Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (1): 60-67.DOI: 10.19852/j.cnki.jtcm.2023.01.007
• Original articles • Previous Articles Next Articles
Astragaloside IV ameliorates insulin induced insulin resistance in HepG2 cells through reactive oxygen species mediated c-Jun N-terminal kinase pathway
YE Xiaomei1,2, XING Xiaowei1, YUAN Kangrui1, WANG Dongming4, WU Dudu1, CHEN Zhi1( ), YU Zhiqiang3(
), YU Zhiqiang3( )
)
- 1 School of Pharmacy, Guangdong Medical University, Dongguan 523808, China
2 Department of Clinical Pharmacy, SSL Central Hospital of Dongguan City (the Third People's Hospital of Dongguan City), Dongguan 523326, China
3 School of Pharmaceutical Sciences, Guangdong Provincial Key Laboratory of New Drug Screening, Southern Medical University, Guangzhou 510515, China
4 Department of Pharmacy, Central People's Hospital of Zhanjiang, Zhanjiang 524037, China
-
Received:2021-03-22Accepted:2021-06-12Online:2023-02-15Published:2023-01-10 -
Contact:CHEN Zhi,YU Zhiqiang -
About author:YU Zhiqiang, School of Pharmaceutical Sciences, Guangdong Provincial Key Laboratory of New Drug Screening, Southern Medical University, Guangzhou 510515, China. zhiqiang.yu.1@asu.edu. Telephone:+86-769-22896321; +86-769-22896560
CHEN Zhi, School of Pharmaceutical Sciences, Guangdong Provincial Key Laboratory of New Drug Screening, Southern Medical University, Guangzhou 510515, China. czwududu@126.com;
-
Supported by:National Natural Science Foundation of Guangdong Province(2017A030310666);National Natural Science Foundation of Guangdong Province(2018A030307003);Guangdong Medical University Nanhai Marine Biomedical Resources R&D Public Service Platform Open Fund Project(2HC18013);Guangdong Medical University Nanhai Marine Biomedical Resources R&D Public Service Platform Open Fund Project(2HC18016);“Group-type” Special Support Project for Education Talents in Universities(4SG19045G);Undergraduate Science & Technology Innovation Foundation of Guangdong Province(201810571046);Undergraduate Science & Technology Innovation Foundation of Guangdong Province(201810571073);Medical Science and Technology Development Foundation of Guangdong Province(A2016355);The Opening Project of State Key Laboratory of Polymer Materials Engineering (Sichuan University)(sklpme2018-4-23)
Cite this article
YE Xiaomei, XING Xiaowei, YUAN Kangrui, WANG Dongming, WU Dudu, CHEN Zhi, YU Zhiqiang. Astragaloside IV ameliorates insulin induced insulin resistance in HepG2 cells through reactive oxygen species mediated c-Jun N-terminal kinase pathway[J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 60-67.
share this article
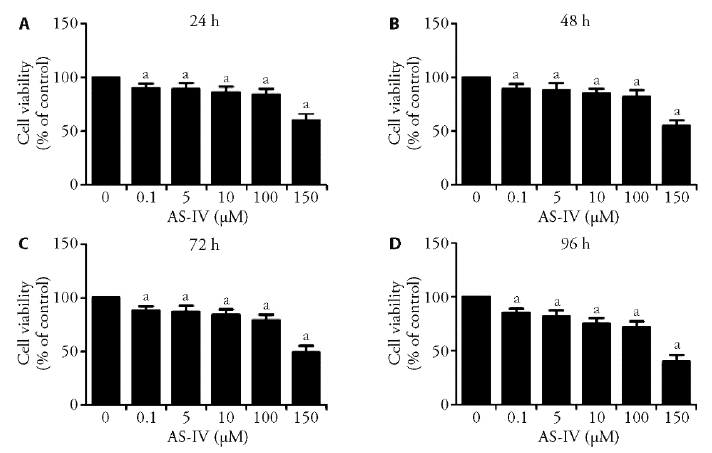
Figure 1 Effects of AS-IV on the viability of HepG2 cells A-D: cells were treated with 0.1, 1, 5, 10, 100 or 150 μM AS-IV for 24 (A), 48 (B), 72 (C) and 96h (D), and inhibition of cell viability was determined by MTT assay. Results are expressed as the mean ± standard deviation of three independent experiments (n = 3). aP < 0.05 compared with untreated cells. MTT: 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide.

