Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (2): 295-302.DOI: 10.19852/j.cnki.jtcm.20221108.002
• Original articles • Previous Articles Next Articles
Efficacy of Baishao Luoshi decoction (白芍络石方) on synaptic plasticity in rats with post stroke spasticity
XIE Le1, MAO Guo1, XIE Yao1, CAO Sijia2, ZHOU Shen1, JIANG Junlin1, YAO Ting1, FAN Jianhu1, LIU Dong3, KANG Fuliang1, WU Dahua1( ), GE Jinwen1,4(
), GE Jinwen1,4( )
)
- 1 Neurology Department, Hunan Academy of Chinese Medicine, Changsha 410006, China
2 School of Humanities and Management, Hunan University of Chinese Medicine, Changsha 410208, China
3 College of pharmacy, Guizhou University of Traditional Chinese Medicine, Guiyang 550002, China
4 College of Integrated Traditional Chinese and Western Medicine, Hunan University of Chinese Medicine, Changsha 410208, China
-
Received:2021-11-18Accepted:2022-02-16Online:2023-04-15Published:2023-03-14 -
Contact:Prof. WU Dahua, Neurology Department, Hunan Academy of Chinese Medicine Affiliated Hospital, Changsha 410006, China. 893049352@qq.com; Prof. GE Jinwen, College of Integrated Traditional Chinese and Western Medicine, Hunan University of Chinese Medicine, Changsha 410208, China 001267@hnucm.edu.cn. Telephone: +86-13508491768; +86-13873172948 -
Supported by:China Postdoctoral Science Foundation: Research on the Mechanism of Baishaoluoshi Decoction in Improving Post-stroke Spasticity by KCC2-GABAAR Based on the Theory of “Gan Zhu Jin”(2020M682571);Natural Science Foundation of Hunan Province China: Research on the Mechanism of Post-stroke Spasticity by KCC2--GABAAR-Motoneuron Excitability and Chinese Traditional Medicine Intervention Based on the Theory of “Gan Zhu Jin”(2019JJ50343);Scientific Research Project of Education Department of Hunan Province: Research on the Mechanism of Axonal Regeneration and TCM Intervention in Post-Stroke Spasticity Based on the Theory of “Gan Zhu Jin” by Slit-Robo Pathway(19C1423)
Cite this article
XIE Le, MAO Guo, XIE Yao, CAO Sijia, ZHOU Shen, JIANG Junlin, YAO Ting, FAN Jianhu, LIU Dong, KANG Fuliang, WU Dahua, GE Jinwen. Efficacy of Baishao Luoshi decoction (白芍络石方) on synaptic plasticity in rats with post stroke spasticity[J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 295-302.
share this article
| Test item | Score |
|---|---|
| Motor function | |
| Raising the rat by the tail: | |
| Flexion of forelimb or hindlimb | 1 |
| Head moved more than 10 ℃ to the vertical axis within 30 s | 1 |
| Walking on the ground | |
| Normal walking | 0 |
| Circling toward the paretic side | 1 |
| Fall down to the paretic side | 2 |
| Abnormal movement | |
| Immobility, staring, tremor and pilo-erection | 1 |
| Myodystony and irritability | 1 |
| Sensory function | |
| Ipsilateral forelimb retracts after acupuncture | 0 |
| Ipsilateral forelimb does not retract after acupuncture | 1 |
| Reflex deficiency | |
| Pinna reflex (head shake when touching the external auditory meatus) | 1 |
| Corneal reflex (blink when lightly touching the cornea with cotton) | 1 |
| Startle reflex (Motor response to sudden appearance of the noise) | 1 |
| Total score | 11 |
Table 1 Modified neurological severity score
| Test item | Score |
|---|---|
| Motor function | |
| Raising the rat by the tail: | |
| Flexion of forelimb or hindlimb | 1 |
| Head moved more than 10 ℃ to the vertical axis within 30 s | 1 |
| Walking on the ground | |
| Normal walking | 0 |
| Circling toward the paretic side | 1 |
| Fall down to the paretic side | 2 |
