Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (4): 815-823.DOI: 10.19852/j.cnki.jtcm.20230517.001
Previous Articles Next Articles
Effectiveness and safety of Xuefu Zhuyu oral liquid (血府逐瘀口服液) on Qi-stagnation and blood-stasis pattern in patients with stable angina, tension-type headache and primary dysmenorrhea: rationale and design of a master protocol
CAO Wencong1, LIAO Shaojun1, ZHANG Yuanwen2, ZHOU Li3,4, LI Geng3,4, OUYANG Wenwei3,4, WEN Zehuai3,4,5( )
)
- 1 Second Clinical Medical College (Second Affiliated Hospital), Guangzhou University of Chinese Medicine, Guangzhou 510405, China
2 First Clinical Medical College (First Affiliated Hospital), Guangzhou University of Chinese Medicine, Guangzhou 510405, China
3 Key Unit of Methodology in Clinical Research, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, China
4 State Key Laboratory of Dampness Syndrome of Chinese Medicine, Second Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510120, China
5 Science and Technology Innovation Center, Guangzhou University of Chinese Medicine, Guangzhou 510405, China
-
Received:2022-03-22Accepted:2022-07-19Online:2023-08-15Published:2023-05-17 -
Contact:WEN Zehuai, Key Unit of Methodology in Clinical Research, Guangdong Provincial Hospital of Chinese Medicine, Guangzhou 510120, China. wenzh@gzucm.edu.cn. Telephone: +86 13903008091 -
Supported by:National Key Research and Development Program of China: Research on “the Pattern Dominating Disease” of Post-market Evaluation on Two Classic Chinese Herbal Formulas based on Basket Design(2018YFC1707407)
Cite this article
CAO Wencong, LIAO Shaojun, ZHANG Yuanwen, ZHOU Li, LI Geng, OUYANG Wenwei, WEN Zehuai. Effectiveness and safety of Xuefu Zhuyu oral liquid (血府逐瘀口服液) on Qi-stagnation and blood-stasis pattern in patients with stable angina, tension-type headache and primary dysmenorrhea: rationale and design of a master protocol[J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 815-823.
share this article
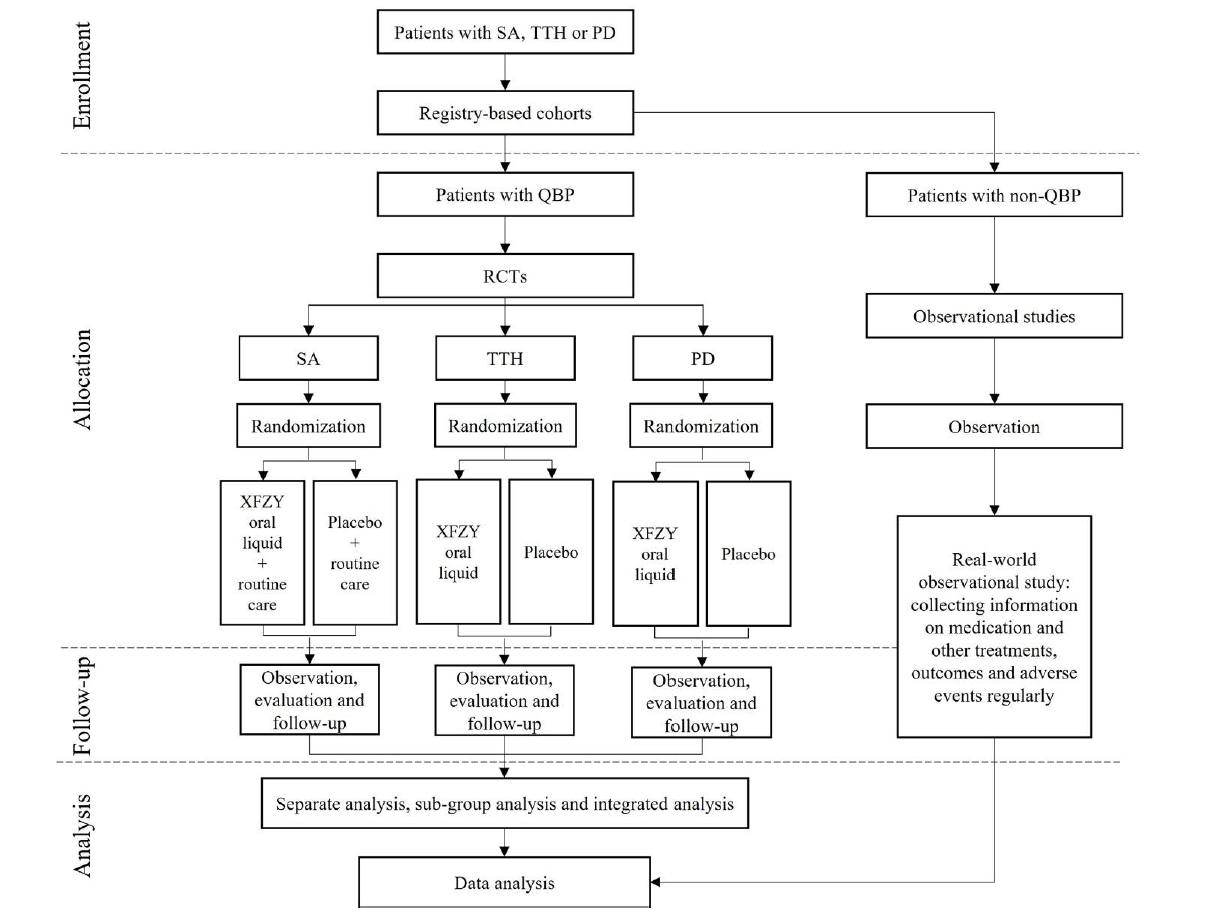
Figure 1 Study flowchart QBP: Qi-stagnation and blood-stasis pattern; RCTs: randomized controlled trials; SA: stable angina; TTH: tension-type headache; PD: primary dysmenorrhea; XFZY: Xuefu Zhuyu.
| Symptoms and signs | Yes | No | Score |
|---|---|---|---|
| Paina | 9 | 0 | - |
