinese Academy of Agricultural Science, Beijing 100098, China
3 Department of Cardiology, Beijing Anzhen Hospital, Capital Medical University, Beijing 100029, China
-
Received:2023-01-12Accepted:2023-05-25Online:2024-06-15Published:2024-04-08 -
Contact:LIU Jianxun, Beijing Key Laboratory of Pharmacology of Chinese Materia Region, Institute of Basic Medical Sciences, Xiyuan Hospital, China Academy of Chinese Medical Sciences, National Clinical Research Center of Cardiovascular Disease of Traditional Chinese Medicine, Beijing 100000, China.liujx0324@sina.com ;LIU Jinghua, Department of Cardiology, Beijing Anzhen Hospital, Capital Medical University, Beijing 100029, China. liujinghua@vip.sina.com Telephone: +86-18810446985 -
Supported by:National Natural Science Foundation of China: Biological Mechanism of Platelet System in the Occurrence and Evolution of Blood Stasis Syndrome(82030124)
Cite this article
WANG Zhibo, LI Ying, WANG Daoping, MA Bo, MIAO Lan, REN Junguo, LIU Jinghua, LIU Jianxun. Proteomics analysis of coronary atherosclerotic heart disease with different Traditional Chinese Medicine syndrome types before and after percutaneous coronary intervention[J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 554-563.
share this article
| Item | QD (n = 211) | QS (n = 227) |
|---|---|---|
| Age (years)a | 60.3±9.0 | 60.8±9.1 |
| Males [n (%)] | 149 (70.6) | 183 (80.6) |
| Body mass index (kg/m2)a | 24.9±3.2 | 25.0±2.9 |
| Systolic pressure (mm Hg)a | 130.2±20.4 | 131.0±20.9 |
| Diastolic pressure (mm Hg)a | 74.9±12.3 | 75.8±12.8 |
| Resting heart rate (bpm)a | 72.0±11.0 | 72.0±13.0 |
| Unstable angina [n (%)] | 123 (58.3) | 151 (66.5) |
| Non-ST-segment elevation myocardial infarction [n (%)] | 32 (15.2) | 24 (10.6) |
| ST-segment elevation myocardial infarction [n (%)] | 56 (26.5) | 52 (22.9) |
| History of hypertension [n (%)] | 127 (60.2) | 136 (59.9) |
| History of hyperlipidemia [n (%)] | 59 (28.0) | 81 (35.7) |
| History of diabetes [n (%)] | 67 (31.8) | 74 (32.6) |
| History of stroke [n (%)] | 15 (7.1) | 23 (10.1) |
| History of smoking [n (%)] | 99 (46.9) | 116 (51.1) |
| Ejection fraction [n (%)] | 62.2±9.2 | 62.5±7.4 |
| Left ventricular internal dimension in diastole (mm)a | 47.7±5.2 | 47.8±4.8 |
| Erythrocyte (×1012/L)b | 4.6 (4.3-4.9) | 4.6 (4.3-5.0) |