| Abnormal movement | |
| Immobility, staring, tremor and pilo-erection | 1 |
| Myodystony and irritability | 1 |
| Sensory function | |
| Ipsilateral forelimb retracts after acupuncture | 0 |
| Ipsilateral forelimb does not retract after acupuncture | 1 |
| Reflex deficiency | |
| Pinna reflex (head shake when touching the external auditory meatus) | 1 |
| Corneal reflex (blink when lightly touching the cornea with cotton) | 1 |
| Startle reflex (Motor response to sudden appearance of the noise) | 1 |
| Total score | 11 |
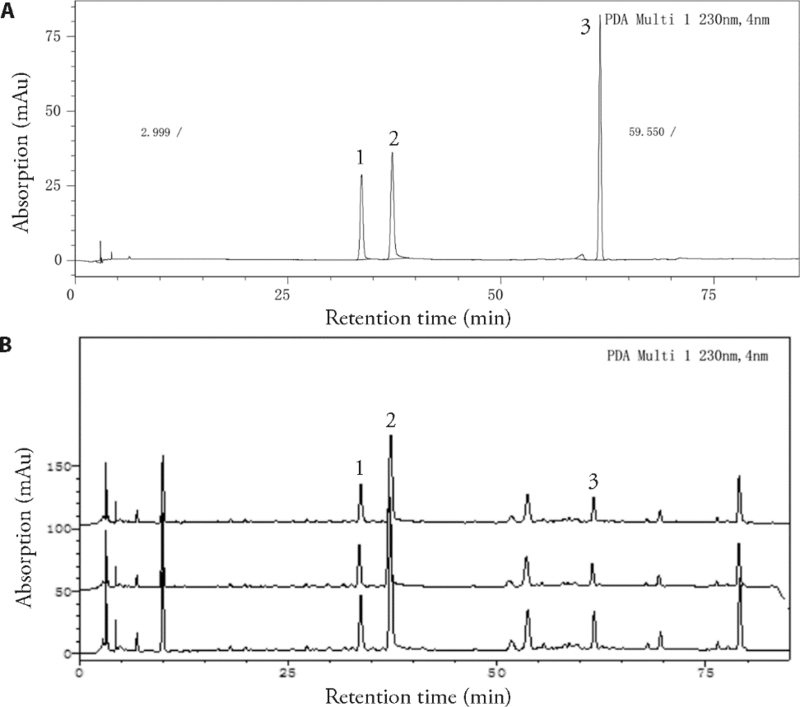
Figure 1 Chromatogram of BD established by HPLC A: reference sample of paeoniflorin, albiflorin and tracheloside, the main component of BD [Baishao (Baishao (Radix Paeoniae Alba) and Luoshiteng (Caulis Trachelospermi Jasminoidis) at a ratio of 1∶1]. Peaks 1-3 represented paeoniflorin, albiflorin and tracheloside respectively; B: HPLC chromatogram of three different batches of BD formula granules [Baishao (Radix Paeoniae Alba) (No. 16050101, 15090121, 16010069) and Luoshiteng (Caulis Trachelospermi Jasminoidis) (No. 15110003, 16082053, 16050059)]. BD: Baishaoluoshi decoction; HPLC: high performance liquid chromatography.

Figure 2 Neuroprotective effects of BD on PSS rats A: comparison of weight in each group at different time. Weight of rats in each group were detected at different time, 3 d after model established, 1, 2, 3, and 4 weeks after intervention; B: comparison of mNSS in each group at different time. mNSS was used to evaluate neurological deficits at different time, 3 d after model established, 1, 2, 3, and 4 weeks after intervention; C: comparison of MAS in each group at different time. MAS was used to evaluate spasticity of the affected fore limb 3 d after model established, 1, 2, 3, and 4 weeks after intervention. The PSS model was established by MCAO, and intervention began 3 d after model established for 4 weeks. Rats were daily treated orally with BD (10.8 g/kg in H and 5.4 g/kg in L) or baclofen (5.4 mg/kg in B), or normal saline (M, S, C) by gavage for 4 weeks. H: high dose BD group (10.8 g/kg); L: low dose BD group (5.4 g/kg); B: baclofen group (5.4 mg/kg); M: model group (normal saline); S: sham-operated group (normal saline); C: control group (normal saline). BD: Baishaoluoshi decoction; PSS: post stroke spasticity; mNSS: modified neurological deficit score; MAS: Modified Ashworth score; MCAO: middle cerebral artery occlusion. aP < 0.01 and bP < 0.05 as compared to model group (n = 12).

Figure 3 Effects of BD on the synaptic structure in PSS rats A: synaptic structure observed by TEM (×20 000). The red arrows represent synapse; B: comparison of PSD in the brain tissue around the infarcts; C: comparison of synaptic curvature in the brain tissue around the infarcts; D: comparison of synaptic gap in the brain tissue around the infarcts. Brain tissue around the infarct was stained by uranium acetate and lead citrate, and observed by TEM and the morphology of Gray 1 synapse was analyzed by image-pro Plus 6.0, 4 weeks after intervention. A1: high dose BD group (10.8 g/kg); A2: low dose BD group (5.4 g/kg); A3: baclofen group (5.4 mg/kg); A4: model group (normal saline); A5: sham-operated group (normal saline); A6: control group (normal saline). BD: Baishaoluoshi decoction; PSS: post stroke spasticity; TEM: transmission electron microscopy; PSD: postsynaptic density. bP < 0.05 and cP < 0.01 as compared to control group; dP < 0.05 and aP < 0.01 as compared to model group (n = 3).