| Irritability/depression | 16 | 0 | - |
| Distending pain | 2 | 0 | - |
| Scurry pain | 6 | 0 | - |
| Chest distress | 0.5 | 0 | - |
| Lumps in body | 7 | 0 | - |
| Petechia in the tongue | 4 | 0 | - |
| Purplish tongue | 1 | 0 | - |
| Uneven pulse | 4 | 0 | - |
| Deep pulse | 2 | 0 | - |
| If the sum score ≥20, QBP can be diagnosed. | Score: | - |
Table 1 Diagnosis scale for Qi-stagnation and blood-stasis syndrome
| Symptoms and signs | Yes | No | Score |
|---|---|---|---|
| Paina | 9 | 0 | - |
| Irritability/depression | 16 | 0 | - |
| Distending pain | 2 | 0 | - |
| Scurry pain | 6 | 0 | - |
| Chest distress | 0.5 | 0 | - |
| Lumps in body | 7 | 0 | - |
| Petechia in the tongue | 4 | 0 | - |
| Purplish tongue | 1 | 0 | - |
| Uneven pulse | 4 | 0 | - |
| Deep pulse | 2 | 0 | - |
| If the sum score ≥20, QBP can be diagnosed. | Score: | - |
| Disease | Experimental group | Control group | Follow-up |
|---|---|---|---|
| SA | Routine care + XFZY oral liquid, 20 mL each dose, three times per day, for 12 weeks | Routine carea+ placebo, 20 mL each dose, three times per day, for 12 weeks | 12-week follow-up |
| TTH | XFZY oral liquid, 20 mL each dose, three times per day, for 4 weeks | Placebo, 20 mL each dose, three times per day, for 4 weeks | 8-week follow-up |
| PD | XFZY oral liquid, 20 mL each dose, three times per day, for 3 menstrual cycles (about 12 weeks) | Placebo, 20 mL each dose, three times per day, for 3 menstrual cycles (about 12 weeks) | 12-week follow-up |
Table 2 Interventions in the experimental and control groups of RCTs for QBP
| Disease | Experimental group | Control group | Follow-up |
|---|---|---|---|
| SA | Routine care + XFZY oral liquid, 20 mL each dose, three times per day, for 12 weeks | Routine carea+ placebo, 20 mL each dose, three times per day, for 12 weeks | 12-week follow-up |
| TTH | XFZY oral liquid, 20 mL each dose, three times per day, for 4 weeks | Placebo, 20 mL each dose, three times per day, for 4 weeks | 8-week follow-up |
| PD | XFZY oral liquid, 20 mL each dose, three times per day, for 3 menstrual cycles (about 12 weeks) | Placebo, 20 mL each dose, three times per day, for 3 menstrual cycles (about 12 weeks) | 12-week follow-up |
| 1. |
Ohman EM. Clinical practice Chronic stable angina. N Engl J Med 2016; 374: 1167-76.