Figure 2 Effects of AS-IV on ameliorating glucose consumption in insulin resistance N: control group, given conventional feed without other interfering factors. M: model group, given high sugar and fat feed but no other interfering factors; ROZ: positive group (30 μM rosiglitazone). [Results are expressed as the mean ± standard deviation of three independent experiments (n = 3). aP < 0.05 versus control, bP < 0.05 versus model].
| Group | n | FBG (mmol/L) | C-P (ng/mL) | TNF-α (ng/mL) | IL-6 (ng/mL) |
|---|---|---|---|---|---|
| N | 8 | 5.46±0.49a | 1.79±0.22a | 149±25.92a | 49±10.45a |
| M | 8 | 20.48±1.48b | 0.84±0.25b | 238±37.18b | 135±16.53b |
| A1 | 8 | 17.58±2.74bc | 1.15±0.38b | 194±33.45c | 96±18.42ab |
| A2 | 8 | 15.65±1.51ab | 1.34±0.51c | 176±38.16a | 79±19.13ab |
| A3 | 8 | 13.89±3.18ab | 1.59±0.36a | 168±29.13a | 71±21.76ad |
| x | 8 | 13.94±1.58ab | 1.67±0.31a | 185±34.43a | 80±19.24ab |
Table 1 Comparison of the levels of FBG, C-P, TNF-α, IL-6 in different groups ($\bar{x}\pm s$)
| Group | n | FBG (mmol/L) | C-P (ng/mL) | TNF-α (ng/mL) | IL-6 (ng/mL) |
|---|---|---|---|---|---|
| N | 8 | 5.46±0.49a | 1.79±0.22a | 149±25.92a | 49±10.45a |
| M | 8 | 20.48±1.48b | 0.84±0.25b | 238±37.18b | 135±16.53b |
| A1 | 8 | 17.58±2.74bc | 1.15±0.38b | 194±33.45c | 96±18.42ab |
| A2 | 8 | 15.65±1.51ab | 1.34±0.51c | 176±38.16a | 79±19.13ab |
| A3 | 8 | 13.89±3.18ab | 1.59±0.36a | 168±29.13a | 71±21.76ad |
| x | 8 | 13.94±1.58ab | 1.67±0.31a | 185±34.43a | 80±19.24ab |
| Group | n | Blood glucose | ||||
|---|---|---|---|---|---|---|
| 0 h | 0.5 h | 1 h | 2 h | AUC | ||
| N | 8 | 5.54±1.89a | 7.55±1.47a | 7.17±1.40a | 5.70±1.39a | 12.92±1.69a |
| M | 8 | 20.59±1.95b | 31.58±2.14b | 27.72±1.81b | 23.44±2.41b | 51.41±3.51b |
| A1 | 8 | 17.63±1.69ab | 28.35±2.97bc | 24.37±0.88ab | 19.22±1.57ab | 44.18±1.94ab |
| A2 | 8 | 15.31±2.29ab | 26.59±3.14ab | 24.16±2.34ab | 17.39±2.19ab | 41.63±3.05ab |
| A3 | 8 | 13.47±2.46ab | 23.40±2.93ab | 18.42±2.31ab | 11.47±1.57ab | 31.63±2.97ab |
| x | 8 | 13.56±0.95ab | 24.39±1.48ab | 19.30±1.83ab | 12.26±2.40ab | 33.15±3.75ab |
Table 2 Comparison of the levels of blood glucose during OGTT in different groups ($\bar{x}\pm s$)
| Group | n | Blood glucose | ||||
|---|---|---|---|---|---|---|
| 0 h | 0.5 h | 1 h | 2 h | AUC | ||
| N | 8 | 5.54±1.89a | 7.55±1.47a | 7.17±1.40a | 5.70±1.39a | 12.92±1.69a |
| M | 8 | 20.59±1.95b | 31.58±2.14b | 27.72±1.81b | 23.44±2.41b | 51.41±3.51b |
| A1 | 8 | 17.63±1.69ab | 28.35±2.97bc | 24.37±0.88ab | 19.22±1.57ab | 44.18±1.94ab |
| A2 | 8 | 15.31±2.29ab | 26.59±3.14ab | 24.16±2.34ab | 17.39±2.19ab | 41.63±3.05ab |
| A3 | 8 | 13.47±2.46ab | 23.40±2.93ab | 18.42±2.31ab | 11.47±1.57ab | 31.63±2.97ab |
| x | 8 | 13.56±0.95ab | 24.39±1.48ab | 19.30±1.83ab | 12.26±2.40ab | 33.15±3.75ab |
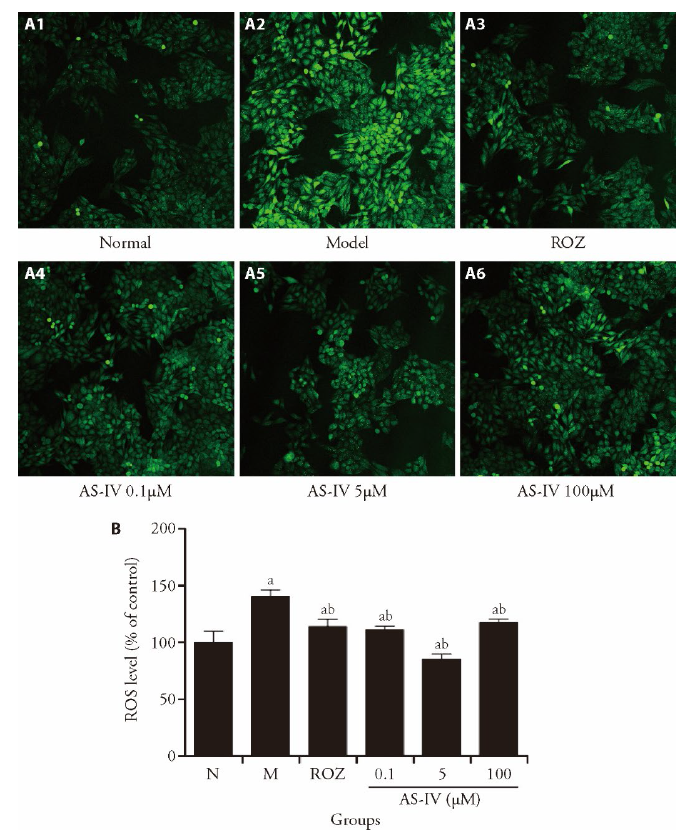