Figure 4 Effects of BD on synaptic plasticity associated protein in PSS rats A: protein expression levels of BDNF, GAP43, p38 and MAP2 in brain tissue around the infarct were detected by Western blot analysis, 4 weeks after intervention. β-actin was used as the internal control. B: the relative expression of BDNF, C: the relative expression of GAP43, D: the relative expression of p38, E: the relative expression of MAP2: Densitometric analysis. H: high dose BD group (10.8 g/kg), L: low dose BD group (5.4 g/kg), B: baclofen group (5.4 mg/kg), M: model group (normal saline), S: sham-operated group (normal saline), and C: control group (normal saline). BD: Baishaoluoshi decoction; PSS: post stroke spasticity; BDNF: brain derived neurotrophic factor; GAP43: growth associated protein-43; MAP2: microtubule-associated protein 2. aP < 0.01, cP < 0.05, as compared to control group; bP < 0.01 and dP < 0.05, as compared to model group (n = 6).
| 1. |
Zhou MG, Wang HD, Zeng XY, et al. Mortality, morbidity, and risk factors in China and its provinces, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019; 394: 1145-58.
DOI PMID |
| 2. | Kuo CL, Hu GC. Post-stroke spasticity: a review of epidemiology, pathophysiology, and treatments. Int J Gerontol 2018; 4: 280-4. |
| 3. |
Schinwelski MJ, Sitek EJ, Waz P, et al. Prevalence and predictors of post-stroke spasticity and its impact on daily living and quality of life. Neurol Neurochir Pol 2019; 53: 449-57.
DOI PMID |
| 4. | Wissel J, Verrier M, Simpson DM, et al. Post-stroke spasticity: predictors of early development and considerations for therapeutic intervention. PMR 2015; 7: 60-7. |
| 5. |
Bakheit AMO. The Pharmacological management of post-stroke muscle spasticity. Drugs Aging 2012; 29: 941-7.
DOI PMID |
| 6. | Civelek GM, Atalay A. Treatment of spasticity in cerebral palsy. Guncel Pediatri 2016; 14: 136-41. |
| 7. | Jacinto J, Lysandropoulos A, Fulford-Smith A. Perceptions of burden of spasticity and treatment satisfaction among post-stroke patients over the course of a botulinum neurotoxin A (BoNT-A) treatment cycle: an ethnographic study. Mov Disord 2020; 35: S589. |
| 8. | Albrecht P, Jansen A, Lee JI, et al. High prevalence of neutralizing antibodies after long-term botulinum neurotoxin therapy. Neurol 2019; 92: E48-54. |
| 9. |
Akturk S, Buyukavci R, Ersoy Y. Functional outcomes following ultrasound-guided botulinum toxin type A injections to reduce spastic equinovarus in adult post-stroke patients. Toxicon 2018; 146: 95-8.
DOI PMID |
| 10. |
Cai YY, Zhang CS, Liu SN, et al. Add-On Effects of Chinese herbal medicine for post-stroke spasticity: a systematic review and Meta-analysis. Front Pharmacol 2019; 10: 1-20.
DOI URL |
| 11. |
Zhang JY, Zhu LW, Tang Q. Electroacupuncture with rehabilitation training for limb spasticity reduction in post-stroke patients: a systematic review and Meta-analysis. Top Stroke Rehabil 2021; 28: 340-61.
DOI URL |
| 12. | Xie L, Wu D, Fang R, Zhang F, Li H, Zhou S. Effect of Baishao Luoshi decoction on spasticity after ischemic stroke. Hunan Zhong Yi Yao Da Xue Xue Bao 2018; 38: 77-81. |
| 13. |
Zhu XQ, Hu HX, Li ZF, et al. Gua Lou Gui Zhi decoction attenuates post-stroke spasticity via the modulation of GABA(B) receptors. Mol Med Rep 2015; 12: 5957-62.
DOI URL |
| 14. |
Pan JJ, Qu MJ, Li YF, et al. MicroRNA-126-3p/-5p overexpression attenuates blood-brain barrier disruption in a mouse model of middle cerebral artery occlusion. Stroke 2020; 51: 619-27.
DOI PMID |
| 15. |
Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther 1987; 67: 206-7.
DOI PMID |
| 16. |
Li GY, Yuan WC, Liu GL, et al. Effects of radial extracorporeal shockwave therapy on spasticity of upper-limb agonist/antagonist muscles in patients affected by stroke: a randomized, single-blind clinical trial. Age Ageing 2020; 49: 246-52.
DOI PMID |
| 17. | Lourenco MV, Frozza RL, de Freitas GB, et al. Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer's models. Nat Med 2019; 25: 165-75. |
| 18. |
Nanou E, Catterall WA. Calcium channels, synaptic plasticity, and neuropsychiatric disease. Neuron 2018; 98: 466-81.