DOI URL |
| 2. |
Walters MA. Management of chronic stable angina. Crit Care Nurs Clin North Am 2017; 29: 487-93.
DOI URL |
| 3. | Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics-2015 update: a report from the American Heart Association. Circulation 2015; 131: e29-322. |
| 4. |
Greaves K, Chen Y, Appadurai V, et al. The prevalence of doctor-diagnosed angina in 4314 older adults in China and comparison with the Rose angina questionnaire: the 4 province study. Int J Cardiol 2014; 177: 627-8.
DOI PMID |
| 5. |
Balla C, Pavasini R, Ferrari R. Treatment of angina: where are we? Cardiology 2018; 140: 52-67.
DOI PMID |
| 6. | Hu K. Study on the circRNA clinical relevant mechanism of Xuefu Zhuyu capsule intervention in patients with stable angina pectoris with Qi stagnation and blood stasis syndrome. Beijing: China Academy of Chinese Medical Sciences, 2020: 12-3. |
| 7. |
Jensen RH. Tension-type headache-the normal and most prevalent headache. Headache 2018; 58: 339-45.
DOI URL |
| 8. |
Yao C, Wang Y, Wang L, et al. Burden of headache disorders in China, 1990-2017: findings from the Global Burden of Disease Study 2017. J Headache Pain 2019; 20: 1-11.
DOI |
| 9. |
Ertsey C, Magyar M, Gyüre T, et al. Tension type headache and its treatment possibilities. Ideggyogy Sz 2019; 72: 13-21.
DOI URL |
| 10. |
Ashina S, Mitsikostas DD, Lee MJ, et al. Tension-type headache. Nat Rev Dis Primers 2021; 7: 24.
DOI PMID |
| 11. |
Bendtsen L, Evers S, Linde M, et al. EFNS guideline on the treatment of tension-type headache-report of an EFNS task force. Eur J Neurol 2010; 17: 1318-25.
DOI PMID |
| 12. |
Bendtsen L, Jensen R. Treating tension-type headache-an expert opinion. Expert Opin Pharmacother 2011; 12: 1099-109.
DOI PMID |
| 13. |
Melchart D, Hager S, Hager U, et al. Treatment of patients with chronic headaches in a hospital for Traditional Chinese Medicine in Germany. a randomised, waiting list controlled trial. Complement Ther Med 2004; 12: 71-8.
PMID |
| 14. |
Ferries-Rowe E, Corey E, Archer JS. Primary dysmenorrhea: diagnosis and therapy. Obstet Gynecol 2020; 136: 1047-58.
DOI PMID |
| 15. |
Burnett M, Lemyre M. No. 345-Primary dysmenorrhea consensus guideline. J Obstet Gynaecol Can 2017; 39: 585-95.
DOI PMID |
| 16. |
Hu Z, Tang L, Chen L, et al. Prevalence and risk factors associated with primary dysmenorrhea among Chinese female university students: a cross-sectional study. J Pediatr Adolesc Gynecol 2020; 33: 15-22.
DOI URL |
| 17. |
Koninckx PR, Ussia A, Adamyan L, et al. Primary dysmenorrhea. J Obstet Gynaecol Can 2017; 39: 578-9.
DOI PMID |
| 18. | Marjoribanks J, Ayeleke RO, Farquhar C, et al. Nonsteroidal anti-inflammatory drugs for dysmenorrhea. Cochrane Database Syst Rev 2015; 2015: CD001751. |
| 19. |
Wang Y, Zhang L, Pan Y, et al. Investigation of invigorating Qi and activating blood circulation prescriptions in treating Qi deficiency and blood stasis syndrome of ischemic stroke patients: study protocol for a randomized controlled trial. Front Pharmacol 2020; 11: 892.
DOI PMID |
| 20. |
He H, Chen G, Gao J, et al. Xuefu Zhuyu capsule in the treatment of Qi stagnation and blood stasis syndrome: a study protocol for a randomised controlled pilot and feasibility trial. Trials 2018; 19: 515.