| Hemoglobin (g/L)a | 139.3±16.0 | 141.1±15.7 |
| White blood cell (×109/L)a | 7.6±2.7 | 7.4±2.7 |
| Percentages of mature neutrophils (%)b | 63.7 (55.6-71.7) | 63.1 (57.0-70.8) |
| Platelet count (×109/L)a | 216.4±55.7 | 210.9±60.7 |
| Uric acid (μmol/L)a | 347.8±98.2 | 355.2±96.3 |
| Total cholesterol (mmol/L)a | 4.3±1.1 | 4.1±1.1 |
| Triglycerides (mmol/L)b | 1.4 (1.0-2.1) | 1.39 (1.0-2.0) |
| Prothrombin time (s)b | 11.3 (10.6-12.5) | 11.3 (10.6-12.5) |
| Activated partial thromboplastin time (s)a | 34.7±15.9 | 34.1±18.2 |
| D-dimer (ng/mL)a | 101.6±54.3 | 102.4±36.6 |
| Fibrinogen (μg/mL)b | 3.2 (2.8-3.7) | 3.1 (2.6-3.7) |
Table 1 Baseline characteristics of the study participants
| Item | QD (n = 211) | QS (n = 227) |
|---|---|---|
| Age (years)a | 60.3±9.0 | 60.8±9.1 |
| Males [n (%)] | 149 (70.6) | 183 (80.6) |
| Body mass index (kg/m2)a | 24.9±3.2 | 25.0±2.9 |
| Systolic pressure (mm Hg)a | 130.2±20.4 | 131.0±20.9 |
| Diastolic pressure (mm Hg)a | 74.9±12.3 | 75.8±12.8 |
| Resting heart rate (bpm)a | 72.0±11.0 | 72.0±13.0 |
| Unstable angina [n (%)] | 123 (58.3) | 151 (66.5) |
| Non-ST-segment elevation myocardial infarction [n (%)] | 32 (15.2) | 24 (10.6) |
| ST-segment elevation myocardial infarction [n (%)] | 56 (26.5) | 52 (22.9) |
| History of hypertension [n (%)] | 127 (60.2) | 136 (59.9) |
| History of hyperlipidemia [n (%)] | 59 (28.0) | 81 (35.7) |
| History of diabetes [n (%)] | 67 (31.8) | 74 (32.6) |
| History of stroke [n (%)] | 15 (7.1) | 23 (10.1) |
| History of smoking [n (%)] | 99 (46.9) | 116 (51.1) |
| Ejection fraction [n (%)] | 62.2±9.2 | 62.5±7.4 |
| Left ventricular internal dimension in diastole (mm)a | 47.7±5.2 | 47.8±4.8 |
| Erythrocyte (×1012/L)b | 4.6 (4.3-4.9) | 4.6 (4.3-5.0) |
| Hemoglobin (g/L)a | 139.3±16.0 | 141.1±15.7 |
| White blood cell (×109/L)a | 7.6±2.7 | 7.4±2.7 |
| Percentages of mature neutrophils (%)b | 63.7 (55.6-71.7) | 63.1 (57.0-70.8) |
| Platelet count (×109/L)a | 216.4±55.7 | 210.9±60.7 |
| Uric acid (μmol/L)a | 347.8±98.2 | 355.2±96.3 |
| Total cholesterol (mmol/L)a | 4.3±1.1 | 4.1±1.1 |
| Triglycerides (mmol/L)b | 1.4 (1.0-2.1) | 1.39 (1.0-2.0) |
| Prothrombin time (s)b | 11.3 (10.6-12.5) | 11.3 (10.6-12.5) |
| Activated partial thromboplastin time (s)a | 34.7±15.9 | 34.1±18.2 |
