Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (3): 458-467.DOI: 10.19852/j.cnki.jtcm.20240423.005
Previous Articles Next Articles
Huoxue Jiedu Huayu recipe (活血解毒化瘀方) inhibits macrophage-secreted vascular endothelial growth factor-a on angiogenesis and alleviates renal fibrosis in the contralateral kidneys of unilateral ureteral obstruction rats
GAO Xiaomeng1, QIANG Panpan1, CHANG Jingyue1, FAN Lili1, YANG Fan2( ), XU Qingyou2(
), XU Qingyou2( )
)
- 1 Graduate School, Hebei University of Chinese Medicine; Hebei Key Laboratory of Integrative Medicine on Liver-Kidney Patterns, Hebei University of Chinese Medicine, Shijiazhuang 050091, China
2 Graduate School, Hebei University of Chinese Medicine; Hebei Key Laboratory of Integrative Medicine on Liver-Kidney Patterns, Hebei University of Chinese Medicine; Institute of Integrative Medicine, Hebei University of Chinese Medicine, Shijiazhuang 050091, China
-
Received:2023-02-10Accepted:2023-07-07Online:2024-06-15Published:2024-04-30 -
Contact:YANG Fan,XU Qingyou -
About author:Prof. YANG Fan, Institute of Integrative Medicine, Hebei University of Chinese Medicine, Shijiazhuang 050091, China. yangfanwy@hebcm.edu.cn Telephone: +86-15732190326
Prof. XU Qingyou, Institute of Integrative Medicine, Hebei University of Chinese Medicine, Shijiazhuang 050091, China. qingyouxu@hebcm.edu.cn;
-
Supported by:Natural Science Foundation-funded Project: Effect of Macrophage-to-Myofibroblast Transition in Contralateral Kidney of Unilateral Ureteral Obstruction Rats Through the Aldosterone/MR/SGK1 Pathway and Inhibition of Chinese Herbs(81873251);Natural Science Foundation-funded Project: Aldosterone Stimulates MR Activation to Induce Lymphangiogenesis in the Contralateral Kidney of UUO Rats and the Protective Effect of Yiqi Jiedu Huayu Herbs(82174317);Hebei Provincial Postgraduate Innovative Ability Cultivation Funding Project: Aldosterone Induced Macrophages to Secrete VEGFA to Participate in Renal Angiogenesis and the Protective Effect of Huoxue Jiedu Huayu Recipe(CXZZBS2023140);Construction Program of New Research and Development Platform and Institution, Hebei Province Innovation Ability Promotion Plan under Grant(20567624H)
Cite this article
GAO Xiaomeng, QIANG Panpan, CHANG Jingyue, FAN Lili, YANG Fan, XU Qingyou. Huoxue Jiedu Huayu recipe (活血解毒化瘀方) inhibits macrophage-secreted vascular endothelial growth factor-a on angiogenesis and alleviates renal fibrosis in the contralateral kidneys of unilateral ureteral obstruction rats[J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 458-467.