DOI |
| 21. | Song Z, Wang ZF, Wu Y, et al. Thoughts on post-marketing evaluation of classical Chinese patent medicines based on clinical value. Zhong Guo Zhong Yao Za Zhi 2021; 46: 1988-93. |
| 22. | Shao YX. Clinical research of the treatment of Xuefu Zhuyu decoction to patients with migraine of Qi stagnancy and blood stasis syndrome. Shandong: Shandong University of Traditional Chinese Medicine, 2018: 18. |
| 23. | Li YF. Effect of Xuefu Zhuyu on primary dysmenorrhea with Qi stagnation and blood stasis. Lin Chuang He Li Yong Yao Za Zhi 2020; 13: 89-90. |
| 24. | Guo SQ, Development and application of Xuefu Zhuyu oral liquid. Jilin Zhong Yi Yao 2005; 25: 61-2. |
| 25. | National Medical Products Administration. Chinese-made medicines. updated 2020-01-14, cited 2021-07-13. Available from URL: http://app1.nmpa.gov.cn/data_nmpa/face3/base.jsp?tableId=25&tableName=TABLE25&title=%B9%FA%B2%FA%D2%A9%C6%B7&bcId=152904713761213296322795806604. |
| 26. | National Pharmacopoeia Committee. The Pharmacopoeia of the People's Republic of China 2015 Edition. Beijing: China Medical Science and Technology Press, 2015: 849. |
| 27. | Li TF, Therapeutic effect of Xuefu Zhuyu Koufuye in the treatment of primary dysmenorrhea. Shi Yong Zhong Xi Yi Jie He Lin Chuang Za Zhi 2014; 14: 27-8. |
| 28. | Wang YM. The effect of Xuefu Zhuyu decoction on thrombosis and hemorheology. Neimenggu Zhong Yi Yao 2016; 35: 108-9. |
| 29. | U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), et al. Master Protocols: Efficient clinical trial design strategies to expedite development of oncology drugs and biologics. updated 2022-03-01, cited 2022-03-22. Available from URL: https://www.fda.gov/media/120721/download. |
| 30. | Hu JY, Zhang XY, Zhao C, et al. Master protocol applied to the prevention and treatment of Traditional Chinese Medicine for coronary heart disease: ideas and methods of evidence-based optimization. Zhong Guo Xun Zheng Yi Xue Za Zhi 2019; 19: 102-6. |
| 31. | Zhao C, Zhang XY, Hu JY, et al. Master protocol design for clinical studies of diseases with the same syndromes in TCM. Zhong Yi Za Zhi 2018; 59: 111-5. |
| 32. | Liao SJ, Zhang Z, Li G, et al. Chinese herbal formula Xuefu Zhuyu for stable angina (CheruSA): study protocol for a multicenter randomized controlled trial. Evid Based Complement Alternat Med 2020; 2020: 1-10. |
| 33. | Zhou L, Zhang Z, Li G, et al. Chinese herbal formula Xuefu Zhuyu for tension-type headache with Qi-stagnation and blood-stasis pattern (CheruXTH): study protocol for a randomized controlled trial. Evid Based Complement Alternat Med 2020; 2020: 1-8. |
| 34. |
Li G, Zhang Z, Zhou L, et al. Chinese herbal formula Xuefu Zhuyu for primary dysmenorrhea patients (CheruPDYS): a study protocol for a randomized placebo-controlled trial. Trials 2021; 22: 95.
DOI PMID |
| 35. |
Dai L, Cheng CW, Tian R, et al. Standard protocol items for clinical trials with Traditional Chinese Medicine 2018: recommendations, explanation and elaboration (SPIRIT-TCM Extension 2018). Chin J Integr Med 2019; 25: 71-9.
DOI |
| 36. | Wang B, Li Y, Ling Y, Guidelines for the diagnosis and treatment of stable coronary artery disease. Zhong Hua Xin Xue Guan Bing Za Zhi 2018; 46: 680-94. |
| 37. | Gibbons RJ, Abrams J, Chatterjee K, et al. ACC/AHA 2002 guideline update for the management of patients with chronic stable angina-summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients With Chronic Stable Angina). Circulation 2003; 107: 149-58. |
| 38. |
Campeau L, Letter: Grading of angina pectoris. Circulation 1976; 54: 522-3.