| D-dimer (ng/mL)a | 101.6±54.3 | 102.4±36.6 |
| Fibrinogen (μg/mL)b | 3.2 (2.8-3.7) | 3.1 (2.6-3.7) |
| Variable | QSA | QSB | P value | QDA | QDB | P value |
|---|---|---|---|---|---|---|
| TNF-α | 4.98±3.08a | 6.56±3.07 | <0.01 | 5.14±2.91c | 7.32±4.34 | <0.01 |
| AST | 25.25±11.54 | 28.67±15.01 | 0.06 | 23.59±7.37c | 28.56±13.74 | <0.01 |
| HDL-C | 1.21±0.24a | 1.04±0.26 | <0.01 | 1.17±0.31c | 1.02±0.27 | <0.01 |
| BSCSO | 18.03±5.80a | 32.27±12.95 | <0.01 | 17.83±6.32c | 33.53±13.94 | <0.01 |
| Gensini | 20.12±23.47a | 44.60±31.48 | <0.01 | 21.44±24.17c | 53.51±36.39 | <0.01 |
| LDL-C | 2.13±0.64b | 2.32±0.75 | <0.05 | 2.25±0.76c | 2.55±1.07 | <0.01 |
Table 2 Disease indicators in patients with two syndrome types ($\bar{x} \pm s$)
| Variable | QSA | QSB | P value | QDA | QDB | P value |
|---|---|---|---|---|---|---|
| TNF-α | 4.98±3.08a | 6.56±3.07 | <0.01 | 5.14±2.91c | 7.32±4.34 | <0.01 |
| AST | 25.25±11.54 | 28.67±15.01 | 0.06 | 23.59±7.37c | 28.56±13.74 | <0.01 |
| HDL-C | 1.21±0.24a | 1.04±0.26 | <0.01 | 1.17±0.31c | 1.02±0.27 | <0.01 |
| BSCSO | 18.03±5.80a | 32.27±12.95 | <0.01 | 17.83±6.32c | 33.53±13.94 | <0.01 |
| Gensini | 20.12±23.47a | 44.60±31.48 | <0.01 | 21.44±24.17c | 53.51±36.39 | <0.01 |
| LDL-C | 2.13±0.64b | 2.32±0.75 | <0.05 | 2.25±0.76c | 2.55±1.07 | <0.01 |
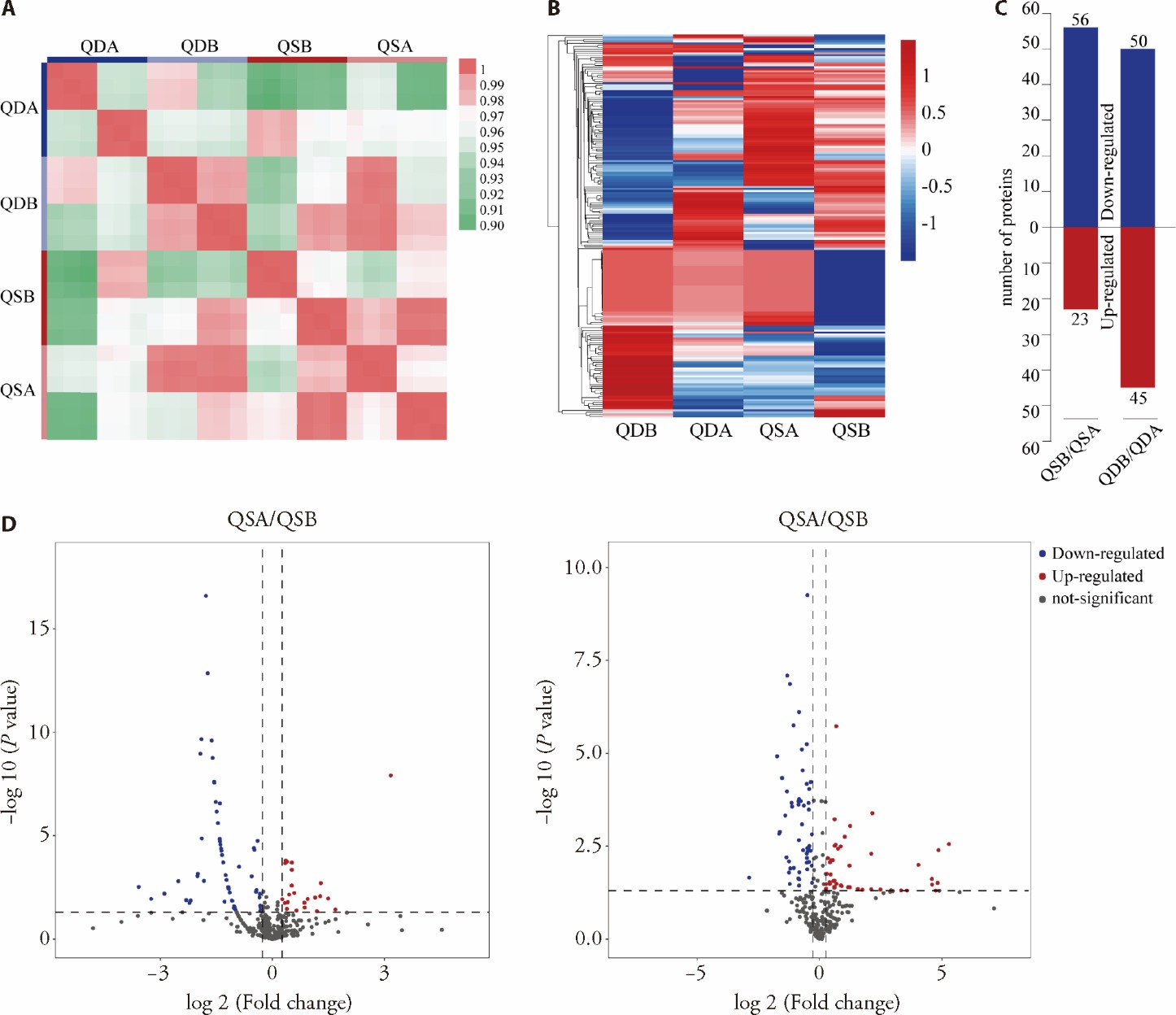
Figure 1 Quality control of the data and integrated analysis of DEPs A: Pearson correlation analysis, the replicates in each group are 6, 3 subjects by each sex; B: enrichment and clustering analyses of DEPs, with quantitative data standardized; C: the count of DEPs between two comparison groups; D: volcano plot illustrating the distribution of proteins in two comparison groups: red points indicate upregulated proteins, blue points denote downregulated proteins, and gray points signify proteins not differentially expressed. QSA: CAD patients with Qi stagnation and blood stasis syndrome post-PCI; QSB: CAD patients with Qi stagnation and blood stasis syndrome pre-PCI; QDA: CAD patients with Qi deficiency and blood stasis syndrome post-PCI; QDB: CAD patients with Qi deficiency and blood stasis syndrome pre-PCI. DEPs: differentially expressed proteins; CAD: coronary artery disease; PCI: percutaneous coronary intervention.