share this article
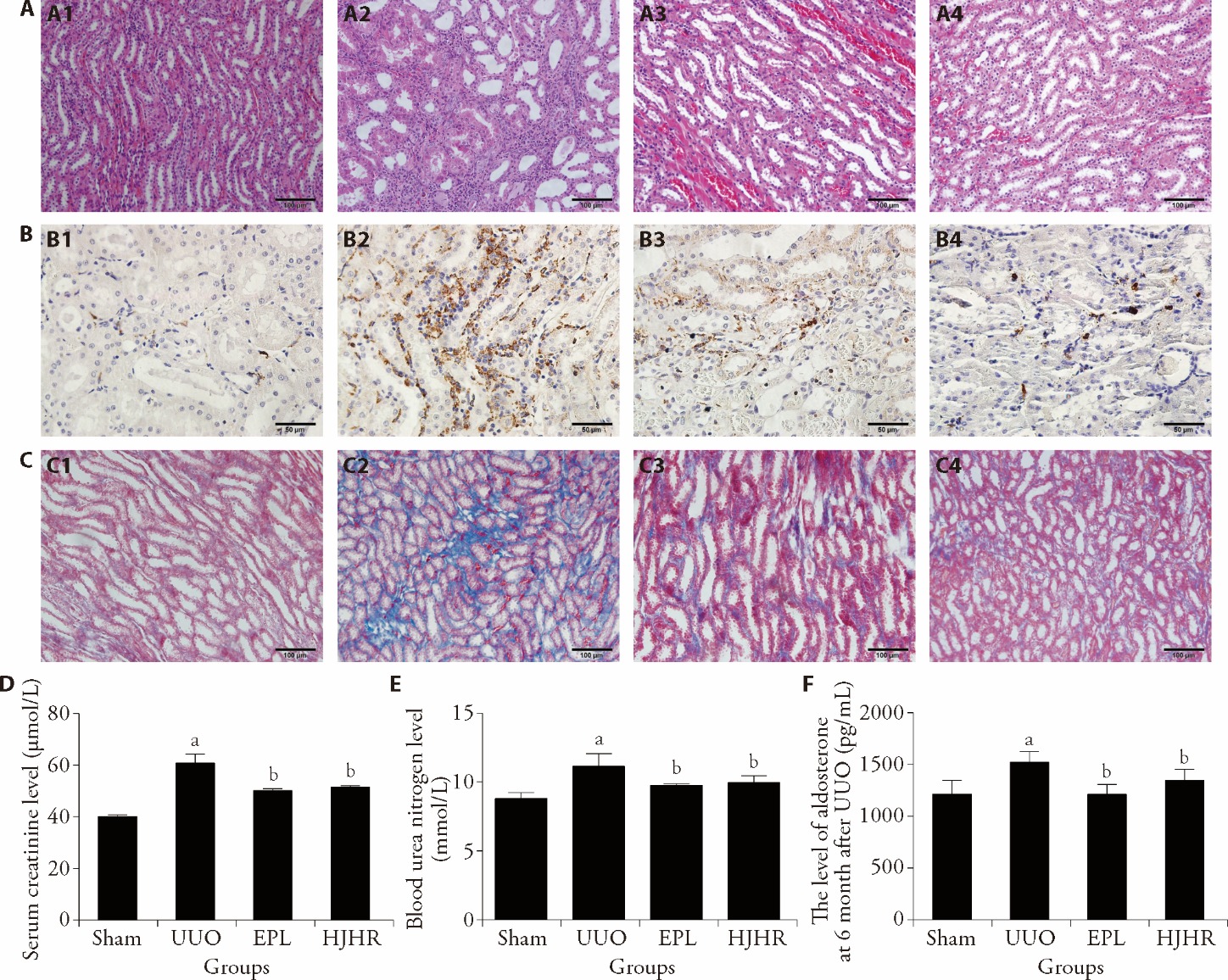
Figure 1 Effects of UUO, HJHR and EPL treatment on renal histology and fibrosis in the contralateral kidneys A: kidney sections from all groups were stained with HE to assess changes in morphology. Scale bars, 100 μm. A1: Sham group; A2: UUO group; A3: EPL group; A4: HJHR group. B: expression of CD68 was measured by immunohistochemical staining. Scale bars, 50 μm. B1: Sham group; B2: UUO group; B3: EPL group; B4: HJHR group. C: Masson's trichrome (Masson) staining was used to assess fibrosis. Scale bars, 100 μm. C1: Sham group; C2: UUO group; C3: EPL group; C4: HJHR group. D: serum creatinine was evaluated for renal function. E: blood urea nitrogen was evaluated for renal function. F: serum aldosterone level was measured. Sham group (the left ureter was exposed but not ligated, 180 d); UUO group (the left ureter was ligated, 180 d); EPL group (EPL, 100 mg·kg-1·d-1, 180 d); HJHR group (HJHR, 11.7 g·kg-1·d-1, 180 d). UUO: unilateral ureteral obstruction; EPL: eplerenone; HJHR: Huoxue Jiedu Huayu recipe; HE: hematoxylin and eosin. Quantification data are presented as the mean ± standard deviation, n = 6. aP < 0.05 vs the sham group; bP < 0.05 vs the UUO group.