DOI PMID |
| 19. |
Bethoux F. Spasticity management after stroke. Phys Med Rehabil Clin N Am 2015; 26: 625-39.
DOI PMID |
| 20. |
Bayes A, van de Lagemaat LN, Collins MO, et al. Characterization of the proteome, diseases and evolution of the human postsynaptic density. Nat Neurosci 2011; 14: 19-21.
DOI PMID |
| 21. |
Bai GH, Wang Y, Zhang MJ. Gephyrin-mediated formation of inhibitory postsynaptic density sheet via phase separation. Cell Res 2021; 31: 312-25.
DOI |
| 22. | Ranneva SV, Maksimov VF, Korostyshevskaja IM, et al. Lack of synaptic protein, calsyntenin-2, impairs morphology of synaptic complexes in mice. Synapse 2020; 74: 1-9. |
| 23. | De Vincenti AP, Rios AS, Paratcha G, et al. Mechanisms that modulate and diversify BDNF functions: implications for hippo-campal synaptic plasticity. Front Cell Neurosci 2019; 13: 1-7. |
| 24. |
Li QF, Wu XF, Na XY, et al. Impaired cognitive function and altered hippocampal synaptic plasticity in mice lacking dermatan sulfotransferase Chst14/D4st1. Front Mol Neurosci 2019; 12: 1-10.
DOI URL |
| 25. |
Kowianski P, Lietzau G, Czuba E, et al. BDNF: a key factor with multipotent impact on brain signaling and synaptic plasticity. Cell Mol Neurobiol 2018; 38: 579-93.
DOI PMID |
| 26. |
Parcerisas A, Pujadas L, Ortega-Gasco A, et al. NCAM2 regulates dendritic and axonal differentiation through the cytoskeletal proteins MAP2 and 14-3-3. Cereb Cortex 2020; 30: 3781-99.
DOI PMID |
| 27. |
Hua HY, Zhang WY, Li JY, et al. Neuroprotection against cerebral ischemia/reperfusion by dietary phytochemical extracts from Tibetan turnip (Brassica rapa L.). J Ethnopharmacol 2021; 265: 113410.
DOI URL |
| 28. |
Tamakoshi K, Kawanaka K, Onishi H, et al. Motor skills training improves sensorimotor dysfunction and increases microtubule-associated protein 2 mRNA expression in rats with intracerebral hemorrhage. J Stroke Cerebrovasc Dis 2016; 25: 2071-7.
DOI PMID |
| 29. |
Li B, Wang ZJ, Yu M, et al. miR-22-3p enhances the intrinsic regenerative abilities of primary sensory neurons via the CBL/p-EGFR/p-STAT3/GAP43/p-GAP43 axis. J Cell Physiol 2020; 235: 4605-17.
DOI URL |
| 30. | Wang JY, Li HZ, Yao Y, et al. Beta-elemene enhances GAP-43 expression and neurite outgrowth by inhibiting RhoA kinase activation in rats with spinal cord injury. Neurosci 2018; 383: 12-21. |
| 31. |
Mascaro ALA, Cesare P, Sacconi L, et al. In vivo single branch axotomy induces GAP-43-dependent sprouting and synaptic remodeling in cerebellar cortex. Proc Natl Acad Sci USA 2013; 110: 10824-9.
DOI URL |
| 32. |
Castagna C, Merighi A, Lossi L. Decreased expression of synaptophysin 1 (SYP 1 major synaptic vesicle protein p38) and contactin 6 (CNTN6/NB3) in the cerebellar vermis of reln haplodeficient mice. Cell Mol Neurobiol 2019; 39: 833-56.
DOI PMID |
| 33. |
Maphis N, Jiang SY, Xu GX, et al. Selective suppression of the alpha isoform of p38 MAPK rescues late-stage tau pathology. Alzheimers Res Ther 2016; 8: 1-15.
DOI URL |
| 34. |
Liu ZW, Chopp M, Ding XS, et al. Axonal remodeling of the corticospinal tract in the spinal cord contributes to voluntary motor recovery after stroke in adult mice. Stroke 2013; 44: 1951-6.
DOI PMID |
| [1] | Esma Anissa Trad Khodja, Abd El Hamid Khabtane, Rabah Arhab, Djamila Benouchenne, Mohamed Sabri Bensaad, Chawki Bensouici, Ramazan Erenler. In vitro assessment of antioxidant, neuroprotective, anti-urease and anti-tyrosinase capacities of Tamarix africana leaves extracts [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 252-264. |
| [2] | Bhakta Prasad Gaire, Hocheol Kim. Neuroprotective effects of Fructus Chebulae extracts on experimental models of cerebral ischemia [J]. Journal of Traditional Chinese Medicine, 2014, 34(01): 69-75. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