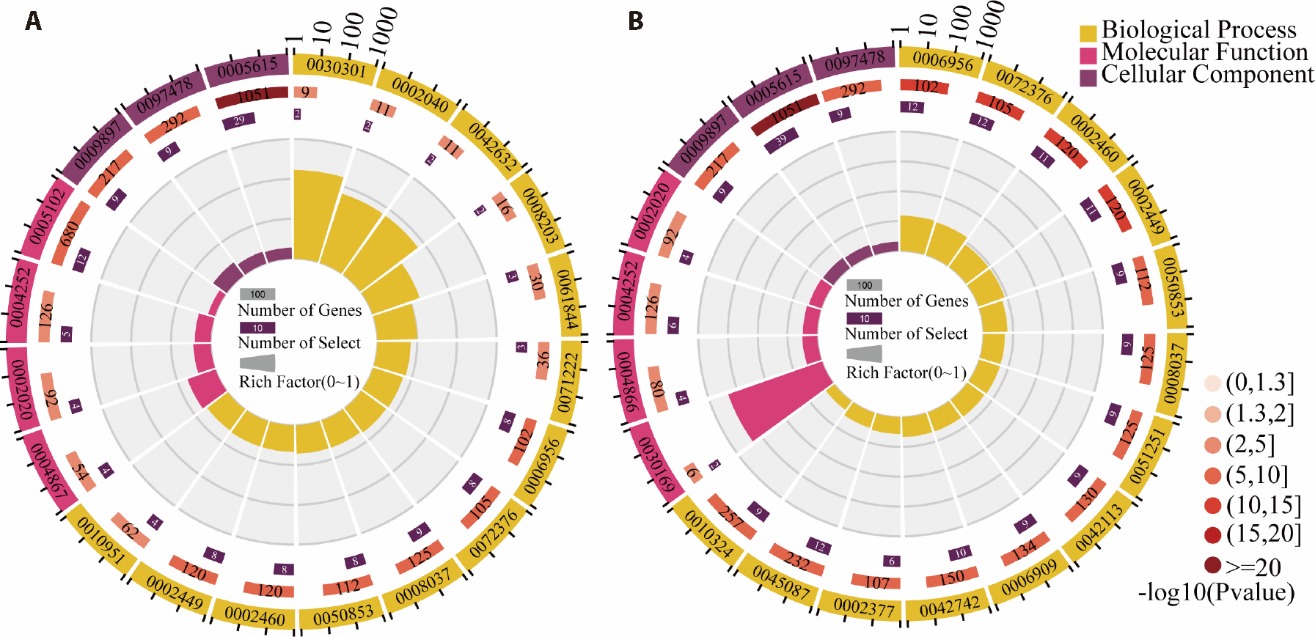
Figure 2 GO annotation enrichment analysis A: functional enrichment maps of GO terms for QSA/QSB; B: functional enrichment maps of GO terms for QDA/QDB. Depicted in four concentric circles: outermost circle displays gene item classification with a coordinate ruler, using different colors for various categories. Second circle shows the count of background genes per entry and corresponding P-values; longer bars indicate more genes, and redder hues denote smaller P-values. Third circle presents the count of input list genes found in each category, without differentiation between up- and down-regulated genes. Innermost circle illustrates the rich factor, with each background gridline representing 0.1. QSA: CAD patients with Qi stagnation and blood stasis syndrome post-PCI; QSB: CAD patients with Qi stagnation and blood stasis syndrome pre-PCI; QDA: CAD patients with Qi deficiency and blood stasis syndrome post-PCI; QDB: CAD patients with Qi deficiency and blood stasis syndrome pre-PCI; GO: Gene Ontology; PCI: percutaneous coronary intervention; CAD: coronary artery disease.