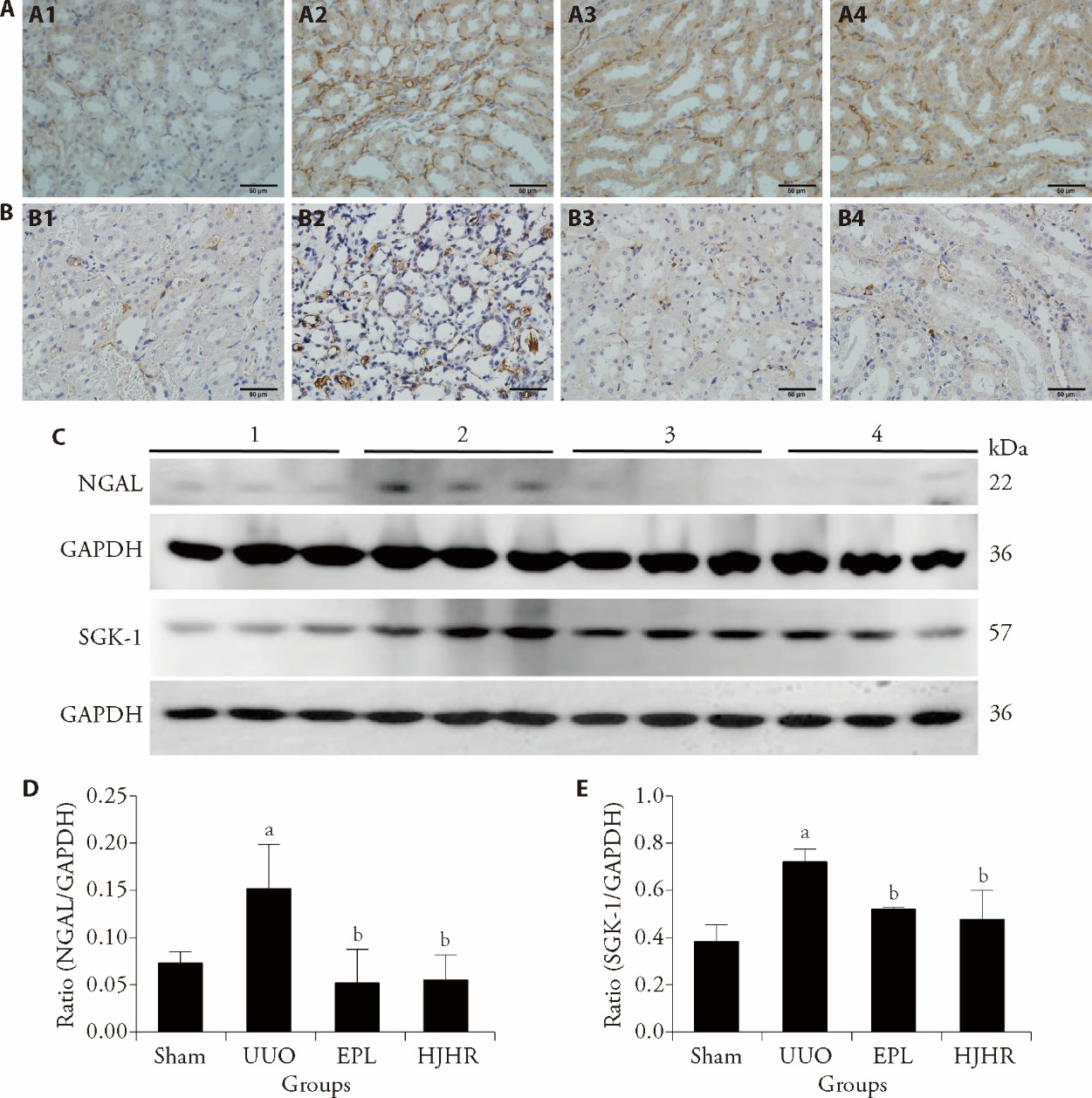
Figure 2 Effects of UUO, HJHR and EPL on angiogenesis in the contralateral kidney tissue of UUO rats A: expression of CD34 was examined by immunohistochemical staining. Scale bars, 50 μm. A1: Sham group; A2: UUO group; A3: EPL group; A4: HJHR group. B: expression of CD105 was examined by immunohistochemical staining. Scale bars, 50 μm. B1: Sham group; B2: UUO group; B3: EPL group; B4: HJHR group. C: expression of NGAL and SGK-1 protein in the contralateral kidney tissue of UUO rats was examined by Western blotting. D: protein ratio of NGAL/GAPDH; E: protein ratio of SGK-1/GAPDH; 1: Sham group; 2: UUO group; 3: EPL group; 4: HJHR group. Sham group (the left ureter was exposed but not ligated, 180 d); UUO group (the left ureter was ligated, 180 d); EPL group (EPL, 100 mg·kg-1·d-1, 180 d); HJHR group (HJHR, 11.7 g·kg-1·d-1, 180 d). UUO: unilateral ureteral obstruction; EPL: eplerenone; HJHR: Huoxue Jiedu Huayu recipe; NGAL: neutropil gelatinase-associated lipocalin; SGK-1: serum/glucocorticoid regulated kinase 1; GAPDH: glyceraldehyde-3-phosphate dehydrogenase. The data are presented as the mean ± standard deviation, n = 3. aP < 0.05 vs the sham group, bP < 0.05 vs the UUO group.

Figure 3 EndMT in the contralateral kidneys of UUO rats A: expression of α-SMA and vimentin was detected by immunohistochemistry (scale bars, 100 μm) and real-time PCR to examine renal infiltration of myofibroblasts. A1-A4: immunohistochemical staining using antibodies against α-SMA. A1: Sham group; A2: UUO group; A3: EPL group; A4: HJHR group. A5: expression of α-SMA mRNA in the contralateral kidney tissue of UUO rats was examined by real-time PCR. A6-A9: immunohistochemical staining using antibodies against vimentin. A6: Sham group; A7: UUO group; A8: EPL group; A9: HJHR group. A10: expression of vimentin mRNA in the contralateral kidney tissue of UUO rats was examined by real-time PCR. B: expression of collagen I was detected by immunohistochemistry (scale bars, 100 μm) and real-time PCR. B1-B4: immunohistochemical staining using antibodies against collagen I. B1: Sham group; B2: UUO group; B3: EPL group; B4: HJHR group. B5: expression of collagen I mRNA in the contralateral kidney tissue of UUO rats was examined by real-time PCR. C: coexpression (Merged, yellow) of vascular endothelial cell (FITC, CD34, green) and myofibroblast (TRITC, α-SMA, red) markers in the UUO group was examined by two-color immunofluorescence staining, and nuclei were stained with DAPI (blue). Scale bars, 50 μm. D: coexpression (Merged, yellow) of vascular endothelial cell (FITC, CD105, green) and myofibroblast (TRITC, α-SMA, red) markers in the UUO group was examined by two-color immunofluorescence staining, and nuclei were stained with DAPI (blue). Scale bars, 50 μm. Sham group (the left ureter was exposed but not ligated, 180 d); UUO group (the left ureter was ligated, 180 d); EPL group (EPL, 100 mg·kg-1·d-1, 180 d); HJHR group (HJHR, 11.7 g·kg-1·d-1, 180 d). UUO: unilateral ureteral obstruction; EPL: eplerenone; HJHR: Huoxue Jiedu Huayu recipe; EndMT: endothelial-to-mesenchymal transition; α-SMA: α-smooth muscle actin. DAPI: 4',6-diamidino-2-phenylindole; FITC: fluorescein isothiocyanate; TRITC: tetraethyl rhodamine isothiocyanate. The data are presented as the mean ± standard deviation, n = 3. aP < 0.05 vs the sham group, bP < 0.05 vs the UUO group.