PMID |
| 39. | Headache Classification Committee of the International Headache Society IHS. The international classification of headache disorders. 3rd ed. Cephalalgia 2018; 38: 1-211. |
| 40. | Wang J, Gao JL, Chen HG, et al. Constructing diagnosis scale for Qi stagnation and blood stasis syndrome. Zhong Guo Shi Yan Fang Ji Xue Za Zhi 2018; 24: 16-20. |
| 41. | Wang DH, Dong B. Retrospective analysis on the pattern/ syndrome distribution of angina pectorisincoronary heart disease. Shi Jie Zhong Xi Yi Jie He Za Zhi 2015; 10: 387-9. |
| 42. | Yu Y. Development and epidemiological investigation on the distribution and combination rule of the elements of pathogenesis and syndrome diagnosis scale for internal injury headache. Nanjing: Nanjing University of Chinese Medicine, 2015: 20. |
| 43. | Cheng F, Cheng H, Cao JH, et al. Exploration of TCM syndrome distribution characteristics of primary dysmenorrhea. Zhong Yi Xue Bao 2013; 28: 1194-6. |
| 44. | Bourdel N, Alves J, Pickering G, et al. Systematic review of endometriosis pain assessment: how to choose a scale? Hum Reprod Update 2015; 21: 136-52. |
| 45. | Liu SH. The evaluation of the reliability and validity of SQA applied in a group of Chinese patients with CHD. Tianjin: Tianjin Medical University, 2003: 33-5. |
| 46. |
Janssen MF, Pickard AS, Golicki D, et al. Measurement properties of the EQ-5D-5L compared to the EQ-5D-3L across eight patient groups: a multi-country study. Qual Life Res 2013; 22: 1717-27.
DOI PMID |
| 47. |
Cox DJ, Meyer RG. Behavioral treatment parameters with primary dysmenorrhea. J Behav Med 1978; 1: 297-310.
DOI PMID |
| 48. | WHO. The use of the WHO-UMC system for standardised case causality assessment. updated 2013-06-05, cited 2022-03-01. Available from URL: https://www.who.int/medicines/areas/quality_safety/safety_efficacy/WHOcausality_assessment.pdf. |
| 49. | ICH Harmonised Tripartite Guideline: Safety Pharmacology Studies for Human Pharmaceuticals S7A. updated 2019-05-06, cited 2022-03-01. Available from URL: https://database.ich.org/sites/default/files/S7A_Guideline.pdf. |
| 50. |
Redman MW, Allegra CJ, The master protocol concept. Semin Oncol 2015; 42: 724-30.
DOI PMID |
| 51. |
Woodcock J, LaVange LM. Master protocols to study multiple therapies, multiple diseases, or both. N Engl J Med 2017; 377: 62-70.
DOI URL |
| 52. |
Cheng CW, Wu TX, Shang HC, et al. CONSORT Extension for Chinese herbal medicine formulas 2017: recommendations, explanation, and elaboration (Traditional Chinese version). Ann Intern Med 2017; 167: W7-20.
DOI URL |
| 53. |
Zhai X, Wang X, Wang L, et al. Treating different diseases with the same method-a Traditional Chinese Medicine concept analyzed for its biological basis. Front Pharmacol 2020; 11: 946.
DOI PMID |
| 54. |
Fields T, Patterson M, Bremova-ErtlT, et al. A master protocol to investigate a novel therapy acetyl-L-leucine for three ultra-rare neurodegenerative diseases: Niemann-Pick type C, the GM2 gangliosidoses, and ataxia telangiectasia. Trials 2021; 22: 84.
DOI PMID |
| 55. |
Slobogean GP, Sprague S, Wells J, et al. Effectiveness of iodophor vs chlorhexidine solutions for surgical site infections and unplanned reoperations for patients who underwent fracture repair. JAMA Netw Open 2020; 3: e202215.
DOI URL |
| [1] | YANG Yi, YE Huijun, ZHENG Huiling, JIN Lihua. Clinical observation on 90 cases of primary dysmenorrhea treated by buccal acupuncture therapy: a randomized controlled study [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 172-181. |
| [2] | Jingjing WEI, Rongjuan GUO, Guojing FU, Xiao LIANG, Zhenmin XU, Min JIA, Zixiu ZENG, Wanqing DU, Weiwei JIAO, Linjuan SUN, Hongmei LIU, Chunli GUO, Chenguang TONG, Yunling ZHANG, Xing LIAO. Registration of intervention trials of Traditional Chinese Medicine for four neurological diseases on Chinese Clinical Trial Registry and ClinicalTrials.gov: a narrative review [J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 148-153. |
| [3] | Hu Bing, Zhang Tong, An Hongmei, Zheng Jialu, Yan Xia, Huang Xiaowei, Tian Jianhui, Li Miao. Effect of Yanggan Jiedu Sanjie formula on human hepatocellular carcinoma Bel-7402 cells [J]. Journal of Traditional Chinese Medicine, 2019, 39(01): 26-33. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