Figure 3 Overview of pathway enrichment and PPI analysis A: Bubble charts for KEGG pathway enrichment analysis in QSA/QSB; B: PPI networks for DEPs in QSA/QSB; C: Bubble charts for KEGG pathway enrichment analysis in QDA/QDB; D: PPI networks for DEPs in QDA/QDB. In the bubble chart, the X-axis indicates the enrichment factor, while bubble size and color denote the count of foreground genes and their statistical significance, respectively. The rich factor indicates the ratio of enriched DEPs to all genes annotated in a pathway, with higher values indicating greater enrichment. GPI: glycosylphosphatidylinositol; PPAR: peroxisome proliferator-activated receptor; ECM: extracellular matrix; QSA: CAD patients with Qi stagnation and blood stasis syndrome post-PCI; QSB: CAD patients with Qi stagnation and blood stasis syndrome pre-PCI; QDA: CAD patients with Qi deficiency and blood stasis syndrome post-PCI; QDB: CAD patients with Qi deficiency and blood stasis syndrome pre-PCI. PPI: protein-protein interaction; KEGG: Kyoto Encyclopedia of Genes and Genomes; DEPs: differentially expressed proteins; CAD: coronary artery disease; PCI: percutaneous coronary intervention.
| 1. |
Guo F, Qiu X, Zhu Y, Tan Z, Li Z, Ouyang D. Association between plasma betaine levels and dysglycemia in patients with coronary artery disease. Biosci Rep 2020; 40: BSR20200676.
DOI URL |
| 2. |
Zhou W, Wang Y. A network-based analysis of the types of coronary artery disease from traditional Chinese medicine perspective: Potential for therapeutics and drug discovery. J Ethnopharmacol 2014; 151: 66-77.
DOI PMID |
| 3. | Ren Y, Zhang M, Chen K, et al. Clinical and epidemiological investigation of TCM syndromes of patients with coronary heart disease in China. Evid-Based Compl Alt 2012; 2012: 714517. |
| 4. |
Wu HP, Lin MJ. Central aortic pressure and long-term outcome in hypertensive patients undergoing percutaneous coronary intervention. Sci Rep 2020; 10: 17420.
DOI |
| 5. |
Lu C, Qv X, Jiang J. Proteomics and syndrome of Chinese medicine. J Cell Mol Med 2010; 14: 2721-8.
DOI URL |
| 6. |
Chan MY, Efthymios M, Tan SH, et al. Prioritizing candidates of post-myocardial infarction heart failure using plasma proteomics and single-cell transcriptomics. Circulation 2020; 142: 1408-21.
DOI URL |
| 7. |
Captur G, Heywood WE, Coats C, et al. Identification of a multiplex biomarker panel for hypertrophic cardiomyopathy using quantitative proteomics and machine learning. Mol Cell Proteomics 2020; 19: 114-27.
DOI PMID |
| 8. |
Xie T, Guo J, Jiang Y, Li L, Jiang L, Wei Y. Screening differentially expressed proteins of coronary heart disease with congenital cold syndrome based on tandem mass tag (TMT) technology. Bioengineered 2021; 12: 1338-50.
DOI PMID |
| 9. |
Delles C, Carrick E, Graham D, Nicklin SA. Utilizing proteomics to understand and define hypertension: where are we and where do we go? Expert Rev Proteomics 2018; 15: 581-92.
DOI URL |
| 10. |
Cox J, Hein MY, Luber CA, Paron I, Nagaraj N, Mann M. Accurate proteome-wide label-free quantification by delayed normalization and maximal peptide ratio extraction, termed MaxLFQ. Mol Cell Proteomics 2014; 13: 2513-26.
DOI PMID |
| 11. |
Danielsen HN, Hansen SH, Herbst F, et al. Direct identification of functional amyloid proteins by label-free quantitative mass spectrometry. Biomolecules 2017; 7: 58.
DOI URL |
| 12. |
Auer J, Berent R, Lassnig E, Eber B. C-reactive protein and coronary artery disease. Jpn Heart J 2002; 43: 607-19.
PMID |
| 13. | Su D, Li Z, Li X, et al. Association between serum interleukin-6 concentration and mortality in patients with coronary artery disease. Mediat Inflamm 2013; 2013: 1-7. |
| 14. | Wei W, Zhang X, Kwong J, Zeng X, Zhang Z, Hua X. Tumor necrosis factor-alpha G-238A polymorphism and coronary artery disease risk: a Meta-analysis of 4222 patients and 4832 controls. Ther Clin Risk Manag 2015; 11: 1429-36. |
| 15. |
Hu K, Wang X, Hu H, et al. Intracoronary application of nicorandil regulates the inflammatory response induced by percutaneous coronary intervention. J Cell Mol Med 2020; 24: 4863-70.