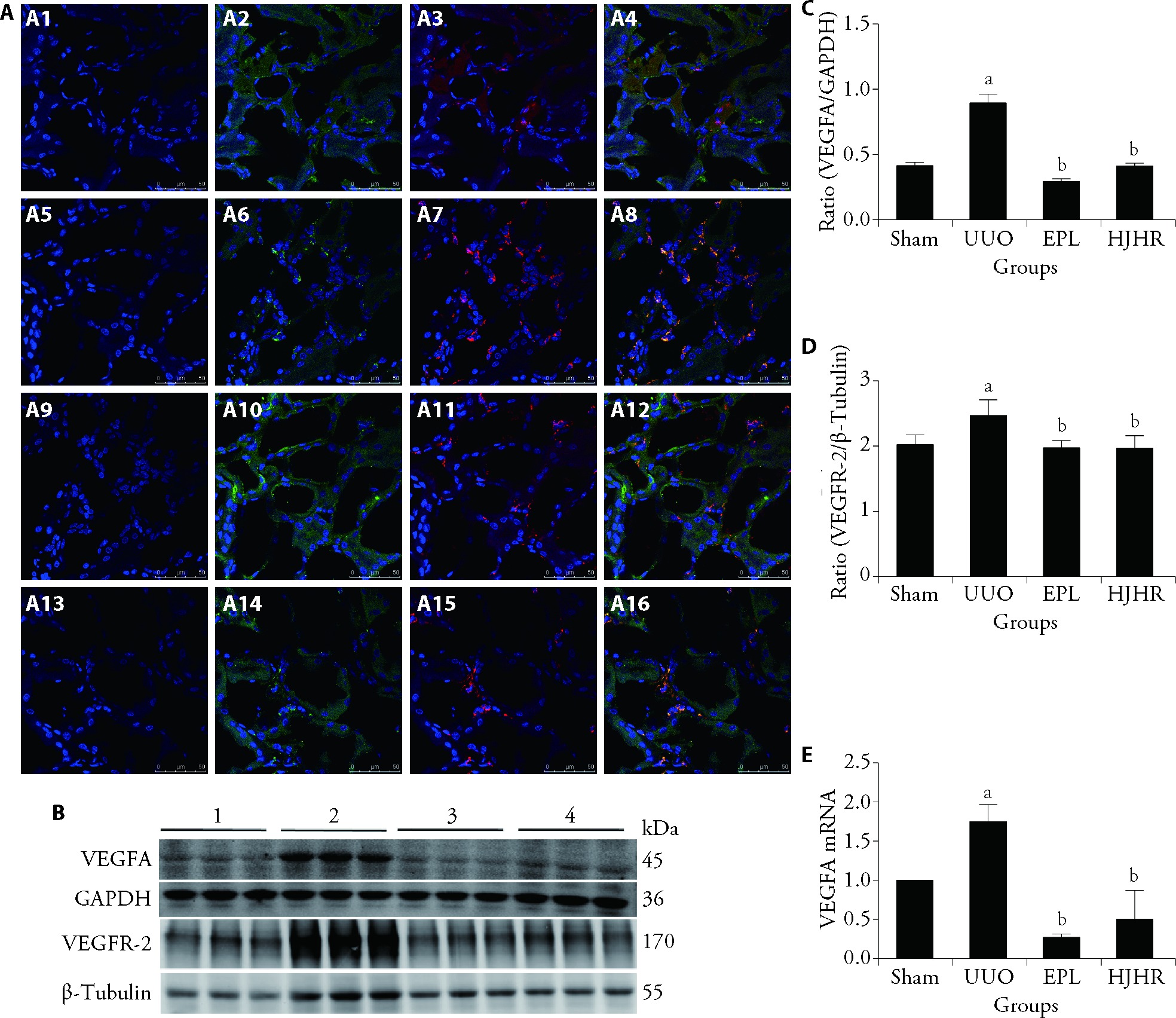
Figure 4 Effects of VEGFA produced by macrophages on angiogenesis in the contralateral kidneys of UUO rats A: coexpression (Merged, yellow) of vascular endothelial cell (FITC, VEGFA, green) and macrophage (TRITC, CD68, red) markers was examined by two-color immunofluorescence staining, and nuclei were stained with DAPI (blue). Scale bars, 50 μm. A1-A4: Sham group; A5-A8: UUO group; A9-A12: EPL group; A13-A16: HJHR group. B: expression of VEGFA and VEGFR-2 protein in the contralateral kidney tissue of UUO rats was examined by Western blotting. C: protein ratio of VEGFA/GAPDH; D: protein ratio of VEGFR-2/β-Tubulin; 1: Sham group; 2: UUO group; 3: EPL group; 4: HJHR group. E: expression of VEGFA mRNA in the contralateral kidney tissue of UUO rats was examined by real-time PCR. Sham group (the left ureter was exposed but not ligated, 180 d); UUO group (the left ureter was ligated, 180 d); EPL group (EPL, 100 mg·kg-1·d-1, 180 d); HJHR group (HJHR, 11.7 g·kg-1·d-1, 180 d). UUO: unilateral ureteral obstruction; EPL: eplerenone; HJHR: Huoxue Jiedu Huayu recipe; DAPI: 4',6-diamidino-2-phenylindole; FITC: fluorescein isothiocyanate; TRITC: tetraethyl rhodamine isothiocyanate; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; VEGFA: vascular endothelial growth factor-A; VEGFR-2: vascular endothelial growth factor receptor-2. The data are presented as the mean ± standard deviation, n = 3. aP < 0.05 vs the sham group, bP < 0.05 vs the UUO group.
| 1. |
Romagnani P, Remuzzi G, Glassock R, et al. Chronic kidney disease. Nat Rev Dis Primers 2017; 3: 17088.
DOI PMID |
| 2. |
Collaboration GBDCKD. Global, regional, and national burden of chronic kidney disease, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020; 395: 709-33.