Figure 3 AS-IV ameliorates ROS accumulation in HepG2 cells A: AS-IV reduced insulin-increased intracellular ROS levels in HepG2 cells. B: ROS levels were quantified. n = 3, aP < 0.05 vs the control group, bP < 0.05 vs the insulin resistant group. N: control group, given conventional feed without other interfering factors. M: model group, given high sugar and fat feed but no other interfering factors; ROZ: positive group (30 μM rosiglitazone). ROS: reactive oxygen species.

Figure 4 Effects of AS-IV on regulation of JNK-AKT-GSK3β signaling pathway in insulin-resistant HepG2 cells A: the expressions of JNK and phosphorylated JNK (p-JNK) in insulin-resistant HepG2 cells. B: Relative p-JNK levels were quantified. C: the expressions of PDK1 in insulin-resistant HepG2 cells. D: relative PDK1 levels were shown. E: the expressions of AKT in insulin-resistant HepG2 cells. F: relative AKT levels were quantified. G: the expressions of GSK3β in insulin-resistant HepG2 cells. H:relative GSK3β levels were displayed. Data are expressed as means ± SD from three individual experiments. aP < 0.001, dP < 0.05, fP < 0.01, compared to normal group; bP < 0.05, eP < 0.01, cP < 0.001 compared to model group. N: control group; M: model group; ROZ: rosiglitazone group (30 μM).
| 1 |
Riddy DM, Delerive P, Summers RJ, Sexton PM, Langmead CJ. G Protein-Coupled Receptors Targeting Insulin Resistance, Obesity, and Type 2 Diabetes Mellitus. Pharmacol Rev 2018; 70: 39-67.
DOI PMID |
| 2 |
Farrokhian A, Bahmani F, Taghizadeh M, et al. Selenium supplementation affects insulin resistance and serum hs-CRP in patients with type 2 diabetes and coronary heart disease. Horm Metab Res 2016; 48: 263-8.
DOI PMID |
| 3 |
Motiani P, Virtanen KA, Motiani KK, et al. Decreased insulin-stimulated brown adipose tissue glucose uptake after short-term exercise training in healthy middle-aged men. Diabetes Obes Metab 2017; 19: 1379-88.
DOI PMID |
| 4 | Keane KN, Cruzat VF, Carlessi R, de Bittencourt PIH, Newsholme P. Molecular events linking oxidative stress and inflammation to insulin resistance and β-cell dysfunction. Oxid Med Cell Longev 2015; 181643. |
| 5 |
Bonnard C, Durand A, Peyrol S, et al. Mitochondrial dysfunction results from oxidative stress in the skeletal muscle of diet-induced insulin-resistant mice. J Clin Invest 2008; 118: 789-800.
DOI PMID |
| 6 |
Rehman K, Akash MSH. Mechanism of generation of oxidative stress and pathophysiology of type 2 diabetes mellitus: how are they interlinked? J Cell Biochem 2017; 118: 3577-85.
DOI PMID |
| 7 | Badeau RM, Honka M-J, Lautamäki R, et al. Systemic metabolic markers and myocardial glucose uptake in type 2 diabetic and coronary artery disease patients treated for 16 weeks with rosiglitazone, a PPARγ agonist. Ann Med 2014; 46: 18-23. |
| 8 |
Zhang J, Liu X, Xie XB, Cheng XC, Wang RL. Multitargeted bioactive ligands for PPARs discovered in the last decade. Chem Biol Drug Des 2016; 88: 635-63.