DOI PMID |
| 16. |
Zhou F, Luo Q, Han L, Shen G, Huang L, Ye H. Proteomics reveals urine apolipoprotein A-I as a potential biomarker of acute kidney injury following percutaneous coronary intervention in elderly patients. Exp Ther Med 2021; 22: 745.
DOI PMID |
| 17. | Monu, Kharb R, Sharma A, et al. Plasma proteome profiling of coronary artery disease patients: downregulation of transthyretin-an important event. Mediators Inflamm 2020; 2020: 3429541. |
| 18. | Ma B, Chen J, Mu Y, et al. Proteomic analysis of rat serum revealed the effects of chronic sleep deprivation on metabolic, cardiovascular and nervous system. PLoS One 2018; 13: e199237. |
| 19. |
Ma J, Chen T, Wu S, et al. iProX: an integrated proteome resource. Nucleic Acids Res 2019; 47: D1211-17.
DOI |
| 20. |
Oksjoki R, Kovanen PT, Mayranpaa MI, et al. Complement regulation in human atherosclerotic coronary lesions. Immunohistochemical evidence that C4b-binding protein negatively regulates the classical complement pathway, and that C5b-9 is formed via the alternative complement pathway. Atherosclerosis 2007; 192: 40-8.
DOI PMID |
| 21. |
Nityanand S, Truedsson L, Mustafa A, Bergmark C, Lefvert AK. Circulating immune complexes and complement C4 null alleles in patients in patients operated on for premature atherosclerotic peripheral vascular disease. J Clin Immunol 1999; 19: 406-13.
PMID |
| 22. | Heja D, Kocsis A, Dobo J, et al. Revised mechanism of complement lectin-pathway activation revealing the role of serine protease MASP-1 as the exclusive activator of MASP-2. Proc Natl Acad Sci USA 2012; 109: 10498-503. |
| 23. |
Schwaeble WJ, Lynch NJ, Clark JE, et al. Targeting of mannan-binding lectin-associated serine protease-2 confers protection from myocardial and gastrointestinal ischemia/reperfusion injury. Proc Natl Acad Sci USA 2011; 108: 7523-28.
DOI PMID |
| 24. |
Arbore G, Kemper C, Kolev M. Intracellular complement-the complosome-in immune cell regulation. Mol Immunol 2017; 89: 2-9.
DOI URL |
| 25. |
Walport MJ. Complement. First of two parts. N Engl J Med 2001; 344: 1058-66.
DOI URL |
| 26. |
Rosado CJ, Kondos S, Bull TE, et al. The MACPF/CDC family of pore-forming toxins. Cell Microbiol 2008; 10: 1765-74.
DOI PMID |
| 27. |
Gilbert RJ, Dalla SM, Froelich CJ, Wallace MI, Anderluh G. Membrane pore formation at protein-lipid interfaces. Trends Biochem Sci 2014; 39: 510-16.
DOI PMID |
| 28. |
Podack ER, Tschopp J. Membrane attack by complement. Mol Immunol 1984; 21: 589-603.
PMID |
| 29. |
van Jaarsveld PP, Edelhoch H, Goodman DS, Robbins J. The interaction of human plasma retinol-binding protein and prealbumin. J Biol Chem 1973; 248: 4698-705.
PMID |
| 30. | Monu, Kharb R, Sharma A, et al. Plasma proteome profiling of coronary artery disease patients: downregulation of transthyretin-an important event. Mediators Inflamm 2020; 2020: 3429541. |
| 31. |
Liz MA, Gomes CM, Saraiva MJ, Sousa MM. ApoA-I cleaved by transthyretin has reduced ability to promote cholesterol efflux and increased amyloidogenicity. J Lipid Res 2007; 48: 2385-95.
DOI PMID |
| 32. |
Sousa MM, Berglund L, Saraiva MJ. Transthyretin in high density lipoproteins: association with apolipoprotein A-I. J Lipid Res 2000; 41: 58-65.
PMID |
| 33. | Feingold KR, Anawalt B, Blackman MR, et al. Introduction to lipids and lipoproteins. 2024; In press. |
| 34. |
Wolf D, Gerhardt T, Winkels H, et al. Pathogenic autoimmunity in atherosclerosis evolves from initially protective apolipoprotein B100-reactive CD4+T-regulatory cells. Circulation 2020; 142: 1279-93.