DOI PMID |
| 3. |
Chevalier RL, Forbes MS, Thornhill BA. Ureteral obstruction as a model of renal interstitial fibrosis and obstructive nephropathy. Kidney Int 2009; 75: 1145-52.
DOI PMID |
| 4. |
Krishnan SM, Ling YH, Huuskes BM, et al. Pharmacological inhibition of the NLRP3 inflammasome reduces blood pressure, renal damage, and dysfunction in salt-sensitive hypertension. Cardiovasc Res 2019; 115: 776-87.
DOI PMID |
| 5. |
Su H, Wan C, Song A, et al. Oxidative stress and renal fibrosis: mechanisms and therapies. Adv Exp Med Biol 2019; 1165: 585-604.
DOI PMID |
| 6. | Zhao XC, Livingston MJ, Liang XL, et al. Cell apoptosis and autophagy in renal fibrosis. Adv Exp Med Biol 2019; 1165: 557-84. |
| 7. |
Crewe C, An YA, Scherer PE. The ominous triad of adipose tissue dysfunction: inflammation, fibrosis, and impaired angiogenesis. J Clin Invest 2017; 127: 74-82.
DOI PMID |
| 8. |
Hanumegowda C, Farkas L, Kolb M. Angiogenesis in pulmonary fibrosis: too much or not enough? Chest 2012; 142: 200-7.
DOI PMID |
| 9. |
Ren Y, Zhang Y, Wang L, et al. Selective targeting of vascular endothelial YAP activity blocks EndMT and ameliorates unilateral ureteral obstruction-induced kidney fibrosis. ACS Pharmacol Transl Sci 2021; 4: 1066-74.
DOI PMID |
| 10. |
Zeisberg EM, Potenta SE, Sugimoto H, et al. Fibroblasts in kidney fibrosis emerge via endothelial-to-mesenchymal transition. J Am Soc Nephrol 2008; 19: 2282-7.
DOI URL |
| 11. |
Carmeliet P. VEGF as a key mediator of angiogenesis in cancer. Oncology 2005; 69 Suppl 3: 4-10.
DOI URL |
| 12. |
Claesson-Welsh L, Welsh M. VEGFA and tumour angiogenesis. J Intern Med 2013; 273: 114-27.