DOI PMID |
| 9 |
Meijun L, Zhicheng L, Bin X, Wei Z, Jianwei C. Review of systematic reviews and Meta-analyses investigating Traditional Chinese Medicine treatment for type 2 diabetes mellitus. J Tradit Chin Med 2016; 36: 555-63.
PMID |
| 10 |
Li L, Hou X, Xu R, Liu C, Tu M. Research review on the pharmacological effects of astragaloside IV. Fund Clin Pharmacol 2017; 31: 17-36.
DOI PMID |
| 11 |
Lv L, Wu SY, Wang GF, et al. Effect of astragaloside IV on hepatic glucose-regulating enzymes in diabetic mice induced by a high-fat diet and streptozotocin. Phytother Res 2010; 24: 219-24.
DOI PMID |
| 12 |
Yin Y, Qi F, Song Z, Zhang B, Teng J. Ferulic acid combined with astragaloside IV protects against vascular endothelial dysfunction in diabetic rats. BioSci Trends 2014; 8: 217-26.
PMID |
| 13 | Ji TJ, Wang ZY, Zhu YF, et al. Effect of astragaloside Ⅳ in regulating PI3K/Akt/FoxO1 pathway and inhibiting hepatic gluconeogenesis in diabetic rats. Zhong Guo Shi Yan Fang Ji Xue Za Zhi 2020; 26: 78-86. |
| 14 | Wang LN, Fang XM, Hu JP, Jia XF, Zhu GQ, Li YJ. Effect of Qidi Xiaoke prescription on C-peptide, tumor necrosis factor-α, interleukin-6, and histopathological changes of the spleen in rats with type 2 diabetes with deficiency of both Qi and Yin. J Anhui Univ Chin Med 2017; 36: 72-5. |
| 15 | Li M, Yang CY, Fu CH, Xiong YG, Ling Y. Study of purslane/ licorice/dandelion compound on insulin resistance in type 2 diabetic rats. Acta Lab Anim Sci Sin 2020: 1-10. |
| 16 | Liu P, Shi L, Cang X, et al. CtBP2 ameliorates palmitate-induced insulin resistance in HepG2 cells through ROS mediated JNK pathway. Gen and Comp Endocr 2017; 247: 66-73. |
| 17 | Lee BC, Lee J. Cellular and molecular players in adipose tissue inflammation in the development of obesity-induced insulin resistance. BBA - Mol Basis Dis 2014; 1842: 446-62. |
| 18 |
Dai B, Wu Q, Zeng C, et al. The effect of Liuwei Dihuang decoction on PI3K/Akt signaling pathway in liver of type 2 diabetes mellitus (T2DM) rats with insulin resistance. J Ethnopharmacol 2016; 192: 382-9.
DOI PMID |
| 19 | Zhang H, Ge Z, Tang S, Meng R, Bi Y, Zhu D. Erythropoietin ameliorates PA-induced insulin resistance through the IRS/AKT/ FOXO1 and GSK-3β signaling pathway, and inhibits the inflammatory response in HepG2 cells. Mol Med Rep 2017; 16: 2295-301. |
| 20 |
Gao W, Du X, Lei L, et al. NEFA-induced ROS impaired insulin signalling through the JNK and p38MAPK pathways in non-alcoholic steatohepatitis. J Cell Mol Med 2018; 22: 3408.
DOI PMID |
| 21 | Dong Y, Gao G, Fan H, Li S, Li X, Liu W. Activation of the liver X receptor by agonist TO901317 improves hepatic insulin resistance via suppressing reactive oxygen species and JNK Pathway. PLoS One 2015; 10: e0124778. |
| 22 | Yang Y, Li W, Li Y, Wang Q, Gao L, Zhao J. Dietary Lycium barbarum polysaccharide induces Nrf2/ARE pathway and ameliorates insulin resistance induced by high-fat via activation of PI3K/AKT sSignaling. Oxid Med Cell Longev 2014; 145641. |
| 23 | He S, Peng WB, Zhou HL. Combination treatment of deep sea water and fucoidan attenuates high glucose-induced insulin-resistance in HepG2 hepatocytes. Mar Drugs 2018; 16: 48. |
| 24 | Cordero-Herrera I, Martín MÁ, Goya L, Ramos S. Cocoa flavonoids attenuate high glucose-induced insulin signalling blockade and modulate glucose uptake and production in human HepG2 cells. Food Chem Toxicol 2014; 64: 10-9. |
| 25 | Song R, Wang X, Mao Y, et al. Resistin disrupts glycogen synthesis under high insulin and high glucose levels by down-regulating the hepatic levels of GSK3β. Gene 2013; 529: 50-6. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