DOI URL |
| 35. | Toth PP, Jones SR, Monsalvo ML, Elliott Davey M, López JAG, Banach M. Effect of evolocumab on non‐high‐density lipoprotein cholesterol, apolipoprotein B, and lipoprotein(a): a pooled analysis of phase 2 and phase 3 studies. J Am Heart Assoc 2020; 9: e014129. |
| 36. | Ference BA, Kastelein JJP, Ray KK, et al. Association of triglyceride-lowering LPL variants and LDL-C-lowering LDLR variants with risk of coronary heart disease. JAMA 2019; 321: 64. |
| 37. |
McLean JW, Tomlinson JE, Kuang WJ, et al. cDNA sequence of human apolipoprotein (a) is homologous to plasminogen. Nature 1987; 330: 132-7.
DOI |
| 38. |
Tirziu D, Moodie KL, Zhuang ZW, et al. Delayed arteriogenesis in hypercholesterolemic mice. Circulation 2005; 112: 2501-09.
PMID |
| 39. |
Vasudev NS, Reynolds AR. Anti-angiogenic therapy for cancer: current progress, unresolved questions and future directions. Angiogenesis 2014; 17: 471-94.
DOI PMID |
| 40. |
Ostos MA, Conconi M, Vergnes L, et al. Antioxidative and antiatherosclerotic effects of human apolipoprotein A-IV in apolipoprotein E-deficient mice. Arterioscl Throm Vas 2001; 21: 1023-28.
DOI URL |
| 41. | Duverger N, Tremp G, Caillaud JM, et al. Protection against atherogenesis in mice mediated by human apolipoprotein A-IV. Science (AAAS) 1996; 273: 966-8. |
| 42. |
Cohen RD, Castellani LW, Qiao JH, Van Lenten BJ, Lusis AJ, Reue K. Reduced aortic lesions and elevated high density lipoprotein levels in transgenic mice overexpressing mouse apolipoprotein A-IV. J Clin Invest 1997; 99: 1906-16.
PMID |
| 43. | Xu XR, Wang Y, Adili R, et al. Apolipoprotein A-IV binds αIIbβ3 integrin and inhibits thrombosis. Nat Commun 2018; 9: 3608. |
| 44. | Qu J, Ko CW, Tso P, Bhargava A. Apolipoprotein A-IV: A multifunctional protein involved in protection against atherosclerosis and diabetes. Cells-Basel 2019; 8: 319. |
| 45. |
McBride PE. Triglycerides and risk for coronary heart disease. JAMA 2007; 298: 336-8.
DOI PMID |
| 46. |
Kannel WB, Vasan RS. Triglycerides as vascular risk factors: new epidemiologic insights. Curr Opin Cardiol 2009; 24: 345-50.
DOI PMID |
| 47. | Qamar A, Khetarpal SA, Khera AV, Qasim A, Rader DJ, Reilly MP. Plasma apolipoprotein C-Ⅲ levels, triglycerides, and coronary artery calcification in type 2 diabetics. Arterioscl Throm Vas 2015; 35: 1880-8. |
| 48. |
Folsom AR, Aleksic N, Park E, Salomaa V, Juneja H, Wu KK. Prospective study of fibrinolytic factors and incident coronary heart disease: the atherosclerosis risk in communities (ARIC) study. Arterioscl Throm Vas 2001; 21: 611-7.
DOI URL |
| 49. |
Sakkinen PA, Cushman M, Psaty BM, et al. Relationship of plasmin generation to cardiovascular disease risk factors in elderly men and women. Arterioscl Throm Vas 1999; 19: 499-504.
DOI PMID |
| 50. |
Plow EF, Hoover-Plow J. The functions of plasminogen in cardiovascular disease. Trends Cardiovasc Med 2004; 14: 180-6.
DOI URL |
| 51. |
Syrovets T, Simmet T. Novel aspects and new roles for the serine protease plasmin. Cell Mol Life Sci 2004; 61: 873-85.
DOI URL |
| 52. |
Ploplis VA, French EL, Carmeliet P, Collen D, Plow EF. Plasminogen deficiency differentially affects recruitment of inflammatory cell populations in mice. Blood 1998; 91: 2005-9.
PMID |
| 53. |
Torzewski M, Suriyaphol P, Paprotka K, et al. Enzymatic modification of low-density lipoprotein in the arterial wall: a new role for plasmin and matrix metalloproteinases in atherogenesis. Arterioscl Throm Vas 2004; 24: 2130-6.