DOI PMID |
| 13. | Lieber GB, Fernandez X, Mingo GG, et al. Mineralocorticoid receptor antagonists attenuate pulmonary inflammation and bleomycin-evoked fibrosis in rodent models. Eur J Pharmacol 2013; 718: 290-8. |
| 14. |
Xiong Y, Chang Y, Hao J, et al. Eplerenone attenuates fibrosis in the contralateral kidney of UUO rats by preventing macrophage-to-myofibroblast transition. Front Pharmacol 2021; 12: 620433.
DOI URL |
| 15. |
Toto RD. Aldosterone blockade in chronic kidney disease: can it improve outcome? Curr Opin Nephrol Hypertens 2010; 19: 444-9.
DOI URL |
| 16. |
Wang CH, Wang Z, Liang LJ, et al. The inhibitory effect of eplerenone on cell proliferation in the contralateral kidneys of rats with unilateral ureteral obstruction. Nephron 2017; 136: 328-38.
DOI URL |
| 17. |
Shen YL, Wang SJ, Rahman K, et al. Chinese herbal formulas and renal fibrosis: an overview. Curr Pharm Des 2018; 24: 2774-81.
DOI URL |
| 18. |
Zhou S, Ai Z, Li W, et al. Deciphering the pharmacological mechanisms of Taohe-chengqi decoction extract against renal fibrosis through integrating network pharmacology and experimental validation in vitro and in vivo. Front Pharmacol 2020; 11: 425.
DOI URL |
| 19. | Ma X, Qiang P, Chen G, et al. Huoxue Jiedu Huayu formula alleviates cell pyroptosis in contralateral kidneys of 6-month-old UUO rats through the NLRP3/Caspase-1/IL-1beta pathway. Evid Based Complement Alternat Med 2021; 2021: 5533911. |
| 20. | Zhang Y, Hao J, Ma X, et al. Huoxue Jiedu Huayu recipe ameliorates mesangial cell pyroptosis in contralateral kidney of UUO rats. Evid Based Complement Alternat Med 2020; 2020: 2530431. |
| 21. |
Arai K, Tsuruoka H, Homma T. CS-3150, a novel non-steroidal mineralocorticoid receptor antagonist, prevents hypertension and cardiorenal injury in Dahl salt-sensitive hypertensive rats. Eur J Pharmacol 2015; 769: 266-73.
DOI PMID |
| 22. |
Truong LD, Gaber L, Eknoyan G. Obstructive uropathy. Contrib Nephrol 2011; 169: 311-26.
DOI PMID |
| 23. | Rabb H, Ramirez G, Saba SR, et al. Renal ischemic-reperfusion injury in L-selectin-deficient mice. Am J Physiol 1996; 271: F408-13. |
| 24. | Eddy AA. Overview of the cellular and molecular basis of kidney fibrosis. Kidney Int Suppl (2011) 2014; 4: 2-8. |
| 25. |
Liu Y. Renal fibrosis: new insights into the pathogenesis and therapeutics. Kidney Int 2006; 69: 213-7.
DOI PMID |
| 26. |
Zhu MQ, De Broe ME, Nouwen EJ. Vimentin expression and distal tubular damage in the rat kidney. Exp Nephrol 1996; 4: 172-83.
PMID |
| 27. |
Klahr S. Obstructive nephropathy. Intern Med 2000; 39: 355-61.
DOI URL |
| 28. |
Klomjit N, Zhu XY, Massat AE, et al. Microvascular remodeling and altered angiogenic signaling in human kidneys distal to occlusive atherosclerotic renal artery stenosis. Nephrol Dial Transplant 2022; 37: 1844-56.
DOI URL |
| 29. |
Da J, Yang Y, Dong R, et al. Therapeutic effect of 1,25(OH)2-VitaminD3 on fibrosis and angiogenesis of peritoneum induced by chlorhexidine. Biomed Pharmacother 2020; 129: 110431.
DOI PMID |
| 30. |
Feng J, Wang C, Liu T, et al. Procyanidin B2 inhibits the activation of hepatic stellate cells and angiogenesis via the Hedgehog pathway during liver fibrosis. J Cell Mol Med 2019; 23: 6479-93.