DOI URL |
| 54. |
Syrovets T, Simmet T. Novel aspects and new roles for the serine protease plasmin. Cell Mol Life Sci 2004; 61: 873-85.
PMID |
| 55. |
Touyz RM, Schiffrin EL. Peroxisome proliferator-activated receptors in vascular biology-molecular mechanisms and clinical implications. Vasc Pharmacol 2006; 45: 19-28.
DOI URL |
| 56. |
Castro A, Lazaro I, Selva DM, et al. APOH is increased in the plasma and liver of type 2 diabetic patients with metabolic syndrome. Atherosclerosis 2010; 209: 201-5.
DOI PMID |
| 57. |
Corona JC, Duchen MR. PPAR gamma as a therapeutic target to rescue mitochondrial function in neurological disease. Free Radic Biol Med 2016; 100: 153-63.
DOI URL |
| 58. |
Budoff MJ, Shaw LJ, Liu ST, et al. Long-term prognosis associated with coronary calcification: observations from a registry of 25 253 patients. J Am Coll Cardiol 2007; 49: 1860-70.
DOI URL |
| 59. |
Blaha M, Budoff MJ, Shaw LJ, et al. Absence of coronary artery calcification and all-cause mortality. JACC-Cardiovasc Imag 2009; 2: 692-700.
DOI URL |
| 60. |
Shao JS, Cai J, Towler DA. Molecular mechanisms of vascular calcification: lessons learned from the aorta. Arterioscl Throm Vas 2006; 26: 1423-30.
DOI URL |
| 61. |
Barinda AJ, Ikeda K, Hirata KI, Emoto N. Macrophages highly express carbonic anhydrase 2 and play a significant role in demineralization of the ectopic calcification. Kobe J Med Sci 2017; 63: E45-50.
PMID |
| 62. |
Darribère T, Schwarzbauer JE. Fibronectin matrix composition and organization can regulate cell migration during amphibian development. Mech Develop 2000; 92: 239-50.
PMID |
| 63. |
Kumra H, Sabatier L, Hassan A, et al. Roles of fibronectin isoforms in neonatal vascular development and matrix integrity. Plos Biol 2018; 16: e2004812.
DOI URL |
| 64. |
Song KS, Kim HK, Shim W, Jee SH. Plasma fibronectin levels in ischemic heart disease. Atherosclerosis 2001; 154: 449-53.
PMID |
| 65. |
Felix SB, Staudt A. Non-specific immunoadsorption in patients with dilated cardiomyopathy: mechanisms and clinical effects. Int J Cardiol 2006; 112: 30-3.
PMID |
| 66. |
Tang YL, Wang YJ, Chen LJ, Pan YH, Zhang L, Weintraub NL. Cardiac-derived stem cell-based therapy for heart failure: progress and clinical applications. Exp Biol Med (Maywood) 2013; 238: 294-300.
DOI PMID |
| [1] | HENG Xianpei, WANG Zhita, LI Liang, YANG Liuqing, HUANG Suping, JIN Lang, HE Weidong. Mechanisms of Dangua Fang (丹瓜方) in multi-target and multi-method regulation of glycolipid metabolism based on phosphoproteomics [J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 334-344. |
| [2] | WEI Lexin, LI Weiyi, TIAN Ting, ZHANG Ning, YANG Shijing, YANG Dongqing, LI Guochun, YE Fang. Identification of novel biomarkers and therapeutic target candidates for stasis-heat symptom pattern of acute intracerebral hemorrhage by quantitative plasma proteomics [J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 622-632. |
| [3] | CHEN Xiaole, WANG Peng, YANG Mengdie, ZHOU Wenjun, CHEN Jian, MENG Zhiqiang, SU Shibing. Therapeutic effect of Jianpi Liqi Fang(健脾理气方) combined with transcatheter arterial chemoembolization in patients with hepatocellular carcinoma and spleen deficiency syndrome [J]. Journal of Traditional Chinese Medicine, 2021, 41(1): 157-166. |
| [4] | Zhizhen Liu, Zongyang Yu, Xuenong OuYang, Jian Du, Xiaopeng Lan, Meng Zhao. Applied research on serum protein fingerprints for prediction of Qi deficiency syndrome and phlegm and blood stasis in patients with non-small cell lung cancer [J]. Journal of Traditional Chinese Medicine, 2012, 32(03): 350-354. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