DOI URL |
| 31. |
Wu X, Reboll MR, Korf-Klingebiel M, et al. Angiogenesis after acute myocardial infarction. Cardiovasc Res 2021; 117: 1257-73.
DOI PMID |
| 32. |
Su CT, Jao TM, Urban Z, et al. LTBP4 affects renal fibrosis by influencing angiogenesis and altering mitochondrial structure. Cell Death Dis 2021; 12: 943.
DOI |
| 33. |
Zhang Y, Nakano D, Guan Y, et al. A sodium-glucose cotransporter 2 inhibitor attenuates renal capillary injury and fibrosis by a vascular endothelial growth factor-dependent pathway after renal injury in mice. Kidney Int 2018; 94: 524-35.
DOI PMID |
| 34. |
Zhang Y, Fan K, Xu X, et al. The TGF-beta1 induces the endothelial-to-mesenchymal transition via the UCA1/miR-455/ZEB1 regulatory axis in human umbilical vein endothelial cells. DNA Cell Biol 2020; 39: 1264-73.
DOI PMID |
| 35. | Srivastava SP, Koya D, Kanasaki K. MicroRNAs in kidney fibrosis and diabetic nephropathy: roles on EMT and EndMT. Biomed Res Int 2013; 2013: 125469. |
| 36. |
Zeisberg EM, Tarnavski O, Zeisberg M, et al. Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat Med 2007; 13: 952-61.
DOI PMID |
| 37. |
Wang Z, Han Z, Tao J, et al. Role of endothelial-to-mesenchymal transition induced by TGF-beta1 in transplant kidney interstitial fibrosis. J Cell Mol Med 2017; 21: 2359-69.
DOI URL |
| 38. |
Siemerink MJ, Hughes MR, Dallinga MG, et al. CD34 promotes pathological epi-retinal neovascularization in a mouse model of oxygen-induced retinopathy. PLoS One 2016; 11: e0157902.
DOI URL |
| 39. |
Zhao T, Zhao W, Chen Y, et al. Vascular endothelial growth factor (VEGF)-A: role on cardiac angiogenesis following myocardial infarction. Microvasc Res 2010; 80: 188-94.
DOI PMID |
| 40. |
Wang J, Yang GY, Sun HY, et al. Dioscin reduces vascular damage in the retina of db/db mice by inhibiting the VEGFA signaling pathway. Front Pharmacol 2021; 12: 811897.
DOI URL |
| 41. |
Lu Y, Qin T, Li J, et al. MicroRNA-140-5p inhibits invasion and angiogenesis through targeting VEGF-A in breast cancer. Cancer Gene Ther 2017; 24: 386-92.
DOI PMID |
| 42. |
Wang R, Ma Y, Zhan S, et al. B7-H 3 promotes colorectal cancer angiogenesis through activating the NF-kappaB pathway to induce VEGFA expression. Cell Death Dis 2020; 11: 55.
DOI |
| 43. |
Zhao H, Kalish FS, Wong RJ, et al. Hypoxia regulates placental angiogenesis via alternatively activated macrophages. Am J Reprod Immunol 2018; 80: e12989.
DOI URL |
| 44. |
Guo L, Akahori H, Harari E, et al. CD163+ macrophages promote angiogenesis and vascular permeability accompanied by inflammation in atherosclerosis. J Clin Invest 2018; 128: 1106-24.
DOI PMID |
| 45. |
Zhang Q, Cunha APD, Li S, et al. IL-27 regulates HIF-1alpha-mediated VEGFA response in macrophages of diabetic retinopathy patients and healthy individuals. Cytokine 2019; 113: 238-47.
DOI PMID |
| 46. |
Li C, Wang W, Norregaard R, et al. Altered expression of epithelial sodium channel in rats with bilateral or unilateral ureteral obstruction. Am J Physiol Renal Physiol 2007; 293: F333-41.
DOI URL |
| 47. |
Ma X, Chang Y, Xiong Y, et al. Eplerenone ameliorates cell pyroptosis in contralateral kidneys of rats with unilateral ureteral obstruction. Nephron 2019; 142: 233-42.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

